Abstract
The discovery of chemosynthetic ecosystems at deep-sea hydrothermal vents in 1977 changed our view of biology. Chemosynthetic bacteria and archaea form the foundation of vent ecosystems by exploiting the chemical disequilibrium between reducing hydrothermal fluids and oxidizing seawater, harnessing this energy to fix inorganic carbon into biomass. Recent research has uncovered fundamental aspects of these microbial communities, including their relationships with underlying geology and hydrothermal geochemistry, interactions with animals via symbiosis and distribution both locally in various habitats within vent fields and globally across hydrothermal systems in diverse settings. Although ‘black smokers’ and symbioses between microorganisms and macrofauna attract much attention owing to their novelty and the insights they provide into life under extreme conditions, habitats such as regions of diffuse flow, subseafloor aquifers and hydrothermal plumes have important roles in the global cycling of elements through hydrothermal systems. Owing to sharp contrasts in physical and chemical conditions between these various habitats and their dynamic, extreme and geographically isolated nature, hydrothermal vents provide a valuable window into the environmental and ecological forces that shape microbial communities and insights into the limits, origins and evolution of microbial life.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Baker, M. C. et al. in Life in the World’s Oceans: Diversity, Distribution, and Abundance (ed. McIntyre, A. D.) 161–182 (Blackwell Publishing Ltd., 2010).
Dick, G. J. Genomic Approaches in Earth and Environmental Sciences (Wiley Blackwell, 2018).
Nakamura, K. & Takai, K. Theoretical constraints of physical and chemical properties of hydrothermal fluids on variations in chemolithotrophic microbial communities in seafloor hydrothermal systems. Prog. Earth Plan. Sci. 1, 5 (2014).
Baas Becking, L. Geobiologie of Inleiding Tot de Milieukunde [Dutch] (Den Haag: W. P. Van Stockum & Zoon, 1934).
Reysenbach, A. L., Banta, A. B., Boone, D. R., Cary, S. C. & Luther, G. W. Microbial essentials at hydrothermal vents. Nature 404, 835 (2000). This paper highlights the importance of microenvironments in defining microbial niches along the steep chemical gradients that are found at hydrothermal vents.
Kelley, D. S., Baross, J. A. & Delaney, J. R. Volcanoes, fluids, and life at mid-ocean ridge spreading centers. Ann. Rev. Earth Plan. Sci. 30, 385–491 (2002). This is a comprehensive and thorough review of the fundamentals of geology, geochemistry and biology of mid-ocean ridge spreading centres.
Orcutt, B. N., Sylvan, J. B., Knab, N. J. & Edwards, K. J. Microbial ecology of the dark ocean above, at, and below the seafloor. Microbiol. Mol. Biol. Rev. 75, 361–422 (2011).
Karl, D. M. The Microbiology of Deep-Sea Hydrothermal Vents (CRC Press, 1995).
Sievert, S. M. & Vetriani, C. Chemoautotrophy at deep-sea vents past, present, and future. Oceanography 25, 218–233 (2012). This paper critically reviews the current understanding and knowledge gaps regarding the physiology and metabolism of chemoautotrophs at deep-sea hydrothermal vents.
Edwards, K. J., Wheat, C. G. & Sylvan, J. B. Under the sea: microbial life in volcanic oceanic crust. Nat. Rev. Microbiol. 9, 703–712 (2011).
Dick, G. J. et al. Hydrothermal vent plume microbiology: ecological and biogeographic linkages to seafloor and water column habitats. Front. Microbiol. 4, 124 (2013).
Anantharaman, K., Breier, J. A. & Dick, G. J. Metagenomic resolution of microbial functions in deep-sea hydrothermal plumes across the Eastern Lau Spreading Center. ISME J. 10, 225–239 (2016).
Fortunato, C. S., Larson, B., Butterfield, D. A. & Huber, J. A. Spatially distinct, temporally stable microbial populations mediate biogeochemical cycling at and below the seafloor in hydrothermal vent fluids. Environ. Microbiol. 20, 769–784 (2018). This study is a good example of how a combination of molecular and geochemical approaches can provide insights into how metabolic functions are distributed across different microbial groups and how the distribution of microorganisms and metabolisms is linked to hydrothermal chemistry.
Stetter, K. O. Extremophiles and their adaptation to hot environments. FEBS Lett. 452, 22–25 (1999).
Takai, K. et al. Cell proliferation at 122 degrees C and isotopically heavy CH4 production by a hyperthermophilic methanogen under high-pressure cultivation. Proc. Natl Acad. Sci. USA 105, 10949–10954 (2008).
Kashefi, K. & Lovley, D. R. Extending the upper temperature limit for life. Science 301, 934 (2003).
Mora, M., Bellack, A., Ugele, M., Hopf, J. & Wirth, R. The temperature gradient-forming device, an accessory unit for normal light microscopes to study the biology of hyperthermophilic microorganisms. Appl. Environ. Microbiol. 80, 4764–4770 (2014).
Reysenbach, A. L. & Shock, E. Merging genomes with geochemistry in hydrothermal ecosystems. Science 296, 1077–1082 (2002).
Prieur, D., Erauso, G. & Jeanthon, C. Hyperthermophilic life at deep-sea hydrothermal vents. Planet. Space Sci. 43, 115–122 (1995).
Tuttle, J. H., Wirsen, C. O. & Jannasch, H. W. Microbial activities in the emitted hydrothermal waters of the Galapagos Rift vents. Mar. Biol. 73, 293–299 (1983).
Wirsen, C. O., Tuttle, J. H. & Jannasch, H. W. Activities of sulfur-oxidizing bacteria at the 21N East Pacific Rise vent site. Mar. Biol. 92, 449–456 (1986).
Reveillaud, J. et al. Subseafloor microbial communities in hydrogen-rich vent fluids from hydrothermal systems along the Mid-Cayman Rise. Environ. Microbiol. 18, 1970–1987 (2016).
Takai, K., Nakagawa, S., Reysenbach, A. L. & Hoek, J. in Back-Arc Spreading Systems: Geological, Biological, Chemical and Physical Interactions Vol. 166 (eds Christie, D. M., Fisher, C. R., Lee, S. M. & Givens, S.) 185–213 (American Geophysical Union, 2006).
Amend, J. P., McCollom, T. M., Hentscher, M. & Bach, W. Catabolic and anabolic energy for chemolithoautotrophs in deep-sea hydrothermal systems hosted in different rock types. Geochim. Cosmochim. Acta 75, 5736–5748 (2011).
Takai, K. & Nakamura, K. in Geomicrobiology: Molecular and Environmental Perspective (eds Loy, A., Mandl, M. & Barton, L. L.) 251–283 (Springer Netherlands, 2010).
McCollum, T. M. Geochemical constraints on primary productivity in submarine hydrothermal vent plumes. Deep Sea Res. Part I Oceanogr. Res. Pap. 47, 85–101 (2000).
McCollom, T. M. & Shock, E. L. Geochemical constraints on chemolithoautotrophic metabolism by microorganisms in seafloor hydrothermal systems. Geochim. Cosmochim. Acta 61, 4375–4391 (1997).
Reed, D. C. et al. Predicting the response of the deep-ocean microbiome to geochemical perturbations by hydrothermal vents. ISME J. 9, 1857–1869 (2015). This study shows how coupled hydrodynamic and thermodynamic models, in tandem with microbiological and physical oceanographic observations, can be used to understand the source, transport and growth of microorganisms in hydrothermal plumes.
Hugler, M. & Sievert, S. M. Beyond the Calvin cycle: autotrophic carbon fixation in the ocean. Ann. Rev. Mar. Sci. 3, 261–289 (2011).
Nunoura, T. et al. A primordial and reversible TCA cycle in a facultatively chemolithoautotrophic thermophile. Science 359, 559–563 (2018).
Goffredi, S. et al. Inorganic carbon acquisition by the hydrothermal vent tubeworm Riftia pachyptila depends upon high external PCO2 and upon proton-equivalent ion transport by the worm. J. Exp. Biol. 200, 883–896 (1997).
Schrenk, M. O., Kelley, D. S., Bolton, S. A. & Baross, J. A. Low archaeal diversity linked to subseafloor geochemical processes at the lost city hydrothermal field, Mid-Atlantic Ridge. Environ. Microbiol. 6, 1086–1095 (2004).
Mangiapia, M. et al. Proteomic and mutant analysis of the CO2 concentrating mechanism of hydrothermal vent chemolithoautotroph thiomicrospira crunogena. J. Bacteriol. 199, e00871–16 (2017).
Johnson, K. S., Childress, J. J. & Beehler, C. L. Short-term temperature variability in the rose garden hydrothermal vent field - an unstable deep-sea environment. Deep Sea Res. A 35, 1711–1721 (1988).
Campbell, B. J., Summers Engel, A., Porter, M. L. & Takai, K. The versatile ε-proteobacteria: key players in sulphidic habitats. Nat. Rev. Microbiol. 4, 458–468 (2006).
Koschinsky, A. et al. Hydrothermal venting at pressure-temperature conditions above the critical point of seawater, 5°S on the Mid-Atlantic Ridge. Geology 36, 615 (2008).
McDermott, J., Sylva, S. P., Ono, S., German, C. R. & Seewald, J. S. Geochemistry of fluids from Earth’s deepest ridge-crest hot-springs: Piccard hydrothermal field, Mid-Cayman Rise. Geochim. Cosmochim. Acta 228, 95–118 (2018).
LaRowe, D. E. et al. Modeling micorbial reaction rates in a submarine hydrothermal vent chimney wall. Geochim. Cosmochim. Acta 124, 72–97 (2014).
Tivey, M. K. in The Subseafloor Biosphere at Mid-Ocean Ridges (eds Wilcock, W. S. D., DeLong, E. F., Kelley, D. S., Baross, J. A. & Cary, S. C.) 137–152 (American Geophysical Union, 2004).
Flores, G. E. et al. Inter-field variability in the microbial communities of hydrothermal vent deposits from a back-arc basin. Geobiology 10, 333–346 (2012).
Takai, K. et al. Geochemical and microbiological evidence for a hydrogen-based, hyperthermophilic subsurface lithoautotrophic microbial ecosystem (HyperSLiME) beneath an active deep-sea hydrothermal field. Extremophiles 8, 269–282 (2004).
Brazelton, W. J., Nelson, B. & Schrenk, M. O. Metagenomic evidence for H2 oxidation and H2 production by serpentine-hosted subsurface microbial communities. Front. Microbiol. 2, 268 (2012).
Wirth, R. Colonization of black smokers by hyperthermophilic microorganisms. Trends Microbiol. 25, 92–99 (2017).
Huber, R., Stoffers, P., Cheminee, J. L., Richnow, H. H. & Stetter, K. O. Hyperthermophilic archaebacteria within the crater and open-sea plume of erupting Macdonald Seamount. Nature 345, 179–182 (1990).
Gonnella, G. et al. Endemic hydrothermal vent species identified in the open ocean seed bank. Nat. Microbiol. 1, 16086 (2016). This study presents evidence that taxa previously thought to be endemic to deep-sea hydrothermal vents are, in fact, present in ambient deep seawater, suggesting frequent dispersal of vent microorganisms between geographically distant vent sites.
Langseth, M. G., Becker, K., Herzen, R. P. V. & Schultheiss, P. Heat and fluid flux through sediment on the western flank of the Mid-Atlantic Ridge: a hydrogeological study of North Pond. Geophys. Res. Lett. 19, 517–520 (1992).
Huber, J. A. & Holden, J. F. in Magma to Microbe: Modeling Hydrothermal Processes at Ocean Spreading Centers (eds Lowell, R. P., Seewald, J. S., Metaxas, A. & Perfit, M. R.) 215–231 (American Geophysical Union, 2008).
McCollom, T. M. Energetic constraints on subsurface biomass production within igneous ocean crust. Geochem. Cosmochim. Acta 69, A200 (2005).
Cowen, J. P. et al. Fluids from aging ocean crust that support microbial life. Science 299, 120–123 (2003).
Santelli, C. M. et al. Abundance and diversity of microbial life in ocean crust. Nature 453, 653–656 (2008).
Walter, S. R. S. et al. Microbial decomposition of marine dissolved organic matter in cool oceanic crust. Nat. Geosci. 11, 334–339 (2018).
Schrenk, M. O., Huber, J. A. & Edwards, K. J. Microbial provinces in the subseafloor. Ann. Rev. Earth Plan. Sci. 2, 279–304 (2010).
Ishibashi, J., Okino, K. & Sunamura, M. Subseafloor Biosphere Linked to Hydrothermal Systems (Springer, Tokyo, 2015).
Huber, J. A., Butterfield, D. A. & Baross, J. A. Bacterial diversity in a subseafloor habitat following a deep-sea volcanic eruption. FEMS Microbiol. Ecol. 43, 393–409 (2003).
Perner, M. et al. In situ chemistry and microbial community compositions in five deep-sea hydrothermal fluid samples from Irina II in the Logatchev field. Environ. Microbiol. 15, 1551–1560 (2013).
Huber, J. A., Butterfield, D. A. & Baross, J. A. Temporal changes in archaeal diversity and chemistry in a mid-ocean ridge subseafloor habitat. Appl. Environ. Microbiol. 68, 1585–1594 (2002).
Vetriani, C. et al. Deep-sea hydrothermal vent epsilonproteobacteria encode a conserved and widespread nitrate reduction pathway (Nap). ISME J. 8, 1510–1521 (2014).
Burggraf, S., Jannasch, H. W., Nicolaus, B. & Stetter, K. O. Archaeoglobus profundus sp. nov., represents a new species within the sulfate-reducing Archaebacteria. Syst. Appl. Microbiol. 13, 24–28 (1990).
Deming, J. A. & Baross, J. A. Deep-sea smokers: windows to a subsurface biosphere? Geochim. Cosmochim. Acta 57, 3219–3230 (1993).
Holden, J. F. & Adams, M. W. Microbe-metal interactions in marine hydrothermal environments. Curr. Opin. Chem. Biol. 7, 160–165 (2003).
Juniper, S. K., Martineu, P., Sarrazin, J. & Gelinas, Y. Microbial-mineral floc associated with nascent hydrothermal activity on coaxial segment, Juan-De-Fuca Ridge. Geophys. Res. Lett. 22, 179–182 (1995).
Moussard, H., Corre, E., Cambon-Bonavita, M. A., Fouquet, Y. & Jeanthon, C. Novel uncultured Epsilonproteobacteria dominate a filamentous sulphur mat from the 13 degrees N hydrothermal vent field, East Pacific Rise. FEMS Microbiol. Ecol. 58, 449–463 (2006).
Sievert, S. M., Hugler, M., Taylor, C. D. & Wirsen, C. O. in Microbial Sulfur Metabolism (eds Dahl, C. & Friedrich, C. G.) 238–258 (Springer, 2008).
Meyer, J. L., Akerman, N. H., Proskurowski, G. & Huber, J. A. Microbiological characterization of post-eruption “snowblower” vents at Axial Seamount, Juan de Fuca Ridge. Front. Microbiol. 4, 153 (2013).
Emerson, D. et al. A novel lineage of proteobacteria involved in formation of marine Fe-oxidizing microbial mat communities. PLOS ONE 2, e667 (2007).
Girguis, P. R. & Lee, R. W. Thermal preference and tolerance of alvinellids. Science 312, 231 (2006).
Cary, S. C., Shank, T. & Stein, J. Worms bask in extreme temperatures. Nature 391, 545–546 (1998).
Dubilier, N., Bergin, C. & Lott, C. Symbiotic diversity in marine animals: the art of harnessing chemosynthesis. Nat. Rev. Microbiol. 6, 725–740 (2008).
Van Dover, C. L. The Ecology of Deep Sea Hydrothermal Vents (Princeton Univ. Press, 2000).
Gardebrecht, A. et al. Physiological homogeneity among the endosymbionts of Riftia pachyptila and Tevnia jerichonana revealed by proteogenomics. ISME J. 6, 766–776 (2012).
Robidart, J. C., Roque, A., Song, P. & Girguis, P. R. Linking hydrothermal geochemistry to organismal physiology: physiological versatility in Riftia pachyptila from sedimented and basalt-hosted vents. PLOS ONE 6, e21692 (2011).
Childress, J. J. & Girguis, P. R. The metabolic demands of endosymbiotic chemoautotrophic metabolism on host physiological capacities. J. Exp. Biol. 214, 312–325 (2011).
Van Dover, C. L. et al. Biogeography and ecological setting of Indian Ocean hydrothermal vents. Science 294, 818–823 (2001).
Petersen, J. M., Wentrup, C., Verna, C., Knittel, K. & Dubilier, N. Origins and evolutionary flexibility of chemosynthetic symbionts from deep-sea animals. Biol. Bull. 223, 123–137 (2012).
Bang, C. et al. Metaorganisms in extreme environments: do microbes play a role in organismal adaptation? Zoology 127, 1–19 (2018).
Petersen, J. M. et al. Hydrogen is an energy source for hydrothermal vent symbioses. Nature 476, 176–180 (2011).
Li, Y., Liles, M. R. & Halanych, K. M. Endosymbiont genomes yield clues of tubeworm success. ISME J. 12, 2785–2795 (2018).
Markert, S. et al. Physiological proteomics of the uncultured endosymbiont of Riftia pachyptila. Science 315, 247–250 (2007).
Robidart, J. C. et al. Metabolic versatility of the Riftia pachyptila endosymbiont revealed through metagenomics. Environ. Microbiol. 10, 727–737 (2008).
Stewart, F. J. & Cavanaugh, C. M. Symbiosis of thioautotrophic bacteria with Riftia pachyptila. Prog. Mol. Subcell. Biol. 41, 197–225 (2006).
Bright, M. & Bulgheresi, S. A complex journey: transmission of microbial symbionts. Nat. Rev. Microbiol. 8, 218–230 (2010).
Fisher, C. R., Brooks, J. M., Vodenichar, J. S., Zande, J. M. & Chidress, J. J. The co-occurrence of methanotrophic and chemoautotrophic sulfur-oxidizing bacterial symbionts in a deep-sea mussel. Mar. Ecol. 14, 277–289 (1993).
Beinart, R. A. et al. Evidence for the role of endosymbionts in regional-scale habitat partitioning by hydrothermal vent symbioses. Proc. Natl Acad. Sci. USA 109, E3241–E3250 (2012). This paper shows how microbial symbionts are linked to habitat partitioning of animals across different geochemical niches at deep-sea hydrothermal vents.
Sanders, J. G., Beinart, R. A., Stewart, F. J., Delong, E. F. & Girguis, P. R. Metatranscriptomics reveal differences in in situ energy and nitrogen metabolism among hydrothermal vent snail symbionts. ISME J. 7, 1556–1567 (2013).
Goffredi, S. K. Indigenous ectosymbiotic bacteria associated with diverse hydrothermal vent invertebrates. Environ. Microbiol. Rep. 2, 479–488 (2010).
Van Dover, C. L., Fry, B., Grassle, J. F., Humphris, S. & Rona, P. A. Feeding biology of the shrimp Rimicaris exoculata at hydrotheraml vents on the Mid-Atlantic Ridge. Mar. Biol. 98, 209–216 (1988).
Watsuji, T. O. et al. Molecular evidence of digestion and absorption of epibiotic bacterial community by deep-sea crab Shinkaia crosnieri. ISME J. 9, 821–831 (2015).
Goffredi, S. K., Waren, A., Orphan, V. J., Van Dover, C. L. & Vrijenhoek, R. C. Novel forms of structural integration between microbes and a hydrothermal vent gastropod from the Indian Ocean. Appl. Environ. Microbiol. 70, 3082–3090 (2004).
Sayavedra, L. et al. Abundant toxin-related genes in the genomes of beneficial symbionts from deep-sea hydrothermal vent mussels. eLife 4, e07966 (2015).
Lesniewski, R. A., Jain, S., Anantharaman, K., Schloss, P. D. & Dick, G. J. The metatranscriptome of a deep-sea hydrothermal plume is dominated by water column methanotrophs and lithotrophs. ISME J. 6, 2257–2268 (2012).
Baker, B. J., Lesniewski, R. A. & Dick, G. J. Genome-enabled transcriptomics reveals archaeal populations that drive nitrification in a deep-sea hydrothermal plume. ISME J. 6, 2269–2279 (2012).
Anantharaman, K., Breier, J. A., Sheik, C. S. & Dick, G. J. Evidence for hydrogen oxidation and metabolic plasticity in widespread deep-sea sulfur-oxidizing bacteria. Proc. Natl Acad. Sci. USA 110, 330–335 (2013).
Dick, G. J. et al. Enzymatic microbial Mn(II) oxidation and Mn biooxide production in the Guaymas Basin hydrothermal plume. Geochim. Cosmochim. Acta 73, 6517–6530 (2009).
Li, M. et al. Microbial iron uptake as a mechanism for dispersing iron from deep-sea hydrothermal vents. Nat. Commun. 6, 8933 (2014).
Levin, L. A. et al. Hydrothermal vents and methane seeps: rethinking the sphere of influence. Front. Mar. Sci. 3, 72 (2016).
Breier, J. A. et al. A large volume particulate and water multi-sampler with in situ preservation for microbial and biogeochemical studies. Deep Sea Res. Part I Oceanogr. Res. Pap. 94, 195–206 (2014).
Dick, G. J. & Tebo, B. M. Microbial diversity and biogeochemistry of the Guaymas Basin deep-sea hydrothermal plume. Environ. Microbiol. 12, 1334–1347 (2010).
German, C. R. et al. Diverse styles of submarine venting on the ultraslow spreading Mid-Cayman Rise. Proc. Natl Acad. Sci. USA 107, 14020–14025 (2010).
Sheik, C. S. et al. Spatially resolved sampling reveals dynamic microbial communities in rising hydrothermal plumes across a back-arc basin. ISME J. 9, 1434–1445 (2015).
Sylvan, J. B., Pyenson, B. C., Rouxel, O., German, C. R. & Edwards, K. J. Time-series analysis of two hydrothermal plumes at 9 degrees 50’N East Pacific Rise reveals distinct, heterogeneous bacterial populations. Geobiology 10, 178–192 (2012).
Cowen, J. P. et al. Geomicrobial transformation of manganese in Gorda Ridge event plumes. Deep Sea Res. Part II Top. Stud. Oceanogr. 45, 2713–2738 (1998).
Klose, J. et al. Endosymbionts escape dead hydrothermal vent tubeworms to enrich the free-living population. Proc. Natl Acad. Sci. USA 112, 11300–11305 (2015).
Summit, M. & Baross, J. A. A novel microbial habitat in the mid-ocean ridge subseafloor. Proc. Natl Acad. Sci. USA 98, 2158–2163 (2001).
Nakagawa, S. et al. Distribution, phylogenetic diversity and physiological characteristics of epsilon-Proteobacteria in a deep-sea hydrothermal field. Environ. Microbiol. 7, 1619–1632 (2005).
Opatkiewicz, A. D., Butterfield, D. A. & Baross, J. A. Individual hydrothermal vents at Axial Seamount harbor distinct subseafloor microbial communities. FEMS Microbiol. Ecol. 70, 413–424 (2009).
Huber, J. A. et al. Isolated communities of epsilonproteobacteria in hydrothermal vent fluids of the Mariana Arc seamounts. FEMS Microbiol. Ecol. 73, 538–549 (2010).
Huber, J. A., Butterfield, D. A. & Baross, J. A. Diversity and distribution of subseafloor thermococcales populations in diffuse hydrothermal vents at an active deep-sea volcano in the northeast Pacific Ocean. J. Geophys. Res. Biogeosci. 111, G04016 (2006).
Luther, G. W. III et al. Chemical speciation drives hydrothermal vent ecology. Nature 410, 813–816 (2001). This paper highlights the importance of chemical speciation, in this case, the availability of free sulfide, in structuring biological communities at deep-sea hydrothermal vents.
Larson, B. I., Houghton, J. L., Lowell, R. P., Farough, A. & Meile, C. D. Subsurface conditions in hydrothermal vents inferred from diffuse flow composition, and models of reaction and transport. Earth Planet. Sci. Lett. 424, 245–255 (2015).
Perner, M. et al. The influence of ultramafic rocks on microbial communities at the Logatchev hydrothermal field, located 15 degrees N on the Mid-Atlantic Ridge. FEMS Microbiol. Ecol. 61, 97–109 (2007).
Ver Eecke, H. C. et al. Hydrogen-limited growth of hyperthermophilic methanogens at deep-sea hydrothermal vents. Proc. Natl Acad. Sci. USA 109, 13674–13679 (2012).
Meier, D. V. et al. Niche partitioning of diverse sulfur-oxidizing bacteria at hydrothermal vents. ISME J. 11, 1545–1558 (2017). This paper shows how the niches of sulfur-oxidizing bacteria are partitioned according to the chemical environment across small spatial and temporal scales.
Coleman, M. L. & Chisholm, S. W. Code and context: Prochlorococcus as a model for cross-scale biology. Trends Microbiol. 15, 398–407 (2007).
Corliss, J. B. et al. Submarine thermal springs on the Galapagos Rift. Science 203, 1073–1083 (1979). This paper describes the discovery of deep-sea hydrothermal-vent ecosystems and lays out the hypothesis that chemosynthesis is the main source of primary production.
Einen, J., Thorseth, I. H. & Ovreas, L. Enumeration of archaea and bacteria in seafloor basalt using real-time quantitative PCR and fluorescence microscopy. FEMS Microbiol. Lett. 282, 182–187 (2008).
Sylvan, J. B., Toner, B. M. & Edwards, K. J. Life and death of deep-sea vents: bacterial diversity and ecosystem succession on inactive hydrothermal sulfides. mBio 3, e00279–11 (2012).
Mino, S. et al. Endemicity of the cosmopolitan mesophilic chemolithoautotroph Sulfurimonas at deep-sea hydrothermal vents. ISME J. 11, 909–919 (2017).
Hanson, C. A., Fuhrman, J. A., Horner-Devine, M. C. & Martiny, J. B. Beyond biogeographic patterns: processes shaping the microbial landscape. Nat. Rev. Microbiol. 10, 497–506 (2012).
Kelley, D. S. et al. A serpentinite-hosted ecosystem: the Lost City hydrothermal field. Science 307, 1428–1434 (2005).
Mccollom, T. M. Geochemical constraints on sources of metabolic energy for chemolithoautotrophy in ultramafic-hosted deep-sea hydrothermal systems. Astrobiology 7, 933–950 (2007).
Flores, G. E. et al. Microbial community structure of hydrothermal deposits from geochemically different vent fields along the Mid-Atlantic Ridge. Environ. Microbiol. 13, 2158–2171 (2011).
German, C. R. et al. Hydrothermal impacts on trace element and isotope ocean biogeochemistry. Philos. Trans. A Math. Phys. Eng. Sci. 374, 20160035 (2016).
Sunamura, M. & Yanagawa, K. in Subseafloor Biosphere Linked to Hydrothermal Systems (eds Ishibashi, J., Okino, K. & Sunamura, M.) 31–38 (Springer, 2015).
Mino, S. et al. Biogeography of persephonella in deep-sea hydrothermal vents of the Western Pacific. Front. Microbiol. 4, 107 (2013).
Anderson, R. E., Sogin, M. L. & Baross, J. A. Biogeography and ecology of the rare and abundant microbial lineages in deep-sea hydrothermal vents. FEMS Microbiol. Ecol. 91, 1–11 (2015).
Ochman, H. & Wilson, A. C. Evolution in bacteria - evidence for a universal substitution rate in cellular genomes. J. Mol. Evol. 26, 74–86 (1987).
Girguis, P. Here, there and everywhere. Nat. Microbiol. 1, 16123 (2016).
Whitaker, R. J., Grogan, D. W. & Taylor, J. W. Geographic barriers isolate endemic populations of hyperthermophilic archaea. Science 301, 976–978 (2003).
Govenar, B. Energy transfer through food webs at hydrothermal vents: linking the lithosphere to the biosphere. Oceanography 25, 246–255 (2012).
Bennett, S. A. et al. Trophic regions of a hydrothermal plume dispersing away from an ultramafic-hosted vent-system: Von Damm vent-site, Mid-Cayman Rise. Geochem. Geophys. Geosyst. 14, 317–327 (2013).
Beinart, R. A., Gartman, A., Sanders, J. G., Luther, G. W. & Girguis, P. R. The uptake and excretion of partially oxidized sulfur expands the repertoire of energy resources metabolized by hydrothermal vent symbioses. Proc. Biol. Sci. 282, 20142811 (2015).
Ortmann, A. C. & Suttle, C. A. High abundances of viruses in a deep-sea hydrothermal vent system indicates viral mediated microbial mortality. Deep Sea Res. Part I Oceanogr. Res. Pap. 52, 1515–1527 (2005).
Yoshida-Takashima, Y. et al. Spatial distribution of viruses associated with planktonic and attached microbial communities in hydrothermal environments. Appl. Environ. Microbiol. 78, 1311–1320 (2012).
Suttle, C. A. Marine viruses - major players in the global ecosystem. Nat. Rev. Microbiol. 5, 801–812 (2007).
Breitbart, M., Bonnain, C., Malki, K. & Sawaya, N. A. Phage puppet masters of the marine microbial realm. Nat. Microbiol. 3, 754–766 (2018).
Williamson, S. J. et al. Lysogenic virus-host interactions predominate at deep-sea diffuse-flow hydrothermal vents. ISME J. 2, 1112–1121 (2008).
Anantharaman, K. et al. Sulfur oxidation genes in diverse deep-sea viruses. Science 344, 757–760 (2014).
He, T. L., Li, H. Y. & Zhang, X. B. Deep-sea hydrothermal vent viruses compensate for microbial metabolism in virus-host interactions. mBio 8, e00893–17 (2017).
Elderfield, H. & Schultz, A. Mid-ocean ridge hydrothermal fluxes and the chemical composition of the ocean. Ann. Rev. Earth Plan. Sci. 24, 191–224 (1996).
Tagliabue, A. et al. Hydrothermal contribution to the oceanic dissolved iron inventory. Nat. Geosci. 3, 252–256 (2010).
Fitzsimmons, J. N. et al. Iron persistence in a distal hydrothermal plume supported by dissolved-particulate exchange. Nat. Geosci. 10, 195–201 (2017).
Toner, B. M. et al. Preservation of iron(II) by carbon-rich matrices in a hydrothermal plume. Nat. Geosci. 2, 197–201 (2009).
Cowen, J. P., Massoth, G. J. & Baker, E. T. Bacterial scavenging of Mn and Fe in a mid- to far-field hydrothermal particle plume. Nature 322, 169–171 (1986).
Feely, R. A., Trefry, J. H., Lebon, G. T. & German, C. R. The relationship between P/Fe and V/Fe ratios in hydrothermal precipitates and dissolved phosphate in seawater. Geophys. Res. Lett. 25, 2253–2256 (1998).
German, C. R. & Von Damm, K. L. in Treatise on Geochemistry Vol. 6 (eds Holland, H. D. & Turekian, K. K.) 181–222 (2004). This paper presents an accessible review of geological and geochemical processes in mid-ocean ridge hydrothermal systems and the impact of hydrothermal vents on the biogeochemistry of the oceans.
Kadko, D. An assessment of the effect of chemical scavenging within submarine hydrothermal plumes upon ocean geochemistry. Earth Plan. Sci. Lett. 120, 361–374 (1993).
Wankel, S. D. et al. Influence of subsurface biosphere on geochemical fluxes from diffuse hydrothermal fluids. Nat. Geosci. 4, 461–468 (2011). This paper shows the importance of in situ measurements in quantifying geochemical fluxes at hydrothermal vents.
Karl, D. M., Knauer, G. A., Martin, J. H. & Ward, B. B. Bacterial chemolithotrophy in the ocean is associated with sinking particles. Nature 309, 54–56 (1984).
Swan, B. K. et al. Potential for chemolithoautotrophy among ubiquitous bacteria lineages in the dark ocean. Science 333, 1296–12300 (2011).
Reed, D. C., Algar, C. K., Huber, J. A. & Dick, G. J. Gene-centric approach to integrating environmental genomics and biogeochemical models. Proc. Natl Acad. Sci. USA 111, 1879–1884 (2014).
Louca, S. et al. Integrating biogeochemistry with multiomic sequence information in a model oxygen minimum zone. Proc. Natl Acad. Sci. USA 113, E5925–E5933 (2016).
Dick, G. J. Embracing the mantra of modellers and synthesizing omics, experiments and models. Environ. Microbiol. Rep. 9, 18–20 (2017).
Nakajima, R. et al. Post-drilling changes in seabed landscape and megabenthos in a deep-sea hydrothermal system, the Iheya North field, Okinawa Trough. PLOS ONE 10, e0123095 (2015).
Van Dover, C. L. et al. Scientific rationale and international obligations for protection of active hydrothermal vent ecosystems from deep-sea mining. Mar. Policy 90, 20–28 (2018).
Boetius, A. & Haeckel, M. Mind the seafloor. Science 359, 34–36 (2018).
Lutz, R. A. & Kennish, M. J. Ecology of deep-sea hydrothermal vent communities: a review. Rev. Geophys. 31, 211–242 (1993).
Lonsdale, P. Clustering of suspension feeding macrobenthos near abyssal hydrothermal vents at oceanic spreading centers. Deep Sea Res. A 24, 857–858 (1977).
Ballard, R. D. Notes on a major oceanographic find. Oceanus 20, 35–44 (1977).
Jannasch, H. W. & Wirsen, C. O. Chemosynthetic primary production at East Pacific sea floor spreading centers. Bioscience 29, 592–598 (1979). This paper describes initial microbiological experiments and evidence for chemosynthesis as the main mode of primary production in deep-sea hydrothermal-vent ecosystems.
Jannasch, H. W. & Mottl, M. J. Geomicrobiology of deep-sea hydrothermal vents. Science 229, 717–725 (1985).
Cavanaugh, C. M., Gardiner, S. L., Jones, M. L., Jannasch, H. W. & Waterbury, J. B. Prokaryotic cells in the hydrothermal vent tube worm Riftia pachyptila Jones: possible chemoautotrophic symbionts. Science 213, 340–342 (1981).
Girguis, P. R. & Holden, J. F. On the potential for bioenergy and biofuels from hydrothermal vent microbes. Oceanography 25, 213–217 (2012).
Martin, W., Baross, J., Kelley, D. & Russell, M. J. Hydrothermal vents and the origin of life. Nat. Rev. Microbiol. 6, 805–814 (2008).
Corliss, J. B., Baross, J. A. & Hoffman, S. E. An hypothesis concerning the relationship between submarine hot springs and the origin of life on Earth. Oceanol. Acta 4, 59–69 (1981).
Baross, J. A. & Hoffman, S. E. Submarine hydrothermal vents and associated gradient environments as sites for origin and evolution of life. Orig. Life Evol. Biosph. 15, 327–345 (1985).
Russell, M. J. & Hall, A. J. The emergence of life from iron monosulphide bubbles at a submarine hydrothermal redox and pH front. J. Geol. Soc. London 154, 377–402 (1997).
Weiss, M. C. et al. The physiology and habitat of the last universal common ancestor. Nat. Microbiol. 1, 16116 (2016).
Dodd, M. S. et al. Evidence for early life in Earth’s oldest hydrothermal vent precipitates. Nature 543, 60–64 (2017).
Spang, A. et al. Complex archaea that bridge the gap between prokaryotes and eukaryotes. Nature 521, 173–179 (2015).
Holden, J. F., Breier, J. A., Rogers, K. L., Schulte, M. D. & Toner, B. M. Biogeochemical processes at hydrothermal vents: microbes and minerals, bioenergetics, and carbon fluxes. Oceanography 25, 196–208 (2012).
Mullineaux, L. S. et al. Exploring the ecology of deep-sea hydrothermal vents in a metacommunity framework. Front. Mar. Sci. 5, 49 (2018).
Adams, D. K., Arellano, S. M. & Govenar, B. Larval dispersal: vent life in the water column. Oceanography 25, 256–268 (2012).
Nakagawa, S. & Takai, K. Deep-sea vent chemoautotrophs: diversity, biochemistry, and ecological significance. FEMS Microbiol. Ecol. 65, 1–14 (2008).
Acknowledgements
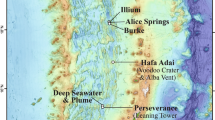
The author thanks C. German for input and S. Beaulieu for guidance about the map in figure 1. The author is grateful to the wonderful group of staff, graduate students, postdocs and collaborators who worked on vents with him and helped develop some of the ideas presented here: K. Anantharaman, B. Baker, S. Jain, R. Lesniewski, M. Li, D. Reed, C. Sheik, B. Toner and J. Breier. The Gordon and Betty Moore Foundation Marine Microbiology Initiative supported this research. The preparation of this manuscript was supported by an Alfred P. Sloan Foundation Fellowship in Ocean Sciences.
Reviewer information
Nature Reviews Microbiology thanks K. Takai and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Chemosynthesis
-
The use of chemical energy to fix inorganic carbon into organic carbon for microbial growth.
- Microbial mats
-
Dense, macroscopic communities of microorganisms attached to a solid surface.
- Chemolithoautotrophic growth
-
The growth of bacteria or archaea using an inorganic, chemical source of energy (for example, reduced forms of iron, sulfur, hydrogen and ammonia) to fix inorganic carbon into organic carbon.
- Reductive tricarboxylic acid cycle
-
(rTCA). A metabolic pathway for carbon fixation in which two molecules of carbon dioxide are converted into acetyl coenzyme A; it uses most of the same enzymes as the oxidative tricarboxylic acid cycle but runs it in reverse by using three alternative enzymes: fumarate reductase, 2-oxoglutarate synthase and ATP citrate lyase.
- Calvin–Benson–Bassham cycle
-
A carbon fixation pathway in which carbon dioxide is converted into glyceraldehyde-3-phosphate using the key enzyme Rubisco.
- Wood–Ljungdahl pathway
-
A metabolic pathway for carbon fixation in which two molecules of carbon dioxide are converted into acetyl coenzyme A by the key enzyme carbon monoxide dehydrogenase–acetyl coenzyme A synthase.
- Dicarboxylate–4-hydroxybutyrate pathway
-
A recently described carbon fixation pathway in archaea in which a molecule of bicarbonate (HCO3−) is fixed onto acetyl coenzyme A via a combination of enzymes from the reductive tricarboxylic acid cycle and the 4-hydroxybutyrate part of the 3-hydroxypropionate–4-hydroxybutyrate cycle.
- Black smokers
-
Hydrothermal vents that eject plumes of black suspended particles, chiefly iron sulfide minerals.
- White smokers
-
Hydrothermal vents that eject plumes of white suspended mineral particles composed mainly of calcium, barium and silica.
- Ultramafic
-
Relating to igneous rocks that have a very low silica content and very high content of magnesium and iron.
- Denitrifying
-
A type of process in which nitrate is used as an electron acceptor for energy metabolism and is reduced to di-nitrogen gas (denitrification).
- Mineral flocs
-
Flocculent materials sometimes emitted from hydrothermal vents, composed of fine mineral particles and often microbial biomass.
- Trophosome
-
An internal organ in tubeworms used to host chemosynthetic symbiotic bacteria.
- Autotrophy
-
The growth of an organism through the fixation of inorganic carbon into organic carbon.
- Heterotrophy
-
A mode of organism growth in which exogenous organic carbon functions as the source of carbon.
- Allopatric speciation
-
The evolutionary divergence of a population into distinct species due to geographic separation.
- Thermohaline conveyor
-
The global circulation of water in the oceans, driven by density gradients due to differences in the temperature and salinity of water masses.
Rights and permissions
About this article
Cite this article
Dick, G.J. The microbiomes of deep-sea hydrothermal vents: distributed globally, shaped locally. Nat Rev Microbiol 17, 271–283 (2019). https://doi.org/10.1038/s41579-019-0160-2
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41579-019-0160-2
This article is cited by
-
Diversity, habitat endemicity and trophic ecology of the fauna of Loki’s Castle vent field on the Arctic Mid-Ocean Ridge
Scientific Reports (2024)
-
Geography, not lifestyle, explains the population structure of free-living and host-associated deep-sea hydrothermal vent snail symbionts
Microbiome (2023)
-
The genome of a vestimentiferan tubeworm (Ridgeia piscesae) provides insights into its adaptation to a deep-sea environment
BMC Genomics (2023)
-
Strategies of chemolithoautotrophs adapting to high temperature and extremely acidic conditions in a shallow hydrothermal ecosystem
Microbiome (2023)
-
Microbe-driven elemental cycling enables microbial adaptation to deep-sea ferromanganese nodule sediment fields
Microbiome (2023)