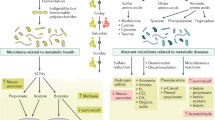
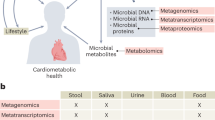
Abstract
Conceptual scientific and medical advances have led to a recent realization that there may be no single, one-size-fits-all diet and that differential human responses to dietary inputs may rather be driven by unique and quantifiable host and microbiome features. Integration of these person-specific host and microbiome readouts into actionable modules may complement traditional food measurement approaches in devising diets that are of benefit to the individual. Although many host-derived factors are hardwired and difficult to modulate, the microbiome may be more readily reshaped by environmental factors such as dietary exposures and is increasingly recognized to potentially impact human physiology by participating in digestion, the absorption of nutrients, shaping of the mucosal immune response and the synthesis or modulation of a plethora of potentially bioactive compounds. Thus, diet-induced microbiota alterations may be harnessed in order to induce changes in host physiology, including disease development and progression. However, major limitations in ‘big-data’ processing and analysis still limit our interpretive and translational capabilities concerning these person-specific host, microbiome and diet interactions. In this Review, we describe the latest advances in understanding diet–microbiota interactions, the individuality of gut microbiota composition and how this knowledge could be harnessed for personalized nutrition strategies to improve human health.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Cho, I. & Blaser, M. J. The human microbiome: at the interface of health and disease. Nat. Rev. Genet. 13, 260–270 (2012).
Le Chatelier, E. et al. Richness of human gut microbiome correlates with metabolic markers. Nature 500, 541–546 (2013).
Ley, R. E., Turnbaugh, P. J., Klein, S. & Gordon, J. I. Microbial ecology: human gut microbes associated with obesity. Nature 444, 1022–1023 (2006).
Gomez de Aguero, M. et al. The maternal microbiota drives early postnatal innate immune development. Science 351, 1296–1302 (2016).
Koenig, J. E. et al. Succession of microbial consortia in the developing infant gut microbiome. Proc. Natl Acad. Sci. USA 108, 4578–4585 (2011).
Arrieta, M. C., Stiemsma, L. T., Amenyogbe, N., Brown, E. M. & Finlay, B. The intestinal microbiome in early life: health and disease. Front. Immunol. 5, 427 (2014).
Laursen, M. F., Bahl, M. I., Michaelsen, K. F. & Licht, T. R. First foods and gut microbes. Front. Microbiol. 8, 356 (2017).
Yatsunenko, T. et al. Human gut microbiome viewed across age and geography. Nature 486, 222–227 (2012).
Claesson, M. J. et al. Gut microbiota composition correlates with diet and health in the elderly. Nature 488, 178–184 (2012).
David, L. A. et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 505, 559–563 (2014).
Muegge, B. D. et al. Diet drives convergence in gut microbiome functions across mammalian phylogeny and within humans. Science 332, 970–974 (2011).
Rajilić-Stojanović, M., Heilig, H. G. H. J., Tims, S., Zoetendal, E. G. & de Vos, W. M. Long-term monitoring of the human intestinal microbiota composition. Environ. Microbiol. 15, 1146–1159, https://doi.org/10.1111/1462-2920.12023 (2013).
Rothschild, D. et al. Environment dominates over host genetics in shaping human gut microbiota. Nature 555, 210–215 (2018).
Zeevi, D. et al. Personalized nutrition by prediction of glycemic responses. Cell 163, 1079–1094 (2015). This seminal study shows that microbiome data accurately predict personalized postprandial glycaemic responses.
Kau, A. L., Ahern, P. P., Griffin, N. W., Goodman, A. L. & Gordon, J. I. Human nutrition, the gut microbiome and the immune system. Nature 474, 327–336 (2011).
Zmora, N., Suez, J. & Elinav, E. You are what you eat: diet, health and the gut microbiota. Nat. Rev. Gastroenterol. Hepatol. 16, 35–56 (2019).
Goodrich, J. K. et al. Human genetics shape the gut microbiome. Cell 159, 789–799 (2014).
Zhernakova, A. et al. Population-based metagenomics analysis reveals markers for gut microbiome composition and diversity. Science 352, 565–569 (2016).
Falony, G. et al. Population-level analysis of gut microbiome variation. Science 352, 560–564 (2016).
Smits, S. A. et al. Seasonal cycling in the gut microbiome of the hadza hunter-gatherers of tanzania. Science 357, 802–806 (2017). This paper provides an excellent example of how the gut microbiome changes dynamically with seasonal dietary shifts as well as westernization.
Davenport, E. R. et al. Seasonal variation in human gut microbiome composition. PLOS ONE 9, e90731 (2014).
Obregon-Tito, A. J. et al. Subsistence strategies in traditional societies distinguish gut microbiomes. Nat. Commun. 6, 6505 (2015).
Schnorr, S. L. et al. Gut microbiome of the Hadza hunter-gatherers. Nat. Commun. 5, 3654 (2014).
De Filippo, C. et al. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl Acad. Sci. USA 107, 14691–14696 (2010).
Vangay, P. et al. US immigration westernizes the human gut microbiome. Cell 175, 962–972.e910 (2018).
Ayeni, F. A. et al. Infant and adult gut microbiome and metabolome in rural Bassa and urban settlers from Nigeria. Cell Rep. 23, 3056–3067 (2018).
Das, B. et al. Analysis of the gut microbiome of rural and urban healthy Indians living in sea level and high altitude areas. Sci. Rep. 8, 10104 (2018).
O’Keefe, S. J. et al. Fat, fibre and cancer risk in African Americans and rural Africans. Nat. Commun. 6, 6342 (2015).
Turnbaugh, P. J. et al. The effect of diet on the human gut microbiome: a metagenomic analysis in humanized gnotobiotic mice. Sci. Transl. Med. 1, 6ra14 (2009).
Korem, T. et al. Bread affects clinical parameters and induces gut microbiome-associated personal glycemic responses. Cell Metab. 25, 1243–1253.e1245 (2017).
Brooks, A. W., Priya, S., Blekhman, R. & Bordenstein, S. R. Gut microbiota diversity across ethnicities in the United States. PLOS Biol. 16, e2006842 (2018).
Sanz, Y. et al. Towards microbiome-informed dietary recommendations for promoting metabolic and mental health: opinion papers of the MyNewGut project. Clin. Nutr. 37, 2191–2197 (2018).
Zhang, C. et al. Structural resilience of the gut microbiota in adult mice under high-fat dietary perturbations. ISME J. 6, 1848–1857 (2012).
Hildebrandt, M. A. et al. High-fat diet determines the composition of the murine gut microbiome independently of obesity. Gastroenterology 137, 1716–1724.e1–2 (2009).
Turnbaugh, P. J., Backhed, F., Fulton, L. & Gordon, J. I. Diet-induced obesity is linked to marked but reversible alterations in the mouse distal gut microbiome. Cell Host Microbe 3, 213–223 (2008).
Parks, B. W. et al. Genetic control of obesity and gut microbiota composition in response to high-fat, high-sucrose diet in mice. Cell Metab. 17, 141–152 (2013).
Lai, M., Chandrasekera, P. C. & Barnard, N. D. You are what you eat, or are you? The challenges of translating high-fat-fed rodents to human obesity and diabetes. Nutr. Diabetes 4, e135 (2014).
Mokkala, K., Houttu, N., Cansev, T. & Laitinen, K. Interactions of dietary fat with the gut microbiota: evaluation of mechanisms and metabolic consequences. Clin. Nutr. https://doi.org/10.1016/j.clnu.2019.05.003 (2019).
Wolters, M. et al. Dietary fat, the gut microbiota, and metabolic health — a systematic review conducted within the MyNewGut project. Clin. Nutr. https://doi.org/10.1016/j.clnu.2018.12.024 (2018).
Laursen, M. F. et al. Infant gut microbiota development is driven by transition to family foods independent of maternal obesity. mSphere 1, e00069-15 (2016).
Wan, Y. et al. Effects of dietary fat on gut microbiota and faecal metabolites, and their relationship with cardiometabolic risk factors: a 6-month randomised controlled-feeding trial. Gut 68, 1417–1429 (2019).
Watson, H. et al. A randomised trial of the effect of omega-3 polyunsaturated fatty acid supplements on the human intestinal microbiota. Gut 67, 1974–1983 (2018).
Lang, J. M. et al. Impact of individual traits, saturated fat, and protein source on the gut microbiome. mBio 9, e01604-18 (2018). This study shows how healthy individuals respond to dietary fat and protein consumption in a highly individualized pattern.
Zhu, Y. et al. Meat, dairy and plant proteins alter bacterial composition of rat gut bacteria. Sci. Rep. 5, 15220 (2015).
Wu, G. D. et al. Linking long-term dietary patterns with gut microbial enterotypes. Science 334, 105–108 (2011).
Swiatecka, D., Narbad, A., Ridgway, K. P. & Kostyra, H. The study on the impact of glycated pea proteins on human intestinal bacteria. Int. J. Food Microbiol. 145, 267–272 (2011).
Dostal Webster, A. et al. Influence of short-term changes in dietary sulfur on the relative abundances of intestinal sulfate-reducing bacteria. Gut Microbes 10, 447–457 (2019).
Riviere, A. et al. The ability of bifidobacteria to degrade arabinoxylan oligosaccharide constituents and derived oligosaccharides is strain dependent. Appl. Environ. Microbiol. 80, 204–217 (2014).
Hansen, L. B. S. et al. A low-gluten diet induces changes in the intestinal microbiome of healthy danish adults. Nat. Commun. 9, 4630 (2018).
Walker, A. W. et al. Dominant and diet-responsive groups of bacteria within the human colonic microbiota. ISME J. 5, 220–230 (2011).
Duncan, S. H. et al. Reduced dietary intake of carbohydrates by obese subjects results in decreased concentrations of butyrate and butyrate-producing bacteria in feces. Appl. Environ. Microbiol. 73, 1073–1078 (2007).
Desai, M. S. et al. A dietary fiber-deprived gut microbiota degrades the colonic mucus barrier and enhances pathogen susceptibility. Cell 167, 1339–1353.e21 (2016).
Townsend, G. E. 2nd et al. Dietary sugar silences a colonization factor in a mammalian gut symbiont. Proc. Natl Acad. Sci. USA 116, 233–238 (2019).
Tap, J. et al. Gut microbiota richness promotes its stability upon increased dietary fibre intake in healthy adults. Environ. Microbiol. 17, 4954–4964 (2015).
Martinez, I., Kim, J., Duffy, P. R., Schlegel, V. L. & Walter, J. Resistant starches types 2 and 4 have differential effects on the composition of the fecal microbiota in human subjects. PLOS ONE 5, e15046 (2010).
Davis, L. M., Martinez, I., Walter, J., Goin, C. & Hutkins, R. W. Barcoded pyrosequencing reveals that consumption of galactooligosaccharides results in a highly specific bifidogenic response in humans. PLOS ONE 6, e25200 (2011).
Bouhnik, Y. et al. The capacity of nondigestible carbohydrates to stimulate fecal bifidobacteria in healthy humans: a double-blind, randomized, placebo-controlled, parallel-group, dose-response relation study. Am. J. Clin. Nutr. 80, 1658–1664 (2004).
Salonen, A. et al. Impact of diet and individual variation on intestinal microbiota composition and fermentation products in obese men. ISME J. 8, 2218–2230 (2014).
Cotillard, A. et al. Dietary intervention impact on gut microbial gene richness. Nature 500, 585–588 (2013). This study highlights that the efficacy of dietary intervention is person-specific and can be predicted by low gene richness of the gut microbiota.
Korpela, K. et al. Gut microbiota signatures predict host and microbiota responses to dietary interventions in obese individuals. PLOS ONE 9, e90702 (2014).
McOrist, A. L. et al. Fecal butyrate levels vary widely among individuals but are usually increased by a diet high in resistant starch. J. Nutr. 141, 883–889 (2011).
Healey, G. et al. Habitual dietary fibre intake influences gut microbiota response to an inulin-type fructan prebiotic: a randomised, double-blind, placebo-controlled, cross-over, human intervention study. Br. J. Nutr. 119, 176–189 (2018).
Chassaing, B. et al. Dietary emulsifiers impact the mouse gut microbiota promoting colitis and metabolic syndrome. Nature 519, 92–96 (2015).
Viennois, E., Merlin, D., Gewirtz, A. T. & Chassaing, B. Dietary emulsifier-induced low-grade inflammation promotes colon carcinogenesis. Cancer Res. 77, 27–40 (2017).
Ruiz-Ojeda, F. J., Plaza-Diaz, J., Saez-Lara, M. J. & Gil, A. Effects of sweeteners on the gut microbiota: a review of experimental studies and clinical trials. Adv. Nutr. 10, S31–S48 (2019).
Bian, X. et al. Saccharin induced liver inflammation in mice by altering the gut microbiota and its metabolic functions. Food Chem. Toxicol. 107, 530–539 (2017).
Uebanso, T. et al. Effects of low-dose non-caloric sweetener consumption on gut microbiota in mice. Nutrients 9, E560 (2017).
Rodriguez-Palacios, A. et al. The artificial sweetener Splenda promotes gut proteobacteria, dysbiosis, and myeloperoxidase reactivity in Crohn’s disease-like ileitis. Inflamm. Bowel Dis. 24, 1005–1020 (2018).
Suez, J. et al. Artificial sweeteners induce glucose intolerance by altering the gut microbiota. Nature 514, 181–186 (2014).
Ferrario, C. et al. Modulation of fecal Clostridiales bacteria and butyrate by probiotic intervention with Lactobacillus paracasei DG varies among healthy adults. J. Nutr. 144, 1787–1796 (2014).
Goossens, D. A., Jonkers, D. M., Russel, M. G., Stobberingh, E. E. & Stockbrugger, R. W. The effect of a probiotic drink with Lactobacillus plantarum 299v on the bacterial composition in faeces and mucosal biopsies of rectum and ascending colon. Aliment. Pharmacol. Ther. 23, 255–263 (2006).
Kristensen, N. B. et al. Alterations in fecal microbiota composition by probiotic supplementation in healthy adults: a systematic review of randomized controlled trials. Genome Med. 8, 52 (2016).
Laursen, M. F. et al. Administration of two probiotic strains during early childhood does not affect the endogenous gut microbiota composition despite probiotic proliferation. BMC Microbiol. 17, 175 (2017).
Zmora, N. et al. Personalized gut mucosal colonization resistance to empiric probiotics is associated with unique host and microbiome features. Cell 174, 1388–1405.e21 (2018). This paper provides important evidence about predictable personalized probiotic colonization patterns in human gut mucosa.
Suez, J. et al. Post-antibiotic gut mucosal microbiome reconstitution is impaired by probiotics and improved by autologous FMT. Cell 174, 1406–1423.e16 (2018).
Maldonado-Gómez, M. X. et al. Stable engraftment of Bifidobacterium longum AH1206 in the human gut depends on individualized features of the resident microbiome. Cell Host Microbe 20, 515–526 (2016).
Kovatcheva-Datchary, P. et al. Dietary fiber-induced improvement in glucose metabolism is associated with increased abundance of Prevotella. Cell Metab. 22, 971–982 (2015).
Martinez, I. et al. Gut microbiome composition is linked to whole grain-induced immunological improvements. ISME J. 7, 269–280 (2013).
Dao, M. C. et al. Akkermansia muciniphila and improved metabolic health during a dietary intervention in obesity: relationship with gut microbiome richness and ecology. Gut 65, 426–436 (2016).
Chumpitazi, B. P. et al. Randomised clinical trial: gut microbiome biomarkers are associated with clinical response to a low FODMAP diet in children with the irritable bowel syndrome. Aliment. Pharmacol. Ther. 42, 418–427 (2015).
Chumpitazi, B. P. et al. Gut microbiota influences low fermentable substrate diet efficacy in children with irritable bowel syndrome. Gut Microbes 5, 165–175 (2014).
Mendes-Soares, H. et al. Assessment of a personalized approach to predicting postprandial glycemic responses to food among individuals without diabetes. JAMA Netw. Open 2, e188102 (2019).
Berry, S. et al. Large inter-individual variation in postprandial lipemia following a mixed meal in over 1000 twins and singletons from the UK and US: The PREDICT I Study (OR19-06-19). Curr. Dev. Nutr. 3, nzz2046.OR19-06-19 (2019).
Mukherji, A., Kobiita, A., Ye, T. & Chambon, P. Homeostasis in intestinal epithelium is orchestrated by the circadian clock and microbiota cues transduced by TLRs. Cell 153, 812–827 (2013).
Thaiss, C. A. et al. Transkingdom control of microbiota diurnal oscillations promotes metabolic homeostasis. Cell 159, 514–529 (2014).
Zarrinpar, A., Chaix, A., Yooseph, S. & Panda, S. Diet and feeding pattern affect the diurnal dynamics of the gut microbiome. Cell Metab. 20, 1006–1017 (2014).
Leone, V. et al. Effects of diurnal variation of gut microbes and high-fat feeding on host circadian clock function and metabolism. Cell Host Microbe 17, 681–689 (2015).
Patterson, R. E. & Sears, D. D. Metabolic effects of intermittent fasting. Annu. Rev. Nutr. 37, 371–393 (2017).
Li, G. et al. Intermittent fasting promotes white adipose browning and decreases obesity by shaping the gut microbiota. Cell Metab. 26, 672–685.e4 (2017).
Cignarella, F. et al. Intermittent fasting confers protection in CNS autoimmunity by altering the gut microbiota. Cell Metab. 27, 1222–1235.e26 (2018).
Thaiss, C. A. et al. Persistent microbiome alterations modulate the rate of post-dieting weight regain. Nature 540, 544–551 (2016).
Carmody, R. N. et al. Diet dominates host genotype in shaping the murine gut microbiota. Cell Host Microbe 17, 72–84 (2015).
Sonnenburg, E. D. et al. Diet-induced extinctions in the gut microbiota compound over generations. Nature 529, 212–215 (2016).
Englyst, H., Wiggins, H. S. & Cummings, J. H. Determination of the non-starch polysaccharides in plant foods by gas–liquid chromatography of constituent sugars as alditol acetates. Analyst 107, 307–318 (1982).
Sajilata, M. G., Singhal, R. S. & Kulkarni, P. R. Resistant starch — a review. Compr. Rev. Food Sci. Food Saf. 5, 1–17 (2006).
McNeil, N. I. The contribution of the large intestine to energy supplies in man. Am. J. Clin. Nutr. 39, 338–342 (1984).
Slavin, J. L., Brauer, P. M. & Marlett, J. A. Neutral detergent fiber, hemicellulose and cellulose digestibility in human subjects. J. Nutr. 111, 287–297 (1981).
Hehemann, J. H. et al. Transfer of carbohydrate-active enzymes from marine bacteria to Japanese gut microbiota. Nature 464, 908–912 (2010).
Pluvinage, B. et al. Molecular basis of an agarose metabolic pathway acquired by a human intestinal symbiont. Nat. Commun. 9, 1043 (2018).
Tasse, L. et al. Functional metagenomics to mine the human gut microbiome for dietary fiber catabolic enzymes. Genome Res. 20, 1605–1612 (2010).
Luis, A. S. & Martens, E. C. Interrogating gut bacterial genomes for discovery of novel carbohydrate degrading enzymes. Curr. Opin. Chem. Biol. 47, 126–133 (2018).
Cummings, J. H. Fermentation in the human large intestine: evidence and implications for health. Lancet 1, 1206–1209 (1983).
Fukuda, S. et al. Bifidobacteria can protect from enteropathogenic infection through production of acetate. Nature 469, 543–547 (2011).
Maslowski, K. M. et al. Regulation of inflammatory responses by gut microbiota and chemoattractant receptor GPR43. Nature 461, 1282–1286 (2009). This study identifies regulatory T cell homeostasis as one of the mechanisms by which SCFAs affect the host.
Smith, P. M. et al. The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science 341, 569–573 (2013).
Sina, C. et al. G protein-coupled receptor 43 is essential for neutrophil recruitment during intestinal inflammation. J. Immunol. 183, 7514–7522 (2009).
Cho, I. et al. Antibiotics in early life alter the murine colonic microbiome and adiposity. Nature 488, 621–626 (2012).
LeBlanc, J. G. et al. Bacteria as vitamin suppliers to their host: a gut microbiota perspective. Curr. Opin. Biotechnol. 24, 160–168 (2013).
Magnusdottir, S., Ravcheev, D., de Crecy-Lagard, V. & Thiele, I. Systematic genome assessment of B-vitamin biosynthesis suggests co-operation among gut microbes. Front. Genet. 6, 148 (2015).
Allison, M. J., Cook, H. M., Milne, D. B., Gallagher, S. & Clayman, R. V. Oxalate degradation by gastrointestinal bacteria from humans. J. Nutr. 116, 455–460 (1986).
Hokama, S., Honma, Y., Toma, C. & Ogawa, Y. Oxalate-degrading Enterococcus faecalis. Microbiol. Immunol. 44, 235–240 (2000).
Koeth, R. A. et al. Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat. Med. 19, 576–585 (2013).
Wang, Z. et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 472, 57–63 (2011). This study identifies a microbially derived compound as a driver of cardiovascular diseases.
Salaspuro, V. et al. Ethanol oxidation and acetaldehyde production in vitro by human intestinal strains of Escherichia coli under aerobic, microaerobic, and anaerobic conditions. Scand. J. Gastroenterol. 34, 967–973 (1999).
Seitz, H. K. et al. Possible role of acetaldehyde in ethanol-related rectal cocarcinogenesis in the rat. Gastroenterology 98, 406–413 (1990).
Johnson, C. H. et al. Metabolism links bacterial biofilms and colon carcinogenesis. Cell Metab. 21, 891–897 (2015).
Hoyles, L. et al. Molecular phenomics and metagenomics of hepatic steatosis in non-diabetic obese women. Nat. Med. 24, 1070–1080 (2018).
Hsiao, E. Y. et al. Microbiota modulate behavioral and physiological abnormalities associated with neurodevelopmental disorders. Cell 155, 1451–1463 (2013).
Lee, J. H. & Lee, J. Indole as an intercellular signal in microbial communities. FEMS Microbiol. Rev. 34, 426–444 (2010).
Roager, H. M. & Licht, T. R. Microbial tryptophan catabolites in health and disease. Nat. Commun. 9, 3294 (2018).
Hubbard, T. D. et al. Adaptation of the human aryl hydrocarbon receptor to sense microbiota-derived indoles. Sci. Rep. 5, 12689 (2015).
Bessede, A. et al. Aryl hydrocarbon receptor control of a disease tolerance defence pathway. Nature 511, 184–190 (2014).
Cervantes-Barragan, L. et al. Lactobacillus reuteri induces gut intraepithelial CD4+CD8αα+ T cells. Science 357, 806–810 (2017).
Monteleone, I. et al. Aryl hydrocarbon receptor-induced signals up-regulate IL-22 production and inhibit inflammation in the gastrointestinal tract. Gastroenterology 141, 237–248.e1 (2011).
Venkatesh, M. et al. Symbiotic bacterial metabolites regulate gastrointestinal barrier function via the xenobiotic sensor PXR and Toll-like receptor 4. Immunity 41, 296–310 (2014).
Chimerel, C. et al. Bacterial metabolite indole modulates incretin secretion from intestinal enteroendocrine L cells. Cell Rep. 9, 1202–1208 (2014).
Krishnan, S. et al. Gut microbiota-derived tryptophan metabolites modulate inflammatory response in hepatocytes and macrophages. Cell Rep. 23, 1099–1111 (2018).
Sayin, S. I. et al. Gut microbiota regulates bile acid metabolism by reducing the levels of tauro-beta-muricholic acid, a naturally occurring FXR antagonist. Cell Metab. 17, 225–235 (2013).
Swann, J. R. et al. Systemic gut microbial modulation of bile acid metabolism in host tissue compartments. Proc. Natl Acad. Sci. USA 108, 4523–4530 (2011).
Ridlon, J. M., Kang, D. J. & Hylemon, P. B. Bile salt biotransformations by human intestinal bacteria. J. Lipid Res. 47, 241–259 (2006).
Groh, H., Schade, K. & Horhold-Schubert, C. Steroid metabolism with intestinal microorganisms. J. Basic Microbiol. 33, 59–72 (1993).
Malik, N. A. Solubilization and interaction studies of bile salts with surfactants and drugs: a review. Appl. Biochem. Biotechnol. 179, 179–201 (2016).
Shapiro, H., Kolodziejczyk, A. A., Halstuch, D. & Elinav, E. Bile acids in glucose metabolism in health and disease. J. Exp. Med. 215, 383–396 (2018).
de Aguiar Vallim, T. Q., Tarling, E. J. & Edwards, P. A. Pleiotropic roles of bile acids in metabolism. Cell Metab. 17, 657–669 (2013).
van Dijk, T. H. et al. An increased flux through the glucose 6-phosphate pool in enterocytes delays glucose absorption in Fxr-/- mice. J. Biol. Chem. 284, 10315–10323 (2009).
Turnbaugh, P. J. et al. A core gut microbiome in obese and lean twins. Nature 457, 480–484 (2009).
Ridaura, V. K. et al. Gut microbiota from twins discordant for obesity modulate metabolism in mice. Science 341, 1241214 (2013).
Ley, R. E. et al. Obesity alters gut microbial ecology. Proc. Natl Acad. Sci. USA 102, 11070–11075 (2005).
Turnbaugh, P. J. et al. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444, 1027–1031 (2006).
Karlsson, F. H. et al. Gut metagenome in European women with normal, impaired and diabetic glucose control. Nature 498, 99–103 (2013).
Tremaroli, V. et al. Roux-en-Y gastric bypass and vertical banded gastroplasty induce long-term changes on the human gut microbiome contributing to fat mass regulation. Cell Metab. 22, 228–238 (2015).
Zhang, H. et al. Human gut microbiota in obesity and after gastric bypass. Proc. Natl Acad. Sci. USA 106, 2365–2370 (2009).
Ussar, S. et al. Interactions between gut microbiota, host genetics and diet modulate the predisposition to obesity and metabolic syndrome. Cell Metab. 22, 516–530 (2015).
Everard, A. et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl Acad. Sci. USA 110, 9066–9071 (2013).
Shin, N. R. et al. An increase in the Akkermansia spp. population induced by metformin treatment improves glucose homeostasis in diet-induced obese mice. Gut 63, 727–735 (2014).
Plovier, H. et al. A purified membrane protein from Akkermansia muciniphila or the pasteurized bacterium improves metabolism in obese and diabetic mice. Nat. Med. 23, 107–113 (2017).
Sokol, H. et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc. Natl Acad. Sci. USA 105, 16731–16736 (2008).
Wang, X. et al. Interleukin-22 alleviates metabolic disorders and restores mucosal immunity in diabetes. Nature 514, 237–241 (2014).
Zhu, Z. et al. Sulfated polysaccharide from sea cucumber and its depolymerized derivative prevent obesity in association with modification of gut microbiota in high-fat diet-fed mice. Mol. Nutr. Food Res. 62, e1800446 (2018).
Cani, P. D. et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 56, 1761–1772 (2007).
Ghoshal, S., Witta, J., Zhong, J., de Villiers, W. & Eckhardt, E. Chylomicrons promote intestinal absorption of lipopolysaccharides. J. Lipid Res. 50, 90–97 (2009).
Caesar, R. et al. Gut-derived lipopolysaccharide augments adipose macrophage accumulation but is not essential for impaired glucose or insulin tolerance in mice. Gut 61, 1701–1707 (2012).
Cani, P. D. et al. Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes 57, 1470–1481 (2008).
Reijnders, D. et al. Effects of gut microbiota manipulation by antibiotics on host metabolism in obese humans: a randomized double-blind placebo-controlled trial. Cell Metab. 24, 341 (2016).
Albers, D. J. et al. Personalized glucose forecasting for type 2 diabetes using data assimilation. PLOS Comput. Biol. 13, e1005232 (2017).
Beura, L. K. et al. Normalizing the environment recapitulates adult human immune traits in laboratory mice. Nature 532, 512–516 (2016).
Ghatak, S., King, Z. A., Sastry, A. & Palsson, B. O. The y-ome defines the 35% of Escherichia coli genes that lack experimental evidence of function. Nucleic Acids Res. 47, 2446–2454 (2019).
Patti, G. J., Yanes, O. & Siuzdak, G. Metabolomics: the apogee of the omics trilogy. Nat. Rev. Mol. Cell Biol. 13, 263–269 (2012).
Grieneisen, L. E. et al. Genes, geology and germs: gut microbiota across a primate hybrid zone are explained by site soil properties, not host species. Proc. Biol. Sci. 286, 20190431 (2019).
Amato, K. R. et al. Evolutionary trends in host physiology outweigh dietary niche in structuring primate gut microbiomes. ISME J. 13, 576–587 (2019).
Knowles, S. C. L., Eccles, R. M. & Baltrunaite, L. Species identity dominates over environment in shaping the microbiota of small mammals. Ecol. Lett. 22, 826–837 (2019).
Goodrich, J. K., Davenport, E. R., Clark, A. G. & Ley, R. E. The relationship between the human genome and microbiome comes into view. Annu. Rev. Genet. 51, 413–433 (2017).
Wang, J. et al. Meta-analysis of human genome–microbiome association studies: the MiBioGen consortium initiative. Microbiome 6, 101 (2018).
Acknowledgements
The authors thank the members of the Elinav lab and the members of the Division of Microbiome and Cancer, DKFZ, for discussions and apologize to researchers whose work was not included due to space constraints. A.A.K. was a recipient of EMBO Long Term Fellowship 2016-1088 and support from the European Union’s Horizon 2020 research and innovation programme under Marie Sklodowska-Curie Grant Agreement No. 747114. D.Z. is the recipient of a European Crohn’s and Colitis Organization (ECCO) Fellowship and is supported by the Ke Lin Program of the First Affiliated Hospital, Sun Yat-sen University. E.E. is supported by Yael and Rami Ungar, the Leona M. and Harry B. Helmsley Charitable Trust, the Adelis Foundation, the Pearl Welinsky Merlo Scientific Progress Research Fund, the Lawrence and Sandra Post Family Foundation, the Daniel Morris Trust, the Park Avenue Charitable Fund, the Hanna and Dr. Ludwik Wallach Cancer Research Fund, the Howard and Nancy Marks Charitable Fund, the Aliza Moussaieff Estate of Malka Moskowitz, the Estate of Myron H. Ackerman, the Estate of Bernard Bishin for the WIS-Clalit Program, Donald and Susan Schwarz, grants funded by the European Research Council, the Israel Science Foundation, Israel Ministry of Science and Technology, Israel Ministry of Health, the Helmholtz Foundation, the Else Kroener Fresenius Foundation, the Garvan Institute, the European Crohn’s and Colitis Organisation, Deutsch-Israelische Projektkooperation and the Wellcome Trust. E.E. is the incumbent of the Sir Marc and Lady Tania Feldmann Professorial Chair, a senior fellow of the Canadian Institute of Advanced Research (CIFAR) and an international scholar of the Bill and Melinda Gates Foundation and the Howard Hughes Medical Institute (HHMI).
Author information
Authors and Affiliations
Contributions
All authors researched the data for this article, made substantial contributions to discussion of the content, and wrote, reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
E.E. is a paid consultant at DayTwo and BiomX. All other authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Microbiology thanks A. Gewirtz and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Personalized medicine
-
A medical approach in which patients are stratified into groups depending on different factors that contribute to treatment outcomes and then receive the tailored treatment predicted to be most effective.
- Urbanization
-
Changes from rural to urban areas and encompasses both the flux of people from rural areas to cities and the growth of urban areas.
- Metabolic derangements
-
Pathological states in which the host metabolism is dysregulated, which are associated with a clustering of metabolic disorders, including obesity, hypertension, insulin resistance, impaired glucose tolerance and dyslipidemia.
- Butyrate
-
A short-chain fatty acid produced by bacteria in the gut from complex carbohydrates.
- Enterotype
-
Proposed classification of human microbiomes into three different types, depending on which bacterial genus is most prevalent: Bacteroides, Prevotella or Ruminococcus.
- Saccharolytic microorganisms
-
Microorganisms that break down sugar to acquire energy.
- Prebiotics
-
Foods, or compounds found in food, that induce the growth of bacterial species that are beneficial.
- Emulsifiers
-
Substances found in food, used to prevent the separation of emulsions in order to achieve the desired textures of food.
- Probiotics
-
Live microorganisms (bacteria or yeast) found in dietary supplements or food.
- Faecal microbiota transplantation
-
(FMT). Process of transferring faecal matter from one or many individuals to another in order to affect the microbiome of a recipient.
- Barley kernel-based bread
-
(BKB). Bread that is made from barley kernels, leading to high resistant starch and non-starch polysaccharide content.
- Postprandial glycaemic response
-
(PPGR). Increase of glucose level in the blood following ingestion of a meal.
- Nonalcoholic steatohepatitis
-
(NASH). A form of nonalcoholic fatty liver disease, characterized by at least 5% hepatic steatosis, with histological liver inflammation and hepatocyte injury.
- Primary bile acids
-
Amphipathic molecules produced by the hepatocytes and released to the intestine to aid the digestion and absorption of lipids.
- Glycaemic index
-
Numeric value, on a scale from 0 to 100, that represents the average glucose-level increase upon consumption of a particular food.
- Chylomicrons
-
Lipoprotein particles, composed of cholesterol, triglicerides, phospholipids and carrier proteins, that allow the transport of fat in the blood.
Rights and permissions
About this article
Cite this article
Kolodziejczyk, A.A., Zheng, D. & Elinav, E. Diet–microbiota interactions and personalized nutrition. Nat Rev Microbiol 17, 742–753 (2019). https://doi.org/10.1038/s41579-019-0256-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41579-019-0256-8
This article is cited by
-
Large-scale causal analysis of gut microbiota and six common complications of diabetes: a mendelian randomization study
Diabetology & Metabolic Syndrome (2024)
-
Deciphering the gut microbiome of grass carp through multi-omics approach
Microbiome (2024)
-
Cluster-specific associations between the gut microbiota and behavioral outcomes in preschool-aged children
Microbiome (2024)
-
Endogenous Coriobacteriaceae enriched by a high-fat diet promotes colorectal tumorigenesis through the CPT1A-ERK axis
npj Biofilms and Microbiomes (2024)
-
Forgetting how we ate: personalised nutrition and the strategic uses of history
History and Philosophy of the Life Sciences (2024)