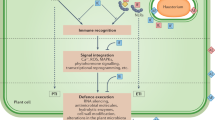
Abstract
Xanthomonas spp. encompass a wide range of plant pathogens that use numerous virulence factors for pathogenicity and fitness in plant hosts. In this Review, we examine recent insights into host–pathogen co-evolution, diversity in Xanthomonas populations and host specificity of Xanthomonas spp. that have substantially improved our fundamental understanding of pathogen biology. We emphasize the virulence factors in xanthomonads, such as type III secreted effectors including transcription activator-like effectors, type II secretion systems, diversity resulting in host specificity, evolution of emerging strains, activation of susceptibility genes and strategies of host evasion. We summarize the genomic diversity in several Xanthomonas spp. and implications for disease outbreaks, management strategies and breeding for disease resistance.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ryan, R. P. et al. Pathogenomics of Xanthomonas: understanding bacterium–plant interactions. Nat. Rev. Microbiol. 9, 344–355 (2011).
Jacques, M.-A. et al. Using ecology, physiology, and genomics to understand host specificity in Xanthomonas. Ann. Rev. Phytopathol. 54, 163–187 (2016).
Quezado-Duval, A. M., Leite Jr, R. P., Truffi, D. & Camargo, L. E. Outbreaks of bacterial spot caused by Xanthomonas gardneri on processing tomato in central-west Brazil. Plant Dis. 88, 157–161 (2004).
Babadoost, M. & Ravanlou, A. Outbreak of bacterial spot (Xanthomonas cucurbitae) in pumpkin fields in Illinois. Plant Dis. 96, 1222–1222 (2012).
Nakato, V., Mahuku, G. & Coutinho, T. Xanthomonas campestris pv. musacearum: a major constraint to banana, plantain and enset production in central and east Africa over the past decade. Mol. Plant Pathol. 19, 525–536 (2018).
Tripathi, L. et al. Xanthomonas wilt: a threat to banana production in east and central Africa. Plant Dis. 93, 440–451 (2009).
Shimwela, M. M. et al. Banana Xanthomonas wilt continues to spread in Tanzania despite an intensive symptomatic plant removal campaign: an impending socio-economic and ecological disaster. Food Sec. 8, 939–951 (2016).
Constantin, E. C. et al. Genetic characterization of strains named as Xanthomonas axonopodis pv. dieffenbachiae leads to a taxonomic revision of the X. axonopodis species complex. Plant Pathol. 65, 792–806 (2016).
Timilsina, S. et al. Reclassification of Xanthomonas gardneri (ex Šutič 1957) Jones et al. 2006 as a later heterotypic synonym of Xanthomonas cynarae Trébaol et al. 2000 and description of X. cynarae pv. cynarae and X. cynarae pv. gardneri based on whole genome analyses. Int. J. Syst. Evol. Micr. 69, 343–349 (2019).
Rademaker, J. L. W. et al. A comprehensive species to strain taxonomic framework for Xanthomonas. Phytopathology 95, 1098–1111 (2005).
Leibniz Institute DSMZ–German Collection of Microorganisms and Cell Cultures GmbH. Prokaryotic Nomenclature Up-to-date. https://www.dsmz.de/services/online-tools/prokaryotic-nomenclature-up-to-date/prokaryotic-nomenclature-up-to-date/genus/516930 (2019).
Poplawsky, A. R. A xanthomonadin-encoding gene cluster for the identification of pathovars of Xanthomonas campestris. Mol. Plant Microbe Interact. 6, 545 (1993).
Midha, S. & Patil, P. B. Genomic insights into the evolutionary origin of Xanthomonas axonopodis pv. citri and its ecological relatives. Appl. Environ. Microbiol. 80, 6266–6279 (2014).
Ferreira, M. A. S. V. et al. Xanthomonas citri pv. viticola affecting grapevine in Brazil: emergence of a successful monomorphic pathogen. Front. Plant Sci. 10, 489 (2019).
Pruvost, O., Couteau, A., Perrier, X. & Luisetti, J. Phenotypic diversity of Xanthomonas sp. mangiferaeindicae. J. Appl. Microbiol. 84, 115–124 (1998).
An, S.-Q. et al. Mechanistic insights into host adaptation, virulence and epidemiology of the phytopathogen Xanthomonas. FEMS Microbiol. Rev. 44, 1–32 (2019).
Zhang, H. & Wang, S. Rice versus Xanthomonas oryzae pv. oryzae: a unique pathosystem. Curr. Opin. Plant Biol. 16, 188–195 (2013).
Cesbron, S. et al. Comparative genomics of pathogenic and nonpathogenic strains of Xanthomonas arboricola unveil molecular and evolutionary events linked to pathoadaptation. Front. Plant Sci. 6, 1126 (2015).
Timilsina, S. et al. Multiple recombination events drive the current genetic structure of Xanthomonas perforans in Florida. Front. Microbiol. 10, 448 (2019).
Huang, C.-L. et al. Ecological genomics in Xanthomonas: the nature of genetic adaptation with homologous recombination and host shifts. BMC Genomics 16, 188 (2015).
Merda, D. et al. Recombination-prone bacterial strains form a reservoir from which epidemic clones emerge in agroecosystems. Environ. Microbiol. Rep. 8, 572–581 (2016).
Newberry, E. A. et al. Independent evolution with the gene flux originating from multiple Xanthomonas species explains genomic heterogeneity in Xanthomonas perforans. Appl. Environ. Microbiol. 85, e00885-19 (2019).
Hsiao, Y.-M., Liao, H.-Y., Lee, M.-C., Yang, T.-C. & Tseng, Y.-H. Clp upregulates transcription of engA gene encoding a virulence factor in Xanthomonas campestris by direct binding to the upstream tandem Clp sites. FEBS Lett. 579, 3525–3533 (2005).
Constantin, E. C. et al. Pathogenicity and virulence gene content of Xanthomonas strains infecting Araceae, formerly known as Xanthomonas axonopodis pv. dieffenbachiae. Plant Pathol. 66, 1539–1554 (2017).
Rossier, O., Wengelnik, K., Hahn, K. & Bonas, U. The Xanthomonas Hrp type III system secretes proteins from plant and mammalian bacterial pathogens. Proc. Natl Acad. Sci. USA 96, 9368–9373 (1999).
White, F. F., Potnis, N., Jones, J. B. & Koebnik, R. The type III effectors of Xanthomonas. Mol. Plant Pathol. 10, 749–766 (2009).
Teper, D. et al. Identification of novel Xanthomonas euvesicatoria type III effector proteins by a machine-learning approach. Mol. Plant Pathol. 17, 398–411 (2016).
Grau, J. et al. AnnoTALE: bioinformatics tools for identification, annotation, and nomenclature of TALEs from Xanthomonas genomic sequences. Sci. Rep. 6, 21077 (2016).
Sgro, G. G. et al. Bacteria-killing type IV secretion systems. Front. Microbiol. 10, 1078 (2019).
Szczesny, R. et al. Functional characterization of the Xcs and Xps type II secretion systems from the plant pathogenic bacterium Xanthomonas campestris pv vesicatoria. N. Phytol. 187, 983–1002 (2010).
Potnis, N. et al. Comparative genomics reveals diversity among xanthomonads infecting tomato and pepper. BMC Genomics 12, 146 (2011).
Weiberg, A. & Jin, H. Small RNAs—the secret agents in the plant–pathogen interactions. Curr. Opin. Plant Biol. 26, 87–94 (2015).
Schmidtke, C. et al. Genome-wide transcriptome analysis of the plant pathogen Xanthomonas identifies sRNAs with putative virulence functions. Nucleic Acids Res. 40, 2020–2031 (2012).
Büttner, D. & Bonas, U. Regulation and secretion of Xanthomonas virulence factors. FEMS Microbiol. Rev. 34, 107–133 (2010).
Russell, A. B., Peterson, S. B. & Mougous, J. D. Type VI secretion system effectors: poisons with a purpose. Nat. Rev. Microbiol. 12, 137–148 (2014).
Silva, A. C. R. da et al. Comparison of the genomes of two Xanthomonas pathogens with differing host specificities. Nature 417, 459–463 (2002).
Bogdanove, A. J. et al. Two new complete genome sequences offer insight into host and tissue specificity of plant pathogenic Xanthomonas spp. J. Bacteriol. 193, 5450–5464 (2011).
Richard, D. et al. Complete genome sequences of six copper-resistant Xanthomonas citri pv. citri strains causing asiatic citrus canker, obtained using long-read technology. Genome Announc. 5, e00010-17 (2017).
Pieretti, I. et al. The complete genome sequence of Xanthomonas albilineans provides new insights into the reductive genome evolution of the xylem-limited Xanthomonadaceae. BMC Genomics 10, 616 (2009).
Pieretti, I. et al. Genomic insights into strategies used by Xanthomonas albilineans with its reduced artillery to spread within sugarcane xylem vessels. BMC Genomics 13, 658 (2012).
Pieretti, I. et al. What makes Xanthomonas albilineans unique amongst xanthomonads? Front. Plant. Sci. 6, 289 (2015).
Mhedbi-Hajri, N. et al. Evolutionary history of the plant pathogenic bacterium Xanthomonas axonopodis. PLOS ONE 8, e58474 (2013).
Schwartz, A. R. et al. Phylogenomics of Xanthomonas field strains infecting pepper and tomato reveals diversity in effector repertoires and identifies determinants of host specificity. Front. Microbiol. 6, 535 (2015).
Kałużna, M., Pulawska, J., Waleron, M. & Sobiczewski, P. The genetic characterization of Xanthomonas arboricola pv. juglandis, the causal agent of walnut blight in Poland. Plant Pathol. 63, 1404–1416 (2014).
Garita-Cambronero, J., Palacio-Bielsa, A., López, M. M. & Cubero, J. Pan-genomic analysis permits differentiation of virulent and non-virulent strains of Xanthomonas arboricola that cohabit Prunus spp. and elucidate bacterial virulence factors. Front. Microbiol. 8, 573 (2017).
Jacobs, J. M., Pesce, C., Lefeuvre, P. & Koebnik, R. Comparative genomics of a cannabis pathogen reveals insight into the evolution of pathogenicity in Xanthomonas. Front. Plant Sci. 6, 431 (2015).
Timilsina, S. et al. Multilocus sequence analysis of xanthomonads causing bacterial spot of tomato and pepper plants reveals strains generated by recombination among species and recent global spread of Xanthomonas gardneri. Appl. Environ. Microbiol. 81, 1520–1529 (2015).
Jibrin, M. O. et al. Genomic inference of recombination-mediated evolution in Xanthomonas euvesicatoria and X. perforans. Appl. Environ. Microbiol. 84, e00136-18 (2018).
Quibod, I. L. et al. The green revolution shaped the population structure of the rice pathogen Xanthomonas oryzae pv. oryzae. ISME J. 14, 492–505 (2019).
Ochiai, H., Inoue, Y., Takeya, M., Sasaki, A. & Kaku, H. Genome sequence of Xanthomonas oryzae pv. oryzae suggests contribution of large numbers of effector genes and insertion sequences to its race diversity. Jpn. Agric. Res. Q. 39, 275–287 (2005).
Rajeshwari, R. & Sonti, R. V. Stationary-phase variation due to transposition of novel insertion elements in Xanthomonas oryzae pv. oryzae. J. Bacteriol. 182, 4797–4802 (2000).
Boch, J. & Bonas, U. Xanthomonas AvrBs3 family-type III effectors: discovery and function. Ann. Rev. Phytopathol. 48, 419–436 (2010).
Gochez, A. M. et al. Pacbio sequencing of copper-tolerant Xanthomonas citri reveals presence of a chimeric plasmid structure and provides insights into reassortment and shuffling of transcription activator-like effectors among X. citri strains. BMC Genomics 19, 16 (2018).
Escalon, A. et al. Variations in type III effector repertoires, pathological phenotypes and host range of Xanthomonas citri pv. citri pathotypes. Mol. Plant Pathol. 14, 483–496 (2013).
Gordon, J. L. et al. Comparative genomics of 43 strains of Xanthomonas citri pv. citri reveals the evolutionary events giving rise to pathotypes with different host ranges. BMC Genomics 16, 1098 (2015).
Rybak, M., Minsavage, G. V., Stall, R. E. & Jones, J. B. Identification of Xanthomonas citri ssp. citri host specificity genes in a heterologous expression host. Mol. Plant Pathol. 10, 249–262 (2009).
Patané, J. S. L. et al. Origin and diversification of Xanthomonas citri subsp. citri pathotypes revealed by inclusive phylogenomic, dating, and biogeographic analyses. BMC Genomics 20, 700 (2019).
Roman-Reyna, V. et al. The rice leaf microbiome has a conserved community structure controlled by complex host–microbe interactions. Preprint at bioRxiv. https://doi.org/10.1101/615278 (2019).
Zhang, J. et al. Insights into endophytic bacterial community structures of seeds among various Oryza sativa L. rice genotypes. J. Plant Growth Regul. 38, 93–102 (2019).
Merda, D. et al. Ancestral acquisitions, gene flow and multiple evolutionary trajectories of the type three secretion system and effectors in Xanthomonas plant pathogens. Mol. Ecol. 26, 5939–5952 (2017).
Baptista, J. C. et al. Mutation in the xpsD gene of Xanthomonas axonopodis pv. citri affects cellulose degradation and virulence. Genet. Mol. Biol. 33, 146–153 (2010).
Bayer-Santos, E. et al. Xanthomonas citri T6SS mediates resistance to Dictyostelium predation and is regulated by an ECF σ factor and cognate Ser/Thr kinase. Environ. Microbiol. 20, 1562–1575 (2018).
Bayer-Santos, E., Ceseti, L. de M., Farah, C. S. & Alvarez-Martinez, C. E. Distribution, function and regulation of type 6 secretion systems of Xanthomonadales. Front. Microbiol. 10, 1635 (2019).
Shrivastava, S. & Mande, S. S. Identification and functional characterization of gene components of type VI secretion system in bacterial genomes. PLOS ONE 3, e2955 (2008).
Records, A. R. The type VI secretion system: a multipurpose delivery system with a phage-like machinery. Mol. Plant Microbe Interact. 24, 751–757 (2011).
Boyer, F., Fichant, G., Berthod, J., Vandenbrouck, Y. & Attree, I. Dissecting the bacterial type VI secretion system by a genome wide in silico analysis: what can be learned from available microbial genomic resources? BMC Genomics 10, 104 (2009).
Kay, S. & Bonas, U. How Xanthomonas type III effectors manipulate the host plant. Curr. Opin. Microbiol. 12, 37–43 (2009).
Mudgett, M. B. et al. Molecular signals required for type III secretion and translocation of the Xanthomonas campestris AvrBs2 protein to pepper plants. Proc. Natl Acad. Sci. USA 97, 13324–13329 (2000).
Roden, J., Eardley, L., Hotson, A., Cao, Y. & Mudgett, M. B. Characterization of the Xanthomonas AvrXv4 effector, a SUMO protease translocated into plant cells. Mol. Plant. Microbe Interact. 17, 633–643 (2004).
Xia, J., Hu, X., Shi, F., Niu, X. & Zhang, C. Support vector machine method on predicting resistance gene against Xanthomonas oryzae pv. oryzae in rice. Expert. Syst. Appl. 37, 5946–5950 (2010).
Midha, S. et al. Population genomic insights into variation and evolution of Xanthomonas oryzae pv. oryzae. Sci. Rep. 7, 1–13 (2017).
Sinha, D., Gupta, M. K., Patel, H. K., Ranjan, A. & Sonti, R. V. Cell wall degrading enzyme induced rice innate immune responses are suppressed by the type 3 secretion system effectors XopN, XopQ, XopX and XopZ of Xanthomonas oryzae pv. oryzae. PLOS ONE 8, e75867 (2013).
Teper, D. et al. The Xanthomonas euvesicatoria type III effector XopAU is an active protein kinase that manipulates plant MAP kinase signaling. PLOS Pathog. 14, e1006880 (2018).
Mondal, K. K. et al. Pathotyping and genetic screening of type III effectors in Indian strains of Xanthomonas oryzae pv. oryzae causing bacterial leaf blight of rice. Physiol. Mol. Plant Pathol. 86, 98–106 (2014).
Roux, B. et al. Genomics and transcriptomics of Xanthomonas campestris species challenge the concept of core type III effectome. BMC Genomics 16, 975 (2015).
Üstün, S. & Börnke, F. Interactions of Xanthomonas type-III effector proteins with the plant ubiquitin and ubiquitin-like pathways. Front. Plant Sci. 5, 736 (2014).
Sonnewald, S. et al. Regulation of cell wall-bound invertase in pepper leaves by Xanthomonas campestris pv. vesicatoria type three effectors. PLOS ONE 7, e51763 (2012).
Feng, F. & Zhou, J.-M. Plant–bacterial pathogen interactions mediated by type III effectors. Curr. Opin. Plant Biol. 15, 469–476 (2012).
Stall, R. E., Jones, J. B. & Minsavage, G. V. Durability of resistance in tomato and pepper to xanthomonads causing bacterial spot. Annu. Rev. Phytopathol. 47, 265–284 (2009).
Han, S. W. & Hwang, B. K. Molecular functions of Xanthomonas type III effector AvrBsT and its plant interactors in cell death and defense signaling. Planta 245, 237–253 (2017).
Teper, D. et al. Xanthomonas euvesicatoria type III effector XopQ interacts with tomato and pepper 14–3–3 isoforms to suppress effector-triggered immunity. Plant J. 77, 297–309 (2014).
Szurek, B., Marois, E., Bonas, U. & Ackerveken, G. V. den. Eukaryotic features of the Xanthomonas type III effector AvrBs3: protein domains involved in transcriptional activation and the interaction with nuclear import receptors from pepper. Plant J. 26, 523–534 (2001).
Song, C. & Yang, B. Mutagenesis of 18 type III effectors reveals virulence function of XopZ(PXO99) in Xanthomonas oryzae pv. oryzae. Mol. Plant Microbe Interact. 23, 893–902 (2010).
Schulze, S. et al. Analysis of new type III effectors from Xanthomonas uncovers XopB and XopS as suppressors of plant immunity. N. Phytol. 195, 894–911 (2012).
Popov, G., Fraiture, M., Brunner, F. & Sessa, G. Multiple Xanthomonas euvesicatoria type III effectors inhibit flg22-triggered immunity. Mol. Plant Microbe Interact. 29, 651–660 (2016).
Salomon, D., Dar, D., Sreeramulu, S. & Sessa, G. Expression of Xanthomonas campestris pv. vesicatoria type III effectors in yeast affects cell growth and viability. Mol. Plant Microbe Interact. 24, 305–314 (2011).
Mutka, A. M. et al. Quantitative, image-based phenotyping methods provide insight into spatial and temporal dimensions of plant disease. Plant Physiol. 172, 650–660 (2016).
Boch, J. et al. Breaking the code of DNA binding specificity of TAL-type III effectors. Science 326, 1509–1512 (2009).
Yang, B., Sugio, A. & White, F. F. Os8N3 is a host disease-susceptibility gene for bacterial blight of rice. Proc. Natl Acad. Sci. USA 103, 10503–10508 (2006).
Moscou, M. J. & Bogdanove, A. J. A simple cipher governs DNA recognition by TAL effectors. Science 326, 1501 (2009).
Thieme, F. et al. Insights into genome plasticity and pathogenicity of the plant pathogenic bacterium Xanthomonas campestris pv. vesicatoria revealed by the complete genome sequence. J. Bacteriol. 187, 7254–7266 (2005).
Hutin, M., Pérez-Quintero, A. L., Lopez, C. & Szurek, B. MorTAL Kombat: the story of defense against TAL effectors through loss-of-susceptibility. Front. Plant. Sci. 6, 535 (2015).
Oliva, R. et al. Broad-spectrum resistance to bacterial blight in rice using genome editing. Nat. Biotechnol. 37, 1344–1350 (2019).
Streubel, J. et al. Five phylogenetically close rice SWEET genes confer TAL effector-mediated susceptibility to Xanthomonas oryzae pv. oryzae. N. Phytol. 200, 808–819 (2013).
Ma, L. et al. Essential role of sugar transporter OsSWEET11 during the early stage of rice grain filling. Plant Cell Physiol. 58, 863–873 (2017).
Römer, P. et al. Promoter elements of rice susceptibility genes are bound and activated by specific TAL effectors from the bacterial blight pathogen, Xanthomonas oryzae pv. oryzae. N. Phytol. 187, 1048–1057 (2010).
Antony, G. et al. Rice xa13 recessive resistance to bacterial blight is defeated by induction of the disease susceptibility gene Os-11N3. Plant. Cell 22, 3864–3876 (2010).
Zhou, J. et al. Gene targeting by the TAL effector PthXo2 reveals cryptic resistance gene for bacterial blight of rice. Plant. J. 82, 632–643 (2015).
Yu, Y. et al. Colonization of rice leaf blades by an African strain of Xanthomonas oryzae pv. oryzae depends on a new TAL effector that induces the rice nodulin-3 Os11N3 gene. Mol. Plant. Microbe Interact. 24, 1102–1113 (2011).
Chen, L.-Q. et al. Sugar transporters for intercellular exchange and nutrition of pathogens. Nature 468, 527–532 (2010).
Peng, Z. et al. Xanthomonas translucens commandeers the host rate-limiting step in ABA biosynthesis for disease susceptibility. Proc. Natl Acad. Sci. USA 116, 20938–20946 (2019).
Falahi Charkhabi, N. et al. Complete genome sequencing and targeted mutagenesis reveal virulence contributions of Tal2 and Tal4b of Xanthomonas translucens pv. undulosa ICMP11055 in bacterial leaf streak of wheat. Front. Microbiol. 8, 1488 (2017).
Hu, Y. et al. Lateral organ boundaries 1 is a disease susceptibility gene for citrus bacterial canker disease. Proc. Natl Acad. Sci. USA 111, E521–E529 (2014).
Li, Z. et al. A potential disease susceptibility gene CsLOB of citrus is targeted by a major virulence effector PthA of Xanthomonas citri subsp. citri. Mol. Plant. 7, 912–915 (2014).
Jia, H. et al. Genome editing of the disease susceptibility gene CsLOB1 in citrus confers resistance to citrus canker. Plant Biotechnol. J. 15, 817–823 (2017).
Peng, A. et al. Engineering canker-resistant plants through CRISPR/Cas9-targeted editing of the susceptibility gene CsLOB1 promoter in citrus. Plant Biotechnol. J. 15, 1509–1519 (2017).
Schornack, S. et al. The tomato resistance protein Bs4 is a predicted non-nuclear TIR-NB-LRR protein that mediates defense responses to severely truncated derivatives of AvrBs4 and overexpressed AvrBs3. Plant. J. 37, 46–60 (2004).
Triplett, L. R. et al. A resistance locus in the American heirloom rice variety Carolina Gold Select is triggered by TAL effectors with diverse predicted targets and is effective against African strains of Xanthomonas oryzae pv. oryzicola. Plant. J. 87, 472–483 (2016).
Ji, Z. et al. Interfering TAL effectors of Xanthomonas oryzae neutralize R-gene-mediated plant disease resistance. Nat. Commun. 7, 13435 (2016).
Yoshimura, S. et al. Expression of Xa1, a bacterial blight-resistance gene in rice, is induced by bacterial inoculation. Proc. Natl Acad. Sci. USA 95, 1663–1668 (1998).
Zhang, J., Yin, Z. & White, F. TAL effectors and the executor R genes. Front. Plant Sci. 6, 641 (2015).
Abendroth, U., Schmidtke, C. & Bonas, U. Small non-coding RNAs in plant-pathogenic Xanthomonas spp. RNA Biol. 11, 457–463 (2014).
Solé, M. et al. Xanthomonas campestris pv. vesicatoria secretes proteases and xylanases via the Xps type II secretion system and outer membrane vesicles. J. Bacteriol. 197, 2879–2893 (2015).
Guerrero-Mandujano, A., Hernández-Cortez, C., Ibarra, J. A. & Castro-Escarpulli, G. The outer membrane vesicles: secretion system type zero. Traffic 18, 425–432 (2017).
Sidhu, V. K., Vorhölter, F.-J., Niehaus, K. & Watt, S. A. Analysis of outer membrane vesicle associated proteins isolated from the plant pathogenic bacterium Xanthomonas campestris pv. campestris. BMC Microbiol. 8, 87 (2008).
Zhou, X. et al. A phosphorylation switch on lon protease regulates bacterial type III secretion system in host. mBio 9, e02146-17 (2018).
Teper, D., Zhang, Y. & Wang, N. TfmR, a novel TetR-family transcriptional regulator, modulates the virulence of Xanthomonas citri in response to fatty acids. Mol. Plant. Pathol. 20, 701–715 (2019).
Cheng, S.-T., Wang, F.-F. & Qian, W. Cyclic-di-GMP binds to histidine kinase RavS to control RavS-RavR phosphotransfer and regulates the bacterial lifestyle transition between virulence and swimming. PLOS Pathog. 15, e1007952 (2019).
Deng, C.-Y. et al. Proteolysis of histidine kinase VgrS inhibits its autophosphorylation and promotes osmostress resistance in Xanthomonas campestris. Nat. Commun. 9, 1–15 (2018).
Wang, F.-F., Cheng, S.-T., Wu, Y., Ren, B.-Z. & Qian, W. A bacterial receptor PcrK senses the plant hormone cytokinin to promote adaptation to oxidative stress. Cell Rep. 21, 2940–2951 (2017).
Zheng, D. et al. Two overlapping two-component systems in Xanthomonas oryzae pv. oryzae contribute to full fitness in rice by regulating virulence factors expression. Sci. Rep. 6, 1–13 (2016).
Kim, H. et al. A genome-scale co-functional network of Xanthomonas genes can accurately reconstruct regulatory circuits controlled by two-component signaling systems. Mol. Cell 42, 166–174 (2019).
Souza, D. P. et al. Bacterial killing via a type IV secretion system. Nat. Commun. 6, 1–9 (2015).
Jackson, R. W., Vinatzer, B., Arnold, D. L., Dorus, S. & Murillo, J. The influence of the accessory genome on bacterial pathogen evolution. Mob. Genet. Elem. 1, 55–65 (2011).
Bartoli, C., Roux, F. & Lamichhane, J. R. Molecular mechanisms underlying the emergence of bacterial pathogens: an ecological perspective. Mol. Plant. Pathol. 17, 303–310 (2015).
Lima, W. C., Sluys, M.-A. V. & Menck, C. F. M. Non-γ-proteobacteria gene islands contribute to the Xanthomonas genome. OMICS 9, 160–172 (2005).
Lima, W. C., Paquola, A. C. M., Varani, A. M., Van Sluys, M.-A. & Menck, C. F. M. Laterally transferred genomic islands in Xanthomonadales related to pathogenicity and primary metabolism. FEMS Microbiol. Lett. 281, 87–97 (2008).
Lang, J. M. et al. A pathovar of Xanthomonas oryzae infecting wild grasses provides insight into the evolution of pathogenicity in rice agroecosystems. Front. Plant. Sci. 10, 507 (2019).
Hert, A. P. et al. Relative importance of bacteriocin-like genes in antagonism of Xanthomonas perforans tomato race 3 to Xanthomonas euvesicatoria tomato race 1 strains. Appl. Environ. Microbiol. 71, 3581–3588 (2005).
Timilsina, S. et al. Analysis of sequenced genomes of Xanthomonas perforans identifies candidate targets for resistance breeding in tomato. Phytopathology 106, 1097–1104 (2016).
Abrahamian, P. et al. The type III effector AvrBsT enhances Xanthomonas perforans fitness in field-grown tomato. Phytopathology 108, 1355–1362 (2018).
Quibod, I. L. et al. Effector diversification contributes to Xanthomonas oryzae pv. oryzae phenotypic adaptation in a semi-isolated environment. Sci. Rep. 6, 1–11 (2016).
Zipfel, C. et al. Bacterial disease resistance in Arabidopsis through flagellin perception. Nature 428, 764–767 (2004).
Zipfel, C. et al. Perception of the bacterial PAMP EF-Tu by the receptor EFR restricts Agrobacterium-mediated transformation. Cell 125, 749–760 (2006).
Willmann, R. et al. Arabidopsis lysin-motif proteins LYM1 LYM3 CERK1 mediate bacterial peptidoglycan sensing and immunity to bacterial infection. Proc. Natl Acad. Sci. USA 108, 19824–19829 (2011).
Kutschera, A. et al. Bacterial medium-chain 3-hydroxy fatty acid metabolites trigger immunity in Arabidopsis plants. Science 364, 178–181 (2019).
Buscaill, P. et al. Glycosidase and glycan polymorphism control hydrolytic release of immunogenic flagellin peptides. Science 364, eaav0748 (2019).
Sun, W., Dunning, F. M., Pfund, C., Weingarten, R. & Bent, A. F. Within-species flagellin polymorphism in Xanthomonas campestris pv campestris and its impact on elicitation of Arabidopsis FLAGELLIN SENSING2-dependent defenses. Plant. Cell 18, 764–779 (2006).
Wang, S. et al. Rice OsFLS2-mediated perception of bacterial flagellins is evaded by Xanthomonas oryzae pvs. oryzae and oryzicola. Mol. Plant. 8, 1024–1037 (2015).
Newman, M.-A., von Roepenack-Lahaye, E., Parr, A., Daniels, M. J. & Dow, J. M. Prior exposure to lipopolysaccharide potentiates expression of plant defenses in response to bacteria. Plant. J. 29, 487–495 (2002).
Erbs, G. et al. Peptidoglycan and muropeptides from pathogens Agrobacterium and Xanthomonas elicit plant innate immunity: structure and activity. Chem. Biol. 15, 438–448 (2008).
Lacombe, S. et al. Interfamily transfer of a plant pattern-recognition receptor confers broad-spectrum bacterial resistance. Nat. Biotechnol. 28, 365–369 (2010).
Proietti, S. et al. Xanthomonas campestris lipooligosaccharides trigger innate immunity and oxidative burst in Arabidopsis. Plant. Physiol. Biochem. 85, 51–62 (2014).
Kakkar, A., Nizampatnam, N. R., Kondreddy, A., Pradhan, B. B. & Chatterjee, S. Xanthomonas campestris cell–cell signalling molecule DSF (diffusible signal factor) elicits innate immunity in plants and is suppressed by the exopolysaccharide xanthan. J. Exp. Bot. 66, 6697–6714 (2015).
Keshavarzi, M. et al. Basal defenses induced in pepper by lipopolysaccharides are suppressed by Xanthomonas campestris pv. vesicatoria. Mol. Plant. Microbe Interact. 17, 805–815 (2004).
Li, S. et al. The type III effector AvrBs2 in Xanthomonas oryzae pv. oryzicola suppresses rice immunity and promotes disease development. Mol. Plant. Microbe Interact. 28, 869–880 (2015).
Priller, J. P. R., Reid, S., Konein, P., Dietrich, P. & Sonnewald, S. The Xanthomonas campestris pv. vesicatoria type-3 effector XopB inhibits plant defence responses by interfering with ROS production. PLOS ONE 11, e0159107 (2016).
Long, J. et al. Non-TAL effectors from Xanthomonas oryzae pv. oryzae suppress peptidoglycan-triggered MAPK activation in rice. Front. Plant. Sci. 9, 1857 (2018).
Medina, C. A. et al. The role of type III effectors from Xanthomonas axonopodis pv. manihotis in virulence and suppression of plant immunity. Mol. Plant. Pathol. 19, 593–606 (2018).
Song, W. Y. et al. A receptor kinase-like protein encoded by the rice disease resistance gene, Xa21. Science 270, 1804–1806 (1995).
Pruitt, R. N. et al. A microbially derived tyrosine-sulfated peptide mimics a plant peptide hormone. N. Phytol. 215, 725–736 (2017).
Liu, F. et al. Variation and inheritance of the Xanthomonas raxX–raxSTAB gene cluster required for activation of XA21-mediated immunity. Mol. Plant. Pathol. 20, 656–672 (2019).
Tai, T. H. et al. Expression of the Bs2 pepper gene confers resistance to bacterial spot disease in tomato. Proc. Natl Acad. Sci. USA 96, 14153–14158 (1999).
Wang, G. et al. The decoy substrate of a pathogen effector and a pseudokinase specify pathogen-induced modified-self recognition and immunity in plants. Cell Host Microbe 18, 285–295 (2015).
Kearney, B. & Staskawicz, B. J. Characterization of IS476 and its role in bacterial spot disease of tomato and pepper. J. Bacteriol. 172, 143–148 (1990).
Kousik, C. & Ritchie, D. F. Race shift in Xanthomonas campestris pv. vesicatoria within a season in field-grown pepper. Phytopathology 86, 952 (1996).
Vera Cruz, C. M. et al. Predicting durability of a disease resistance gene based on an assessment of the fitness loss and epidemiological consequences of avirulence gene mutation. Proc. Natl Acad. Sci. USA 97, 13500–13505 (2000).
Gassmann, W. et al. Molecular evolution of virulence in natural field strains of Xanthomonas campestris pv. vesicatoria. J. Bacteriol. 182, 7053–7059 (2000).
Swords, K. M., Dahlbeck, D., Kearney, B., Roy, M. & Staskawicz, B. J. Spontaneous and induced mutations in a single open reading frame alter both virulence and avirulence in Xanthomonas campestris pv. vesicatoria avrBs2. J. Bacteriol. 178, 4661–4669 (1996).
Read, A. C. et al. Suppression of Xo1-mediated disease resistance in rice by a truncated, non-DNA-binding TAL effector of Xanthomonas oryzae. Front. Plant Sci. 7, 1516 (2016).
Jones, J. B. et al. A non-hypersensitive resistance in pepper to the bacterial spot pathogen is associated with two recessive genes. Phytopathology 92, 273–277 (2002).
Schornack, S., Minsavage, G. V., Stall, R. E., Jones, J. B. & Lahaye, T. Characterization of AvrHah1, a novel AvrBs3-like effector from Xanthomonas gardneri with virulence and avirulence activity. N. Phytol. 179, 546–556 (2008).
Narvel, J. M. et al. Molecular mapping of Rxp conditioning reaction to bacterial pustule in soybean. J. Hered. 92, 267–270 (2001).
Chu, Z. et al. Promoter mutations of an essential gene for pollen development result in disease resistance in rice. Genes. Dev. 20, 1250–1255 (2006).
Huang, S. et al. The broadly effective recessive resistance gene xa5 of rice is a virulence effector-dependent quantitative trait for bacterial blight. Plant. J. 86, 186–194 (2016).
Kunwar, S. et al. Transgenic expression of EFR and Bs2 genes for field management of bacterial wilt and bacterial spot of tomato. Phytopathology 108, 1402–1411 (2018).
Sun, L. et al. Citrus genetic engineering for disease resistance: past, present and future. Int. J. Mol. Sci. 20, 5256 (2019).
Blanvillain-Baufumé, S. et al. Targeted promoter editing for rice resistance to Xanthomonas oryzae pv. oryzae reveals differential activities for SWEET14-inducing TAL effectors. Plant. Biotechnol. J. 15, 306–317 (2017).
Li, T., Liu, B., Spalding, M. H., Weeks, D. P. & Yang, B. High-efficiency TALEN-based gene editing produces disease-resistant rice. Nat. Biotechnol. 30, 390–392 (2012).
Shantharaj, D. et al. An engineered promoter driving expression of a microbial avirulence gene confers recognition of TAL effectors and reduces growth of diverse Xanthomonas strains in citrus. Mol. Plant. Pathol. 18, 976–989 (2017).
Page, A. J. et al. Roary: rapid large-scale prokaryote pan genome analysis. Bioinformatics 31, 3691–3693 (2015).
Boch, J., Bonas, U. & Lahaye, T. TAL effectors — pathogen strategies and plant resistance engineering. N. Phytol. 204, 823–832 (2014).
Richter, A. et al. A TAL effector repeat architecture for frameshift binding. Nat. Commun. 5, 1–10 (2014).
Pérez-Quintero, A. L. et al. An improved method for TAL effectors DNA-binding sites prediction reveals functional convergence in TAL repertoires of Xanthomonas oryzae strains. PLOS ONE 8, e68464 (2013).
Wernham, C. C. The species value of pathogenicity in the genus Xanthomonas. Phytopathology 38, 283–291 (1948).
Hajri, A. et al. A ‘repertoire for repertoire’ hypothesis: repertoires of type three effectors are candidate determinants of host specificity in Xanthomonas. PLOS ONE 4, e6632 (2009).
Figueiredo, J. F. L., Minsavage, G. V., Graham, J. H., White, F. F. & Jones, J. B. Mutational analysis of type III effector genes from Xanthomonas citri subsp. citri. Eur. J. Plant. Pathol. 130, 339–347 (2011).
Lu, H. et al. Acquisition and evolution of plant pathogenesis-associated gene clusters and candidate determinants of tissue-specificity in Xanthomonas. PLOS ONE 3, e3828 (2008).
Jacobs, J. M. et al. Evolutionary and biological basis of Xanthomonas systemic pathogenesis of plants [abstract 347-P]. Phytopathology 107, S5.1. (2017).
Karasov, T. L. et al. Arabidopsis thaliana and Pseudomonas pathogens exhibit stable associations over evolutionary timescales. Cell Host Microbe 24, 168–179.e4 (2018).
Salanoubat, M. et al. Genome sequence of the plant pathogen Ralstonia solanacearum. Nature 415, 497–502 (2002).
Wu, D. et al. A plant pathogen type III effector protein subverts translational regulation to boost host polyamine levels. Cell Host Microbe 26, 638–649 (2019).
Pérez-Quintero, A. L. et al. daTALbase: a database for genomic and transcriptomic data related to TAL effectors. Mol. Plant. Microbe Interact. 31, 471–480 (2018).
Erkes, A., Mücke, S., Reschke, M., Boch, J. & Grau, J. PrediTALE: a novel model learned from quantitative data allows for new perspectives on TALE targeting. PLOS Comput. Biol. 15, e1007206 (2019).
Mücke, S. et al. Transcriptional reprogramming of rice cells by Xanthomonas oryzae TALEs. Front. Plant. Sci. 10, 162 (2019).
Acknowledgements
The authors acknowledge A. M. Gochez and M. M. Shimwela for the images of Xanthomonas disease symptoms in citrus and banana, respectively.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Microbiology thanks Jian-Min Zhou and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Overview of T3SEs inXanthomonasResource: http://xanthomonas.org/t3e.html
Supplementary information
Glossary
- Vascular tissue
-
Tissue involved in transporting nutrients and fluids in plants. The primary components include xylem and phloem.
- Mesophyll tissue
-
Leaf tissue between the epidermis layers that carries out photosynthesis.
- Recombination
-
Genetic exchange between bacteria resulting in the incorporation of homologous and non-homologous sequences.
- Type III secretion system
-
(T3SS). A secretion system composed of ~20 proteins that forms a syringe-like structure to deliver bacterial proteins to eukaryotic cells. Also referred to as the injectisome.
- Effector-triggered immunity
-
(ETI). Innate immune response triggered by recognition of the type III translocated effector proteins by host resistance gene products.
- Type II secretion system
-
(T2SS). A secretion system formed by secretin proteins, which form characteristic β-barrels for passage of secreted proteins.
- Type VI secretion system
-
(T6SS). A secretion system that delivers bacterial proteins across a cellular envelope to adjacent target cells. Primarily known for interbacterial antagonism.
- Hypersensitive response
-
A response mechanism found in plant hosts, characterized typically by a rapid cell death to prevent the spread of the pathogen.
- Accessions
-
Groups of related plant material from the same species collected from a specific location. The accessions are collections to capture the diversity in a given plant species.
- Pathogen or damage-associated molecular pattern (P/DAMP)-triggered immunity
-
(PTI/DTI). PTI refers to the immune response in hosts triggered by recognizing patterns associated with pathogen, for example, flagellin or lipopolysaccharide. DTI refers to the host immune response triggered as a result of recognition of cell wall-degradation products that are generated by the action of pathogen-secreted cell wall-degrading enzymes during pathogen invasion. PTI and DTI pathways have a significant overlap in their signalling components.
- Receptor-like cytoplasmic kinases
-
Kinase-mediated signalling proteins that regulate plant cellular activities in response to biotic or abiotic stresses and endogenous extracellular signalling molecules.
- Receptor-like kinase superfamily
-
Transmembrane proteins with versatile amino-terminal extracellular domains and carboxy-terminal intracellular kinases. They control a wide range of physiological responses in plants and belong to one of the largest gene families in the Arabidopsis thaliana genome, with more than 600 members.
- MAPK
-
Protein kinases involved in regulating cellular responses to an extensive array of stimuli, including mitogens, heat shock and stress. Specific to serine and threonine amino acids.
- Protoplast
-
The entire cell excluding the cell wall.
- SWEET genes
-
Sugar will eventually be exported transporter (SWEET) genes encode membrane proteins with diverse function, typically facilitating sucrose and glucose efflux.
- Recessive resistance
-
Resistance conferred by recessive allele of a gene in a plant host. The term is also used to refer to resistance conferred by mutation in disease-susceptibility genes.
- Abscisic acid
-
A plant hormone with numerous functions in the plant developmental process, including dormancy and stress response.
- Nucleotide binding, leucine-rich repeat (NLR) resistance genes
-
Resistance genes named after their characteristic nucleotide binding and leucine-rich repeat domains.
- Two-component system
-
(TCS). Mediators of signal transduction in bacteria to detect the surrounding changes and relay the signal for modulating gene expression.
- Pathogenic races
-
Groups of strains that belong to the same or closely related bacterial species, characterized by differential responses (compatible or incompatible reaction) on an array of hosts.
- Dominant resistance
-
Resistance conferred by a single dominant resistance gene in plant hosts.
Rights and permissions
About this article
Cite this article
Timilsina, S., Potnis, N., Newberry, E.A. et al. Xanthomonas diversity, virulence and plant–pathogen interactions. Nat Rev Microbiol 18, 415–427 (2020). https://doi.org/10.1038/s41579-020-0361-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41579-020-0361-8
This article is cited by
-
Constructed Rice Tracers Identify the Major Virulent Transcription Activator-Like Effectors of the Bacterial Leaf Blight Pathogen
Rice (2024)
-
High genomic plasticity and unique features of Xanthomonas translucens pv. graminis revealed through comparative analysis of complete genome sequences
BMC Genomics (2023)
-
Xanthomonas euvesicatoria pv. perforans is the causative agent of bacterial leaf spot on Ipomoea aquatica from Fujian Province in China
Australasian Plant Pathology (2023)
-
Identification and population features of Xanthomonas arboricola pv. pruni, X. arboricola pv. coryli, and X. arboricola pv. juglandis strains in Iran
Journal of Plant Pathology (2023)
-
Involvement of oxidative stress in localization of bacterial spot infection in pepper plants
Journal of Biosciences (2023)