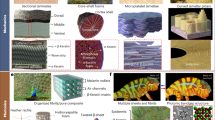
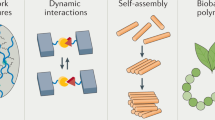
Abstract
Transition-metal coordination complexes are emerging as a broad class of supramolecular crosslinks that can be used to engineer the mechanical properties of advanced structural materials. Unlike conventional covalent bonds, metal-coordination bonds have the capacity to reform after rupture, thereby enabling dynamic, tunable and reversible (self-healing) mechanical properties. Several biological organisms, such as marine mussels, have been found to take advantage of these unique properties of metal-coordinate complexes in the assembly of load-bearing materials for complex extraorganismal functions. Accordingly, efforts to integrate metal-coordinate crosslinking in bioinspired synthetic protein and polymer hydrogels are an increasingly active area of research. However, a deeper understanding of how metal-coordination bonds affect bulk mechanical properties is still missing, rendering predicting the mechanical properties of metal-coordinated materials challenging. In this Review, we survey recent advances and open questions in our understanding of how chemical properties of metal-coordinate complexes influence multiscale mechanical behaviour, with the aim of presenting metal-coordination bonding as a rich, inorganic crosslinking chemistry tool. We also review applications of metal-coordinate crosslinking in the design of novel materials with tunable mechanical properties, ranging from tough gels to soft robots. These applications highlight the opportunities arising from the integration of this class of load-bearing crosslinks in structural materials design.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Williams, R. J. Metal ions in biological systems. Biol. Rev. 28, 381–412 (1953).
Sundberg, R. J. & Martin, R. B. Interactions of histidine and other imidazole derivatives with transition metal ions in chemical and biological systems. Chem. Rev. 74, 471–517 (1974).
Pyle, A. M. Metal ions in the structure and function of RNA. J. Biol. Inorg. Chem. 7, 679–690 (2002).
Waite, J. H. Adhesion à la moule. Integr. Comp. Biol. 42, 1172–1180 (2002).
Sun, C. J. & Waite, J. H. Mapping chemical gradients within and along a fibrous structural tissue, mussel byssal threads. J. Biol. Chem. 280, 39332–39336 (2005).
Holten-Andersen, N. et al. pH-induced metal-ligand cross-links inspired by mussel yield self-healing polymer networks with near-covalent elastic moduli. Proc. Natl Acad. Sci. USA 108, 2651–2655 (2011).
Fullenkamp, D. E., He, L., Barrett, D. G., Burghardt, W. R. & Messersmith, P. B. Mussel-inspired histidine-based transient network metal coordination hydrogels. Macromolecules 46, 1167–1174 (2013).
Li, C. H. et al. A highly stretchable autonomous self-healing elastomer. Nat. Chem. 8, 618–624 (2016).
Hartmann, M. A. & Fratzl, P. Sacrificial ionic bonds need to be randomly distributed to provide shear deformability. Nano Lett. 9, 3603–3607 (2009).
Broomell, C. C., Chase, S. F., Laue, T. & Waite, J. H. Cutting edge structural protein from the jaws of Nereis virens. Biomacromolecules 9, 1669–1677 (2008).
Broomell, C. C., Zok, F. W. & Waite, J. H. Role of transition metals in sclerotization of biological tissue. Acta Biomater. 4, 2045–2051 (2008).
Broomell, C. C., Mattoni, M. A., Zok, F. W. & Waite, J. H. Critical role of zinc in hardening of Nereis jaws. J. Exp. Biol. 209, 3219–3225 (2006).
Waite, J. H., Andersen, N. H., Jewhurst, S. & Sun, C. Mussel adhesion: finding the tricks worth mimicking. J. Adhes. 81, 297–317 (2005).
Degtyar, E., Harrington, M. J., Politi, Y. & Fratzl, P. The mechanical role of metal ions in biogenic protein-based materials. Angew. Chem. Int. Ed. 53, 12026–12044 (2014).
Wegst, U. G. K., Bai, H., Saiz, E., Tomsia, A. P. & Ritchie, R. O. Bioinspired structural materials. Nat. Mater. 14, 23–36 (2015).
Launey, M. E. & Ritchie, R. O. On the fracture toughness of advanced materials. Adv. Mater. 21, 2103–2110 (2009).
Ritchie, R. O. The conflicts between strength and toughness. Nat. Mater. 10, 817–822 (2011).
Creton, C. 50th anniversary perspective: Networks and gels: Soft but dynamic and tough. Macromolecules 50, 8297–8316 (2017).
Fantner, G. E. et al. Sacrificial bonds and hidden length dissipate energy as mineralized fibrils separate during bone fracture. Nat. Mater. 4, 612–616 (2005).
Barthelat, F., Yin, Z. & Buehler, M. J. Structure and mechanics of interfaces in biological materials. Nat. Rev. Mater. 1, 16007 (2016).
Gu, G. X., Takaffoli, M. & Buehler, M. J. Hierarchically enhanced impact resistance of bioinspired composites. Adv. Mater. 29, 1700060 (2017).
Holten-Andersen, N. et al. Metal-coordination: using one of nature’s tricks to control soft material mechanics. J. Mater. Chem. B 2, 2467–2472 (2014).
Grindy, S. C. et al. Control of hierarchical polymer mechanics with bioinspired metal-coordination dynamics. Nat. Mater. 14, 1210–1216 (2015).
Mozhdehi, D. et al. Tuning dynamic mechanical response in metallopolymer networks through simultaneous control of structural and temporal properties of the networks. Macromolecules 49, 6310–6321 (2016).
Harrington, M. J., Masic, A., Holten-Andersen, N., Waite, J. H. & Fratzl, P. Iron-clad fibers: a metal-based biological strategy for hard flexible coatings. Science 328, 216–220 (2010).
Grindy, S. C., Lenz, M. & Holten-Andersen, N. Engineering elasticity and relaxation time in metal-coordinate cross-linked hydrogels. Macromolecules 49, 8306–8312 (2016).
Li, H., Yang, P., Pageni, P. & Tang, C. Recent advances in metal-containing polymer hydrogels. Macromol. Rapid Commun. 38, 1700109 (2017).
Cazzell, S. A. & Holten-Andersen, N. Expanding the stoichiometric window for metal cross-linked gel assembly using competition. Proc. Natl Acad. Sci. USA 22, 201906349 (2019).
Buehler, M. J. Materials by design — A perspective from atoms to structures. MRS Bull. 38, 169–176 (2013).
Giesa, T., Jagadeesan, R., Spivak, D. I. & Buehler, M. J. Matriarch: a Python library for materials architecture. ACS Biomater. Sci. Eng. 1, 1009–1015 (2015).
Huang, W. et al. Synergistic integration of experimental and simulation approaches for the de novo design of silk-based materials. Acc. Chem. Res. 50, 866–876 (2017).
Mozhdehi, D., Ayala, S., Cromwell, O. R. & Guan, Z. Self-healing multiphase polymers via dynamic metal–ligand interactions. J. Am. Chem. Soc. 136, 16128–16131 (2014).
Zheng, S. Y. et al. Metal-coordination complexes mediated physical hydrogels with high toughness, stick-slip tearing behavior, and good processability. Macromolecules 49, 9637–9646 (2016).
Tang, Q., Zhao, D., Yang, H., Wang, L. & Zhang, X. A pH-responsive self-healing hydrogel based on multivalent coordination of Ni2+ with polyhistidine-terminated PEG and IDA-modified oligochitosan. J. Mater. Chem. B 7, 30–42 (2019).
Yeo, J. et al. Multiscale modeling of keratin, collagen, elastin and related human diseases: Perspectives from atomistic to coarse-grained molecular dynamics simulations. Extrem. Mech. Lett. 20, 112–124 (2018).
Bertoldi, K. & Boyce, M. C. Mechanics of the hysteretic large strain behavior of mussel byssus threads. J. Mater. Sci. 42, 8943–8956 (2007).
Cohen, N., Waite, J. H., McMeeking, R. M. & Valentine, M. T. Force distribution and multiscale mechanics in the mussel byssus. Philos. Trans. R. Soc. B Biol. Sci. 374, 20190202 (2019).
Priemel, T., Degtyar, E., Dean, M. N. & Harrington, M. J. Rapid self-assembly of complex biomolecular architectures during mussel byssus biofabrication. Nat. Commun. 8, 14539 (2017).
Bell, E. C. & Gosline, J. M. Mechanical design of mussel byssus: material yield enhances attachment strength. J. Exp. Biol. 199, 1005–1017 (1996).
Carrington, E. & Gosline, J. M. Mechanical design of mussel byssus: Load cycle and strain rate dependence. Am. Malacol. Bull. 18, 135–142 (2004).
Waite, J. H., Lichtenegger, H. C., Stucky, G. D. & Hansma, P. Exploring molecular and mechanical gradients in structural bioscaffolds. Biochemistry 43, 7653–7662 (2004).
Qin, Z. & Buehler, M. J. Impact tolerance in mussel thread networks by heterogeneous material distribution. Nat. Commun. 4, 2187 (2013).
Zhao, H. & Waite, J. H. Proteins in load-bearing junctions: the histidine-rich metal-binding protein of mussel byssus. Biochemistry 45, 14223–14231 (2006).
Harrington, M. J. & Waite, J. H. Holdfast heroics: comparing the molecular and mechanical properties of Mytilus californianus byssal threads. J. Exp. Biol. 210, 4307–4318 (2007).
Vaccaro, E. & Waite, J. H. Yield and post-yield behavior of mussel byssal thread: A self-healing biomolecular material. Biomacromolecules 2, 906–911 (2001).
Zechel, S., Hager, M., Priemel, T. & Harrington, M. Healing through histidine: Bioinspired pathways to self-healing polymers via imidazole–metal coordination. Biomimetics 4, 20 (2019).
Schmitt, C. N. Z., Politi, Y., Reinecke, A. & Harrington, M. J. Role of sacrificial protein–metal bond exchange in mussel byssal thread self-healing. Biomacromolecules 16, 2852–2861 (2015).
Schmidt, S. et al. Metal-mediated molecular self-healing in histidine-rich mussel peptides. Biomacromolecules 15, 1644–1652 (2014).
Krauss, S., Metzger, T. H., Fratzl, P. & Harrington, M. J. Self-repair of a biological fiber guided by an ordered elastic framework. Biomacromolecules 14, 1520–1528 (2013).
Reinecke, A., Bertinetti, L., Fratzl, P. & Harrington, M. J. Cooperative behavior of a sacrificial bond network and elastic framework in providing self-healing capacity in mussel byssal threads. J. Struct. Biol. 196, 329–339 (2016).
Claussen, K. U., Giesa, R., Scheibel, T. & Schmidt, H. W. Learning from nature: Synthesis and characterization of longitudinal polymer gradient materials inspired by mussel byssus threads. Macromol. Rapid Commun. 33, 206–211 (2011).
Holten-Andersen, N. et al. Metals and the integrity of a biological coating: the cuticle of mussel byssus. Langmuir 25, 3323–3326 (2009).
Saiz-Poseu, J., Mancebo-Aracil, J., Nador, F., Busqué, F. & Ruiz-Molina, D. The chemistry behind catechol-based adhesion. Angew. Chem. Int. Ed. Engl. 58, 696–714 (2019).
Hofman, A. H., van Hees, I. A., Yang, J. & Kamperman, M. Bioinspired underwater adhesives by using the supramolecular toolbox. Adv. Mater. 30, 1704640 (2018).
Kord Forooshani, P. & Lee, B. P. Recent approaches in designing bioadhesive materials inspired by mussel adhesive protein. J. Polym. Sci. A Polym. Chem. 55, 9–33 (2017).
Lichtenegger, H. C. et al. Zinc and mechanical prowess in the jaws of Nereis, a marine worm. Proc. Natl Acad. Sci. USA 100, 9144–9149 (2003).
Lichtenegger, H. C., Schoberl, T., Bartl, M. H., Waite, H. & Stucky, G. D. High abrasion resistance with sparse mineralization: copper biomineral in worm jaws. Science 298, 389–392 (2002).
Khan, R. K., Stoimenov, P. K., Mates, T. E., Waite, J. H. & Stucky, G. D. Exploring gradients of halogens and zinc in the surface and subsurface of Nereis jaws. Langmuir 22, 8465–8471 (2006).
Chou, C. C. et al. Ion effect and metal-coordinated cross-linking for multiscale design of Nereis jaw inspired mechanomutable materials. ACS Nano 11, 1858–1868 (2017).
Politi, Y. et al. A spider’s fang: How to design an injection needle using chitin-based composite material. Adv. Funct. Mater. 22, 2519–2528 (2012).
Schofield, R. M. S., Nesson, M. H., Richardson, K. A. & Wyeth, P. Zinc is incorporated into cuticular “tools” after ecdysis: The time course of the zinc distribution in “tools” and whole bodies of an ant and a scorpion. J. Insect Physiol. 49, 31–44 (2003).
Horbelt, N., Eder, M., Bertinetti, L., Fratzl, P. & Harrington, M. J. Unraveling the rapid assembly process of stiff cellulosic fibers from mistletoe berries. Biomacromolecules 20, 3094–3103 (2019).
Pasche, D. et al. A new twist on sea silk: the peculiar protein ultrastructure of fan shell and pearl oyster byssus. Soft Matter 14, 5654–5664 (2018).
Fung, T.-M., Gallego Lazo, C. & Smith, A. M. Elasticity and energy dissipation in the double network hydrogel adhesive of the slug Arion subfuscus. Philos. Trans. R. Soc. B Biol. Sci. 374, 20190201 (2019).
Lipscomb, W. N. & Sträter, N. Recent advances in zinc enzymology. Chem. Rev. 96, 2375–2433 (1996).
Hromada, S. E. et al. Protein oxidation involved in Cys-Tyr post-translational modification. J. Inorg. Biochem. 176, 168–174 (2017).
Birkedal, H. et al. Halogenated veneers: Protein cross-linking and halogenation in the jaws of Nereis, a marine polychaete worm. ChemBioChem 7, 1392–1399 (2006).
Yu, M., Hwang, J. & Deming, T. J. Role of 1-3,4-dihydroxyphenylalanine in mussel adhesive proteins. J. Am. Chem. Soc. 121, 5825–5826 (1999).
Yamamoto, H. Synthesis and adhesive studies of marine polypeptides. J. Chem. Soc. Perkin Trans. 1 1987, 613–618 (1987).
Parada, G. A. & Zhao, X. Ideal reversible polymer networks. Soft Matter 14, 5186–5196 (2018).
Tang, Q. et al. Polyhistidine-based metal coordination hydrogels with physiologically relevant pH responsiveness and enhanced stability through a novel synthesis. Macromol. Rapid Commun. 39, e1800109 (2018).
Yu, M. & Deming, T. J. Synthetic polypeptide mimics of marine adhesives. Macromolecules 31, 4739–4745 (1998).
Sanoja, G. E. et al. Ion transport in dynamic polymer networks based on metal–ligand coordination: effect of cross-linker concentration. Macromolecules 51, 2017–2026 (2018).
Guo, W. et al. Plant oil and amino acid-derived elastomers with rapid room temperature self-healing ability. J. Mater. Chem. A 7, 21927–21933 (2019).
Srivastava, A., Holten-Andersen, N., Stucky, G. D. & Waite, J. H. Ragworm jaw-inspired metal ion cross-linking for improved mechanical properties of polymer blends. Biomacromolecules 9, 2873–2880 (2008).
Enke, M. et al. Self-healing response in supramolecular polymers based on reversible zinc–histidine interactions. Polymer 69, 274–282 (2015).
Enke, M. et al. A translation of the structure of mussel byssal threads into synthetic materials by the utilization of histidine-rich block copolymers. Polym. Chem. 9, 3543–3551 (2018).
Wang, R. et al. Classical challenges in the physical chemistry of polymer networks and the design of new materials. Acc. Chem. Res. 49, 2786–2795 (2016).
Rossow, T. & Seiffert, S. Supramolecular polymer gels with potential model-network structure. Polym. Chem. 5, 3018–3029 (2014).
Tang, S. & Olsen, B. D. Relaxation processes in supramolecular metallogels based on histidine–nickel coordination bonds. Macromolecules 49, 9163–9175 (2016).
Yu, W. et al. Tuning of the dynamics of metal ion crosslinked hydrogels by network structures. Soft Matter 15, 4423–4427 (2019).
Ahmadi, M., Jangizehi, A., van Ruymbeke, E. & Seiffert, S. Deconvolution of the effects of binary associations and collective assemblies on the rheological properties of entangled side-chain supramolecular polymer networks. Macromolecules 52, 5255–5267 (2019).
Hwang, D. S., Yoo, H. J., Jun, J. H., Moon, W. K. & Cha, H. J. Expression of functional recombinant mussel adhesive protein Mgfp-5 in Escherichia coli. Appl. Environ. Microbiol. 70, 3352–3359 (2004).
Hwang, D. S., Sim, S. B. & Cha, H. J. Cell adhesion biomaterial based on mussel adhesive protein fused with RGD peptide. Biomaterials 28, 4039–4046 (2007).
Kim, B. J., Choi, Y. S. & Cha, H. J. Reinforced multifunctionalized nanofibrous scaffolds using mussel adhesive proteins. Angew. Chem. Int. Ed. Engl. 51, 675–678 (2012).
Kim, B. J. et al. Mussel-inspired adhesive protein-based electrospun nanofibers reinforced by Fe(III)–DOPA complexation. J. Mater. Chem. B 3, 112–118 (2013).
Kim, B. J. et al. Mussel-mimetic protein-based adhesive hydrogel. Biomacromolecules 15, 1579–1585 (2014).
Gupta, M. K. et al. Programmable mechanical properties from a worm jaw-derived biopolymer through hierarchical ion exposure. ACS Appl. Mater. Interfaces 10, 31928–31937 (2018).
Statz, A. R., Meagher, R. J., Barron, A. E. & Messersmith, P. B. New peptidomimetic polymers for antifouling surfaces. J. Am. Chem. Soc. 127, 7972–7973 (2005).
Yang, W., Gao, X., Springsteen, G. & Wang, B. Catechol pendant polystyrene for solid-phase synthesis. Tetrahedron Lett. 43, 6339–6342 (2002).
Trapaidze, A., D’Antuono, M., Fratzl, P. & Harrington, M. J. Exploring mussel byssus fabrication with peptide-polymer hybrids: Role of pH and metal coordination in self-assembly and mechanics of histidine-rich domains. Eur. Polym. J. 109, 229–236 (2018).
Jehle, F., Fratzl, P. & Harrington, M. J. Metal-tunable self-assembly of hierarchical structure in mussel-inspired peptide films. ACS Nano 12, 2160–2168 (2018).
Wang, X.-W., Liu, D., Yin, G.-Z. & Zhang, W.-B. in Bioinspired Materials Science and Engineering Ch. 15 (eds Yang, G., Xiao, L. & Lamboni, L.) 295–309 (Wiley, 2018).
Reinecke, A., Brezesinski, G. & Harrington, M. J. pH-responsive self-organization of metal-binding protein motifs from biomolecular junctions in mussel byssus. Adv. Mater. Interfaces 4, 1600416 (2017).
Przybyla, D. E. & Chmielewski, J. Metal-triggered collagen peptide disk formation. J. Am. Chem. Soc. 132, 7866–7867 (2010).
Salgado, E. N. et al. Metal templated design of protein interfaces. Proc. Natl Acad. Sci. USA 107, 1827–1832 (2010).
Salgado, E. N., Radford, R. J. & Tezcan, F. A. Metal-directed protein self-assembly. Acc. Chem. Res. 43, 661–672 (2010).
Brodin, J. D. et al. Metal-directed, chemically tunable assembly of one-, two- and three-dimensional crystalline protein arrays. Nat. Chem. 4, 375–382 (2012).
Tunn, I., Harrington, M. J. & Blank, K. G. Bioinspired histidine–Zn2+ coordination for tuning the mechanical properties of self-healing coiled coil cross-linked hydrogels. Biomimetics 4, 25 (2019).
Sun, W. et al. Molecular engineering of metal coordination interactions for strong, tough, and fast-recovery hydrogels. Sci. Adv. 6, eaaz9531 (2020).
Tunn, I., de Léon, A. S., Blank, K. G. & Harrington, M. J. Tuning coiled coil stability with histidine-metal coordination. Nanoscale 10, 22725–22729 (2018).
Cao, Y., Wei, X., Lin, Y. & Sun, F. Synthesis of bio-inspired viscoelastic molecular networks by metal-induced protein assembly. Mol. Syst. Des. Eng. 5, 117–124 (2020).
Mirabello, C. & Wallner, B. rawMSA: proper Deep Learning makes protein sequence profiles and feature extraction obsolete. Preprint at https://www.biorxiv.org/content/10.1101/394437v1 (2018).
Yu, C. H., Qin, Z., Martin-Martinez, F. J. & Buehler, M. J. A self-consistent sonification method to translate amino acid sequences into musical compositions and application in protein design using artificial intelligence. ACS Nano 13, 7471–7482 (2019).
Sjöberg, S. Critical evaluation of stability constants of metal-imidazole and metal-histamine systems (Technical Report). Pure Appl. Chem. 69, 1549–1570 (1997).
Irving, H. & Williams, R. J. P. 637. The stability of transition-metal complexes. J. Chem. Soc. https://doi.org/10.1039/JR9530003192 (1953).
Remko, M., Fitz, D. & Rode, B. M. Effect of metal ions (Li+, Na+, K+, Mg2+, Ca2+, Ni2+, Cu2+ and Zn2+) and water coordination on the structure and properties of l-histidine and zwitterionic l-histidine. Amino Acids 39, 1309–1319 (2010).
Smith, R. M. & Martell, A. E. Critical Stability Constants Vol. 6 (Springer, 1976).
Gensler, M. et al. Mechanical rupture of mono- and bivalent transition metal complexes in experiment and theory. J. Phys. Chem. C 119, 4333–4343 (2015).
Li, P. & Merz, K. M. Metal ion modeling using classical mechanics. Chem. Rev. 117, 1564–1686 (2017).
Duboué-Dijon, E., Mason, P. E., Fischer, H. E. & Jungwirth, P. Hydration and ion pairing in aqueous Mg2+ and Zn2+ solutions: force-field description aided by neutron scattering experiments and ab initio molecular dynamics simulations. J. Phys. Chem. B 122, 3296–3306 (2018).
Rodríguez-Santiago, L., Alí-Torres, J., Vidossich, P. & Sodupe, M. Coordination properties of a metal chelator clioquinol to Zn2+ studied by static DFT and ab initio molecular dynamics. Phys. Chem. Chem. Phys. 17, 13582–13589 (2015).
Assifaoui, A. et al. Structural behaviour differences in low methoxy pectin solutions in the presence of divalent cations (Ca2+ and Zn2+): A process driven by the binding mechanism of the cation with the galacturonate unit. Soft Matter 11, 551–560 (2015).
Tetteh, S. Coordination behavior of Ni2+, Cu2+, and Zn2+ in tetrahedral 1-methylimidazole complexes: A DFT/CSD study. Bioinorg. Chem. Appl. https://doi.org/10.1155/2018/3157969 (2018).
Senftle, T. P. et al. The ReaxFF reactive force-field: development, applications and future directions. npj Comput. Mater. 2, 15011 (2016).
Raymand, D., van Duin, A. C. T., Baudin, M. & Hermansson, K. A reactive force field (ReaxFF) for zinc oxide. Surf. Sci. 602, 1020–1031 (2008).
Van Duin, A. C. T. et al. Development and validation of a ReaxFF reactive force field for Cu cation/water interactions and copper metal/metal oxide/metal hydroxide condensed phases. J. Phys. Chem. A 114, 9507–9514 (2010).
Huang, L., Joshi, K. L., van Duin, A. C. T., Bandosz, T. J. & Gubbins, K. E. ReaxFF molecular dynamics simulation of thermal stability of a Cu3(BTC)2 metal–organic framework. Phys. Chem. Chem. Phys. 14, 11327–11332 (2012).
Zeng, L. et al. A highly stretchable, tough, fast self-healing hydrogel based on peptide–metal ion coordination. Biomimetics 4, 36 (2019).
Shabbir, H. & Hartmann, M. A. Influence of reversible cross-link coordination on the mechanical behavior of a linear polymer chain. New J. Phys. 19, 093024 (2017).
Shabbir, H., Dellago, C. & Hartmann, M. A high coordination of cross-links is beneficial for the strength of cross-linked fibers. Biomimetics 4, 12 (2019).
Rulíšek, L. & Vondrášek, J. Coordination geometries of selected transition metal ions (Co2+, Ni2+, Cu2+, Zn2+, Cd2+, and Hg2+) in metalloproteins. J. Inorg. Biochem. 71, 115–127 (1998).
Sever, M. J. & Wilker, J. J. Absorption spectroscopy and binding constants for first-row transition metal complexes of a DOPA-containing peptide. Dalton Trans. https://doi.org/10.1039/b509586g (2006).
Li, Y. et al. Single-molecule mechanics of catechol-iron coordination bonds. ACS Biomater. Sci. Eng. 3, 979–989 (2017).
Xu, Z. Mechanics of metal-catecholate complexes: The roles of coordination state and metal types. Sci. Rep. 3, 2914 (2013).
Langford, C. H. & Gray, H. B. Ligand Substitution Processes (W. A. Benjamin, 1966).
Li, C. P. & Du, M. Role of solvents in coordination supramolecular systems. Chem. Commun. 47, 5958–5972 (2011).
Redfern, P. C., Zapol, P., Curtiss, L. A., Rajh, T. & Thurnauer, M. C. Computational studies of catechol and water interactions with titanium oxide nanoparticles. J. Phys. Chem. B 107, 11419–11427 (2003).
Helm, L. & Merbach, A. E. Inorganic and bioinorganic solvent exchange mechanisms. Chem. Rev. 105, 1923–1959 (2005).
Buck, C. C. et al. Anion-mediated effects on the size and mechanical properties of enzymatically crosslinked suckerin hydrogels. Macromol. Biosci. 19, e1800238 (2019).
Maier, G. P., Rapp, M. V., Waite, J. H., Israelachvili, J. N. & Butler, A. Adaptive synergy between catechol and lysine promotes wet adhesion by surface salt displacement. Science 349, 628–632 (2015).
Li, Y. et al. Single-molecule study of the synergistic effects of positive charges and Dopa for wet adhesion. J. Mater. Chem. B 5, 4416–4420 (2017).
Li, Y. et al. Hidden complexity of synergistic roles of Dopa and lysine for strong wet adhesion. Mater. Chem. Front. 1, 2664–2668 (2017).
Sever, M. J., Weisser, J. T., Monahan, J., Srinivasan, S. & Wilker, J. J. Metal-mediated cross-linking in the generation of a marine-mussel adhesive. Angew. Chem. Int. Ed. Engl. 43, 448–450 (2004).
Barrett, D. G. et al. pH-based regulation of hydrogel mechanical properties through mussel-inspired chemistry and processing. Adv. Funct. Mater. 23, 1111–1119 (2013).
Auletta, J. T. et al. Stimuli-responsive iron-cross-linked hydrogels that undergo redox-driven switching between hard and soft states. Macromolecules 48, 1736–1747 (2015).
Mou, C., Ali, F., Malaviya, A. & Bettinger, C. J. Electrochemical-mediated gelation of catechol-bearing hydrogels based on multimodal crosslinking. J. Mater. Chem. B 7, 1690–1696 (2019).
Wegner, S. V., Schenk, F. C., Witzel, S., Bialas, F. & Spatz, J. P. Cobalt cross-linked redox-responsive PEG hydrogels: from viscoelastic liquids to elastic solids. Macromolecules 49, 4229–4235 (2016).
Okamoto, S. & Eltis, L. D. The biological occurrence and trafficking of cobalt. Metallomics 3, 963–970 (2011).
Outten, F. W. & Theil, E. C. Iron-based redox switches in biology. Antioxid. Redox Signal. 11, 1029–1046 (2009).
Campanella, A., Döhler, D. & Binder, W. H. Self-healing in supramolecular polymers. Macromol. Rapid Commun. 39, 1700739 (2018).
Duboué-Dijon, E., Mason, P. E., Fischer, H. E. & Jungwirth, P. Changes in the hydration structure of imidazole upon protonation: Neutron scattering and molecular simulations. J. Chem. Phys. 146, 185102 (2017).
Kim, S., Peterson, A. M. & Holten-Andersen, N. Enhanced water retention maintains energy dissipation in dehydrated metal-coordinate polymer networks: another role for Fe-catechol cross-links? Chem. Mater. 30, 3648–3655 (2018).
Liu, Q. et al. Probing the reversible Fe3+–DOPA-mediated bridging interaction in mussel foot protein-1. J. Phys. Chem. C 120, 21670–21677 (2016).
Castillo-Tejas, J. et al. Associative polymers. Part III: Shear rheology from molecular dynamics. Colloids Surf. A Physicochem. Eng. Asp. 491, 37–49 (2016).
Castillo-Tejas, J., Carro, S. & Manero, O. Shear banding in telechelic associative polymers by molecular dynamics. ACS Macro Lett. 6, 190–193 (2017).
Liu, D., Liu, F., Zhou, W., Chen, F. & Wei, J. Molecular dynamics simulation of self-assembly and viscosity behavior of PAM and CTAC in salt-added solutions. J. Mol. Liq. 268, 131–139 (2018).
Zhang, Q. et al. Disassociation and reformation under strain in polymer with dynamic metal–ligand coordination cross-linking. Macromolecules 52, 660–668 (2019).
Song, J. et al. Programmable anisotropy and percolation in supramolecular patchy particle gels. ACS Nano, 1–10 https://doi.org/10.1021/acsnano.0c06389 (2020).
Sénéchal, K., Maury, O., Le Bozec, H., Ledoux, I. & Zyss, J. Zinc(II) as a versatile template for the design of dipolar and octupolar NLO-phores. J. Am. Chem. Soc. 124, 4560–4561 (2002).
Qin, H. et al. Dynamic Au-thiolate interaction induced rapid self-healing nanocomposite hydrogels with remarkable mechanical behaviors. Chem 3, 691–705 (2017).
Menyo, M. S., Hawker, C. J. & Waite, J. H. Versatile tuning of supramolecular hydrogels through metal complexation of oxidation-resistant catechol-inspired ligands. Soft Matter 9, 10314–10323 (2013).
Hwang, D. S., Zeng, H., Lu, Q., Israelachvili, J. & Waite, J. H. Adhesion mechanism in a DOPA-deficient foot protein from green mussels. Soft Matter 8, 5640–5648 (2012).
Budisa, N. & Schneider, T. Expanding the DOPA universe with genetically encoded, mussel-inspired bioadhesives for material sciences and medicine. ChemBioChem 20, 2163–2190 (2019).
Zhan, K., Kim, C., Sung, K., Ejima, H. & Yoshie, N. Tunicate-inspired gallol polymers for underwater adhesive: a comparative study of catechol and gallol. Biomacromolecules 18, 2959–2966 (2017).
Liu, F. et al. Gallol-containing homopolymers and block copolymers: ROMP synthesis and gelation properties by metal-coordination and oxidation. Polymer 143, 212–227 (2018).
Wang, D. P. et al. Distinct mechanical and self-healing properties in two polydimethylsiloxane coordination polymers with fine-tuned bond strength. Inorg. Chem. 57, 3232–3242 (2018).
Lai, J. C. et al. Thermodynamically stable whilst kinetically labile coordination bonds lead to strong and tough self-healing polymers. Nat. Commun. 10, 1164 (2019).
Park, J. P. et al. Vanadyl-catecholamine hydrogels inspired by ascidians and mussels. Chem. Mater. 27, 105–111 (2015).
Li, Q., Barrett, D. G., Messersmith, P. B. & Holten-Andersen, N. Controlling hydrogel mechanics via bio-inspired polymer–nanoparticle bond dynamics. ACS Nano 10, 1317–1324 (2016).
Du, L. et al. A fascinating metallo-supramolecular polymer network with thermal/magnetic/light-responsive shape-memory effects anchored by Fe3O4 nanoparticles. Macromolecules 51, 705–715 (2018).
Xu, J. H., Ye, S. & Fu, J. J. Novel sea cucumber-inspired material based on stiff, strong yet tough elastomer with unique self-healing and recyclable functionalities. J. Mater. Chem. A 6, 24291–24297 (2018).
Xu, Z. et al. Tough and self-recoverable hydrogels crosslinked by triblock copolymer micelles and Fe3+ coordination. J. Polym. Sci. B Polym. Phys. 56, 865–876 (2018).
Filippidi, E. et al. Toughening elastomers using mussel-inspired iron-catechol complexes. Science 358, 502–505 (2017).
Zhang, Y. et al. Mussel-inspired approach to cross-linked functional 3D nanofibrous aerogels for energy-efficient filtration of ultrafine airborne particles. Appl. Surf. Sci. 479, 700–708 (2019).
Bellinger, M. A., Sauer, J. A. & Hara, M. Tensile fracture properties of rigid-rigid blends made of sulfonated polystyrene ionomer and polystyrene. Macromolecules 27, 6147–6155 (1994).
Shi, L. et al. Self-healing silk fibroin-based hydrogel for bone regeneration: dynamic metal-ligand self-assembly approach. Adv. Funct. Mater. 27, 1700591 (2017).
Burnworth, M. et al. Optically healable supramolecular polymers. Nature 472, 334–337 (2011).
Zhang, Z. et al. Eco-friendly, self-healing hydrogels for adhesive and elastic strain sensors, circuit repairing, and flexible electronic devices. Macromolecules 52, 2531–2541 (2019).
Rao, Y. L. et al. Stretchable self-healing polymeric dielectrics cross-linked through metal–ligand coordination. J. Am. Chem. Soc. 138, 6020–6027 (2016).
Zhu, Q., Zhang, L., Van Vliet, K., Miserez, A. & Holten-Andersen, N. White light-emitting multistimuli-responsive hydrogels with lanthanides and carbon dots. ACS Appl. Mater. Interfaces 10, 10409–10418 (2018).
Lee, B. P. & Konst, S. Novel hydrogel actuator inspired by reversible mussel adhesive protein chemistry. Adv. Mater. 26, 3415–3419 (2014).
Lee, B. P., Lin, M. H., Narkar, A., Konst, S. & Wilharm, R. Modulating the movement of hydrogel actuator based on catechol–iron ion coordination chemistry. Sens. Actuators B Chem. 206, 456–462 (2015).
Yang, L., Zhang, G., Zheng, N., Zhao, Q. & Xie, T. A metallosupramolecular shape-memory polymer with gradient thermal plasticity. Angew. Chem. Int. Ed. Engl. 56, 12773–12776 (2017).
Foster, J. A. et al. Differentially addressable cavities within metal–organic cage-cross-linked polymeric hydrogels. J. Am. Chem. Soc. 137, 9722–9729 (2015).
Li, L., Smitthipong, W. & Zeng, H. Mussel-inspired hydrogels for biomedical and environmental applications. Polym. Chem. 6, 353–358 (2015).
Sharma, N., Sharma, P. K., Singh, Y. & Nagaraja, C. M. A self-healing metal–organic gel (MOG) exhibiting pH-responsive release of a chemotherapeutic agent, doxorubicin: modulation of release kinetics by partial dehydration of matrix. ACS Omega 4, 1354–1363 (2019).
Kim, B. J., Cheong, H., Hwang, B. H. & Cha, H. J. Mussel-inspired protein nanoparticles containing iron(III)–DOPA complexes for pH-responsive drug delivery. Angew. Chem. Int. Ed. Engl. 54, 7318–7322 (2015).
Jeong, Y. et al. Sprayable adhesive nanotherapeutics: mussel-protein-based nanoparticles for highly efficient locoregional cancer therapy. ACS Nano 12, 8909–8919 (2018).
Brisson, E. R. L., Griffith, J. C., Bhaskaran, A., Franks, G. V. & Connal, L. A. Temperature-induced self-assembly and metal-ion stabilization of histidine functional block copolymers. J. Polym. Sci. A Polym. Chem. 57, 1964–1973 (2019).
Huang, W. C., Bettinger, C. J., Rhee, K. & Bettinger, C. J. Ultrasound-mediated self-healing hydrogels based on tunable metal–organic bonding. Biomacromolecules 18, 1162–1171 (2017).
Yi, X. et al. Tunable mechanical, antibacterial, and cytocompatible hydrogels based on a functionalized dual network of metal coordination bonds and covalent crosslinking. ACS Appl. Mater. Interfaces 10, 6190–6198 (2018).
Yang, J., Bai, R., Chen, B. & Suo, Z. Hydrogel adhesion: A supramolecular synergy of chemistry, topology, and mechanics. Adv. Funct. Mater. 30, 1901693 (2020).
Li, Y. & Cao, Y. The molecular mechanisms underlying mussel adhesion. Nanoscale Adv. 1, 4246–4257 (2019).
Oh, D. X., Kim, S., Lee, D. & Hwang, D. S. Tunicate-mimetic nanofibrous hydrogel adhesive with improved wet adhesion. Acta Biomater. 20, 104–112 (2015).
Prajatelistia, E. et al. Tunicate-inspired gallic acid/metal ion complex for instant and efficient treatment of dentin hypersensitivity. Adv. Healthc. Mater. 5, 919–927 (2016).
Ju, S. W. et al. Aesthetically improved and efficient tannin–metal chelates for the treatment of dentinal hypersensitivity. RSC Adv. 7, 87–94 (2017).
Ahn, B. K. et al. High-performance mussel-inspired adhesives of reduced complexity. Nat. Commun. 6, 8663 (2015).
Gebbie, M. A. et al. Tuning underwater adhesion with cation–π interactions. Nat. Chem. 9, 473–479 (2017).
Dai, C. et al. A multifunctional metallohydrogel with injectability, self-healing, and multistimulus-responsiveness for bioadhesives. Macromol. Mater. Eng. 303, 1800305 (2018).
Zeng, H., Hwang, D. S., Israelachvili, J. N. & Waite, J. H. Strong reversible Fe3+-mediated bridging between dopa-containing protein films in water. Proc. Natl Acad. Sci. USA 107, 12850–12853 (2010).
Yang, B., Lim, C., Hwang, D. S. & Cha, H. J. Switch of surface adhesion to cohesion by Dopa-Fe3+ complexation, in response to microenvironment at the mussel plaque/substrate interface. Chem. Mater. 28, 7982–7989 (2016).
Choi, Y. C., Choi, J. S., Jung, Y. J. & Cho, Y. W. Human gelatin tissue-adhesive hydrogels prepared by enzyme-mediated biosynthesis of DOPA and Fe3+ ion crosslinking. J. Mater. Chem. B 2, 201–209 (2014).
Kang, H. et al. An in situ reversible heterodimeric nanoswitch controlled by metal-ion–ligand coordination regulates the mechanosensing and differentiation of stem cells. Adv. Mater. 30, 1803591 (2018).
Lai, E., Keshavarz, B. & Holten-Andersen, N. Deciphering how the viscoelastic properties of mussel-inspired metal-coordinate transiently cross-linked gels dictate their tack behavior. Langmuir 35, 15979–15984 (2019).
Schauser, N. S. et al. Decoupling bulk mechanics and mono- and multivalent ion transport in polymers based on metal–ligand coordination. Chem. Mater. 30, 5759–5769 (2018).
Shi, Y. et al. A conductive self-healing hybrid gel enabled by metal–ligand supramolecule and nanostructured conductive polymer. Nano Lett. 15, 6276–6281 (2015).
Gao, H., Sun, Y., Zhou, J., Xu, R. & Duan, H. Mussel-inspired synthesis of polydopamine-functionalized graphene hydrogel as reusable adsorbents for water purification. ACS Appl. Mater. Interfaces 5, 425–432 (2013).
Yan, L. et al. An efficient supramolecular adsorbent for co-adsorption of dyes and metal ions from wastewater and its application in self-healing materials. Soft Matter 13, 8772–8780 (2017).
Lin, Q. et al. Rewritable security display material and Cl− sensor: based on a bimetal competitive coordination controlled supramolecular gel. Mater. Lett. 137, 444–446 (2014).
Lin, Q. et al. A novel strategy for the design of smart supramolecular gels: Controlling stimuli-response properties through competitive coordination of two different metal ions. Chem. Commun. 50, 10669–10671 (2014).
Zhu, Q., Van Vliet, K., Holten-Andersen, N. & Miserez, A. A double-layer mechanochromic hydrogel with multidirectional force sensing and encryption capability. Adv. Funct. Mater. 29, 1808191 (2019).
Ma, Y. et al. Dynamic metal-ligand coordination for multicolour and water-jet rewritable paper. Nat. Commun. 9, 3 (2018).
Bhowmik, S., Ghosh, B. N., Marjomäki, V. & Rissanen, K. Nanomolar pyrophosphate detection in water and in a self-assembled hydrogel of a simple terpyridine-Zn2+ complex. J. Am. Chem. Soc. 136, 5543–5546 (2014).
Ejima, H. et al. One-step assembly of coordination complexes for versatile film and particle engineering. Science 341, 154–157 (2013).
Yun, G., Richardson, J. J., Biviano, M. & Caruso, F. Tuning the mechanical behavior of metal–phenolic networks through building block composition. ACS Appl. Mater. Interfaces 11, 6404–6410 (2019).
Björnmalm, M. et al. Nanoengineering particles through template assembly. Chem. Mater. 29, 289–306 (2017).
Cranford, S. W., Tarakanova, A., Pugno, N. M. & Buehler, M. J. Nonlinear material behaviour of spider silk yields robust webs. Nature 482, 72–76 (2012).
Keten, S., Xu, Z., Ihle, B. & Buehler, M. J. Nanoconfinement controls stiffness, strength and mechanical toughness of β-sheet crystals in silk. Nat. Mater. 9, 359–367 (2010).
Su, I. et al. Imaging and analysis of a three-dimensional spider web architecture. J. R. Soc. Interface 15, 20180193 (2018).
Vidavsky, Y. et al. Tuning the mechanical properties of metallopolymers via ligand interactions: a combined experimental and theoretical study. Macromolecules 53, 2021–2030 (2020).
Marco-Dufort, B., Iten, R. & Tibbitt, M. W. Linking molecular behavior to macroscopic properties in ideal dynamic covalent networks. J. Am. Chem. Soc. 142, 15371–15385 (2020).
Ryu, J., Ku, S. H., Lee, H. & Park, C. B. Mussel-inspired polydopamine coating as a universal route to hydroxyapatite crystallization. Adv. Funct. Mater. 20, 2132–2139 (2010).
Zhou, Y. Z., Cao, Y., Liu, W., Chu, C. H. & Li, Q. L. Polydopamine-induced tooth remineralization. ACS Appl. Mater. Interfaces 4, 6901–6910 (2012).
Regitsky, A. U., Keshavarz, B., McKinley, G. H. & Holten-Andersen, N. Rheology as a mechanoscopic method to monitor mineralization in hydrogels. Biomacromolecules 18, 4067–4074 (2017).
Schmolke, W., Ahmadi, M. & Seiffert, S. Enhancement of metallo-supramolecular dissociation kinetics in telechelic terpyridine-capped poly(ethylene glycol) assemblies in the semi-dilute regime. Phys. Chem. Chem. Phys. 21, 19623–19638 (2019).
Dawson, J. H. Probing structure-function relations in heme-containing oxygenases and peroxidases. Science 240, 433–439 (1988).
Holm, R. H., Kennepohl, P. & Solomon, E. I. Structural and functional aspects of metal sites in biology. Chem. Rev. 96, 2239–2314 (1996).
Lippard, S. J. & Berg, J. M. in Principles of Bioinorganic Chemistry Ch. 12 (eds Lippard, S. J. & Berg, J. M.) (University Science Books, 1994).
Lippard, S. J. & Berg, J. M. in Principles of Bioinorganic Chemistry Ch. 4 (eds Lippard, S. J. & Berg, J. M.) (University Science Books, 1994).
Meyer, T. J. Photochemistry of metal coordination complexes: metal to ligand charge transfer excited states. Pure Appl. Chem. 58, 1193–1206 (1986).
Rosi, N. L. et al. Hydrogen storage in microporous metal-organic frameworks. Science 300, 1127–1129 (2003).
Furukawa, H. et al. Ultrahigh porosity in metal-organic frameworks. Science 329, 424–428 (2010).
Lee, J. et al. Metal–organic framework materials as catalysts. Chem. Soc. Rev. 38, 1450–1459 (2009).
Furukawa, H., Cordova, K. E., O’Keefe, M. & Yaghi, O. M. The chemistry and applications of metal-organic frameworks. Science 341, 1230444 (2013).
Pedersen, C. J. Cyclic polyethers and their complexes with metal salts. J. Am. Chem. Soc. 89, 7017–7036 (1967).
Cram, D. J. The design of molecular hosts, guests, and their complexes (Nobel Lecture). Angew. Chem. Int. Ed. Engl. 27, 1009–1020 (1988).
Lehn, J. & Supramolecular, M. Chemistry — scope and perspectives molecules, supermolecules, and molecular devices (Nobel Lecture). Angew. Chem. Int. Ed. Engl. 27, 89–112 (1988).
Wang, F. et al. Metal coordination mediated reversible conversion between linear and cross-linked supramolecular polymers. Angew. Chem. Int. Ed. Engl. 49, 1090–1094 (2010).
Lehn, J. M. et al. Spontaneous assembly of double-stranded helicates from oligobipyridine ligands and copper(I) cations: structure of an inorganic double helix. Proc. Natl Acad. Sci. USA 84, 2565–2569 (1987).
Saalfrank, R. W., Maid, H. & Scheurer, A. Supramolecular coordination chemistry: the synergistic effect of serendipity and rational design. Angew. Chem. Int. Ed. Engl. 47, 8794–8824 (2008).
Bentz, K. C. & Cohen, S. M. Supramolecular metallopolymers: from linear materials to infinite networks. Angew. Chem. Int. Ed. Engl. 57, 14992–15001 (2018).
Paquin, F., Rivnay, J., Salleo, A., Stingelin, N. & Silva, C. Multi-phase semicrystalline microstructures drive exciton dissociation in neat plastic semiconductors. J. Mater. Chem. C 3, 10715–10722 (2015).
Wiester, M. J., Ulmann, P. A. & Mirkin, C. A. Enzyme mimics based upon supramolecular coordination chemistry. Angew. Chem. Int. Ed. Engl. 50, 114–137 (2011).
Shimazaki, Y., Takani, M. & Yamauchi, O. Metal complexes of amino acids and amino acid side chain groups. Structures and properties. Dalton Trans. https://doi.org/10.1039/b905871k (2009).
Yang, J., Cohen Stuart, M. A. & Kamperman, M. Jack of all trades: Versatile catechol crosslinking mechanisms. Chem. Soc. Rev. 43, 8271–8298 (2014).
D’Ischia, M., Napolitano, A., Ball, V., Chen, C. T. & Buehler, M. J. Polydopamine and eumelanin: From structure–property relationships to a unified tailoring strategy. Acc. Chem. Res. 47, 3541–3550 (2014).
Yount, W. C., Loveless, D. M. & Craig, S. L. Strong means slow: Dynamic contributions to the bulk mechanical properties of supramolecular networks. Angew. Chem. Int. Ed. Engl. 44, 2746–2748 (2005).
Yount, W. C., Loveless, D. M. & Craig, S. L. Small-molecule dynamics and mechanisms underlying the macroscopic mechanical properties of coordinatively cross-linked polymer networks. J. Am. Chem. Soc. 127, 14488–14496 (2005).
Bell, G. I. Models for the specific adhesion of cells to cells. Science 200, 618–627 (1978).
Evans, E. A. & Ritchie, K. Strength of a weak bond connecting flexible polymer chains. Biophys. J. 76, 2439–2447 (1999).
Bertaud, J., Hester, J., Jimenez, D. D. & Buehler, M. J. Energy landscape, structure and rate effects on strength properties of alpha-helical proteins. J. Phys. Condens. Matter 22, 035102 (2010).
Ackbarow, T., Chen, X., Keten, S. & Buehler, M. J. Hierarchies, multiple energy barriers, and robustness govern the fracture mechanics of α-helical and β-sheet protein domains. Proc. Natl Acad. Sci. USA 104, 16410–16415 (2007).
Keten, S. & Buehler, M. J. Strength limit of entropic elasticity in beta-sheet protein domains. Phys. Rev. E 78, 061913 (2008).
Husson, J. & Pincet, F. Analyzing single-bond experiments: Influence of the shape of the energy landscape and universal law between the width, depth, and force spectrum of the bond. Phys. Rev. E 77, 026108 (2008).
Acknowledgements
This work was supported by ONR (N00014-19-1-2375), U.S. Air Force Office of Scientific Research (FA9550-15-1-0514), NIH (U01 EB014976), ARO (W911NF1920098) and NSF Graduate Research Fellowship, as well as MIT CAST through a grant from the Mellon Foundation.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khare, E., Holten-Andersen, N. & Buehler, M.J. Transition-metal coordinate bonds for bioinspired macromolecules with tunable mechanical properties. Nat Rev Mater 6, 421–436 (2021). https://doi.org/10.1038/s41578-020-00270-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41578-020-00270-z
This article is cited by
-
DFT and TDDFT exploration on electronic transitions and bonding aspect of DPA and PTDC ligated transition metal complexes
Journal of Molecular Modeling (2024)
-
Covalently Bonded Ni Sites in Black Phosphorene with Electron Redistribution for Efficient Metal-Lightweighted Water Electrolysis
Nano-Micro Letters (2024)
-
Direct synthesis of amorphous coordination polymers and metal–organic frameworks
Nature Reviews Chemistry (2023)
-
Azomethine-functionalized organic–inorganic framework: an overview
Chemical Papers (2023)
-
Preparation of dual-cross network polymers by the knitting method and evaluation of their mechanical properties
NPG Asia Materials (2022)