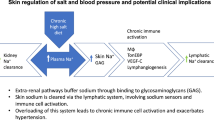
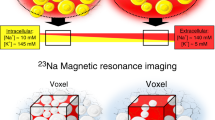
Abstract
During tissue inflammation, immune cells infiltrate the interstitial space of target organs, where they sense and adapt to local environmental stimuli. Such stimuli include not only pathogens but also local factors such as the levels of oxygenation, nutrients and electrolytes. An important electrolyte in this regard is sodium (Na+). Recent in vivo findings have shown a role of Na+ storage in the skin for electrolyte homeostasis. Thereby, Na+ intake may influence the activation status of the immune system through direct effects on T helper cell subsets and innate immune cells in tissues such as the skin and other target organs. Furthermore, high Na+ intake has been shown to alter the composition of the intestinal microbiota, with indirect effects on immune cells. The results suggest regulatory roles for Na+ in cardiovascular disease, inflammation, infection and autoimmunity.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
O’Donnell, M. et al. Urinary sodium and potassium excretion, mortality, and cardiovascular events. N. Engl. J. Med. 371, 612–623 (2014).
Manzel, A. et al. Role of “Western diet” in inflammatory autoimmune diseases. Curr. Allergy Asthma Rep. 14, 404 (2014).
Mente, A. et al. Associations of urinary sodium excretion with cardiovascular events in individuals with and without hypertension: a pooled analysis of data from four studies. Lancet 388, 465–475 (2016).
Mente, A. et al. Association of urinary sodium and potassium excretion with blood pressure. N. Engl. J. Med. 371, 601–611 (2014).
Mozaffarian, D. et al. Global sodium consumption and death from cardiovascular causes. N. Engl. J. Med. 371, 624–634 (2014).
Weber, M. A. et al. Clinical practice guidelines for the management of hypertension in the community a statement by the American Society of Hypertension and the International Society of Hypertension. J. Hypertens. 32, 3–15 (2014).
Taylor, J. 2013 ESH/ESC guidelines for the management of arterial hypertension. Eur. Heart J. 34, 2108–2109 (2013).
World Health Organization. Sodium intake for adults and children. WHO http://www.who.int/nutrition/publications/guidelines/sodium_intake/en/ (2012).
European Commission. Implementation of the EU salt reduction framework results of member states survey. European Commission https://ec.europa.eu/health/sites/health/files/nutrition_physical_activity/docs/salt_report_en.pdf (2012).
Berry, M. R. et al. Renal sodium gradient orchestrates a dynamic antibacterial defense zone. Cell 170, 860–874 (2017). This study shows that Na + recruits macrophages to the renal medulla through epithelial CCL2 production, thus establishing a defence zone against infections.
Jantsch, J. et al. Cutaneous Na+ storage strengthens the antimicrobial barrier function of the skin and boosts macrophage-driven host defense. Cell Metab. 21, 493–501 (2015). This study shows that Na + accumulates at the site of bacterial skin infections in humans and in mice and that a high salt diet promotes skin Na + storage and ameliorates cutaneous leishmaniasis.
Machnik, A. et al. Macrophages regulate salt-dependent volume and blood pressure by a vascular endothelial growth factor-C-dependent buffering mechanism. Nat. Med. 15, 545–552 (2009). This study shows that mononuclear phagocytic cells have a role in regulating Na + homeostasis in the skin in a nonosmotic manner and independently from the kidney.
Wiig, H. et al. Immune cells control skin lymphatic electrolyte homeostasis and blood pressure. J. Clin. Invest. 123, 2803–2815 (2013).
Titze, J. et al. Osmotically inactive skin Na+ storage in rats. Am. J. Physiol. Renal Physiol. 285, F1108–F1117 (2003).
Go, W. Y., Liu, X., Roti, M. A., Liu, F. & Ho, S. N. NFAT5/TonEBP mutant mice define osmotic stress as a critical feature of the lymphoid microenvironment. Proc. Natl Acad. Sci. USA 101, 10673–10678 (2004).
Junger, W. G., Liu, F. C., Loomis, W. H. & Hoyt, D. B. Hypertonic saline enhances cellular immune function. Circ. Shock 42, 190–196 (1994).
Szabo, G. & Magyar, Z. Electrolyte concentrations in subcutaneous tissue fluid and lymph. Lymphology 15, 174–177 (1982).
Knepper, M. A., Kwon, T. H. & Nielsen, S. Molecular physiology of water balance. N. Engl. J. Med. 372, 1349–1358 (2015).
Kitada, K. et al. High salt intake reprioritizes osmolyte and energy metabolism for body fluid conservation. J. Clin. Invest. 127, 1944–1959 (2017).
Ivanova, L. N., Archibasova, V. K. & Shterental, I. Sodium-depositing function of the skin in white rats [Russian]. Fiziol. Zh. SSSR Im. I M Sechenova 64, 358–363 (1978).
Titze, J. et al. Spooky sodium balance. Kidney Int. 85, 759–767 (2014).
Titze, J. et al. Glycosaminoglycan polymerization may enable osmotically inactive Na+ storage in the skin. Am. J. Physiol. Heart Circ. Physiol. 287, H203–H208 (2004).
Karlsen, T. V. et al. High-salt diet causes expansion of the lymphatic network and increased lymph flow in skin and muscle of rats. Arterioscler. Thromb. Vasc. Biol. 38, 2054–2064 (2018).
Nikpey, E. et al. High-salt diet causes osmotic gradients and hyperosmolality in skin without affecting interstitial fluid and lymph. Hypertension 69, 660–668 (2017).
Cannon, W. B. The Wisdom of the Body (W. W. Norton & Company, Inc., 1932).
Rakova, N. et al. Long-term space flight simulation reveals infradian rhythmicity in human Na+ balance. Cell Metab. 17, 125–131 (2013).
Kopp, C. et al. 23Na magnetic resonance imaging of tissue sodium. Hypertension 59, 167–172 (2012).
Kopp, C. et al. 23Na magnetic resonance imaging-determined tissue sodium in healthy subjects and hypertensive patients. Hypertension 61, 635–640 (2013).
Paling, D. et al. Sodium accumulation is associated with disability and a progressive course in multiple sclerosis. Brain 136, 2305–2317 (2013).
Dahlmann, A. et al. Magnetic resonance-determined sodium removal from tissue stores in hemodialysis patients. Kidney Int. 87, 434–441 (2015).
Wang, P. et al. Sex differences in sodium deposition in human muscle and skin. Magn. Reson. Imaging 36, 93–97 (2017).
Karg, M. V. et al. SGLT-2-inhibition with dapagliflozin reduces tissue sodium content: a randomised controlled trial. Cardiovasc. Diabetol. 17, 5 (2018).
Hammon, M. et al. 23Na magnetic resonance imaging of the lower leg of acute heart failure patients during diuretic treatment. PLOS ONE 10, e0141336 (2015).
Shapiro, L. & Dinarello, C. A. Osmotic regulation of cytokine synthesis in vitro. Proc. Natl Acad. Sci. USA 92, 12230–12234 (1995).
Kleinewietfeld, M. et al. Sodium chloride drives autoimmune disease by the induction of pathogenic TH17 cells. Nature 496, 518–522 (2013).
Wu, C. et al. Induction of pathogenic TH17 cells by inducible salt-sensing kinase SGK1. Nature 496, 513–517 (2013). References 35 and 36 are pivotal studies describing a role for salt and SGK1 in T H 17 cell differentiation and in T H 17 cell-mediated autoimmune disease.
Hernandez, A. L. et al. Sodium chloride inhibits the suppressive function of FOXP3+ regulatory T cells. J. Clin. Invest. 125, 4212–4222 (2015).
Binger, K. J. et al. High salt reduces the activation of IL-4- and IL-13-stimulated macrophages. J. Clin. Invest. 125, 4223–4238 (2015).
Wilck, N. et al. Salt-responsive gut commensal modulates TH17 axis and disease. Nature 551, 585–589 (2017). This study shows that intestinal lactobacilli are depleted by dietary salt, thus contributing to the development of T H 17 cell-mediated disease, which can be reverted by Lactobacillus spp. repletion.
Wen, W. et al. Potassium supplementation inhibits IL-17A production induced by salt loading in human T lymphocytes via p38/MAPK-SGK1 pathway. Exp. Mol. Pathol. 100, 370–377 (2016).
Yi, B. et al. Effects of dietary salt levels on monocytic cells and immune responses in healthy human subjects: a longitudinal study. Transl Res. 166, 103–110 (2015).
Hammer, A. et al. Impact of combined sodium chloride and saturated long-chain fatty acid challenge on the differentiation of T helper cells in neuroinflammation. J. Neuroinflamm. 14, 184 (2017).
Heikamp, E. B. et al. The AGC kinase SGK1 regulates TH1 and TH2 differentiation downstream of the mTORC2 complex. Nat. Immunol. 15, 457–464 (2014).
Wu, C. et al. SGK1 governs the reciprocal development of Th17 and regulatory T cells. Cell Rep. 22, 653–665 (2018).
Sumida, T. et al. Activated β-catenin in Foxp3+ regulatory T cells links inflammatory environments to autoimmunity. Nat. Immunol. 19, 1391–1402 (2018).
Yang, X. et al. Exacerbation of lupus nephritis by high sodium chloride related to activation of SGK1 pathway. Int. Immunopharmacol. 29, 568–573 (2015).
Safa, K. et al. Salt accelerates allograft rejection through serum- and glucocorticoid-regulated kinase-1-dependent inhibition of regulatory T cells. J. Am. Soc. Nephrol. 26, 2341–2347 (2015).
Eil, R. et al. Ionic immune suppression within the tumour microenvironment limits T cell effector function. Nature 537, 539–543 (2016). This study reports that increased K + levels constitute an ionic checkpoint within tumours with the potential to modulate T cell function.
Zhang, W. C. et al. High salt primes a specific activation state of macrophages, M(Na). Cell Res. 25, 893–910 (2015).
Hucke, S. et al. Sodium chloride promotes pro-inflammatory macrophage polarization thereby aggravating CNS autoimmunity. J. Autoimmun. 67, 90–101 (2016).
Ip, W. K. & Medzhitov, R. Macrophages monitor tissue osmolarity and induce inflammatory response through NLRP3 and NLRC4 inflammasome activation. Nat. Commun. 6, 6931 (2015). This study identifies the macrophage inflammasome as a sensor of hyperosmotic stress, which is crucial for the induction of inflammatory T H 17 cell-mediated responses.
Ronchi, F. et al. Experimental priming of encephalitogenic Th1/Th17 cells requires pertussis toxin-driven IL-1beta production by myeloid cells. Nat. Commun. 7, 11541 (2016).
Scharschmidt, T. C. & Fischbach, M. A. What lives on our skin: ecology, genomics and therapeutic opportunities of the skin microbiome. Drug Discov. Today Dis. Mech. 10, e83–e89 (2013).
Belkaid, Y. & Segre, J. A. Dialogue between skin microbiota and immunity. Science 346, 954–959 (2014).
Olekhnovitch, R. & Bousso, P. Induction, propagation, and activity of host nitric oxide: lessons from Leishmania infection. Trends Parasitol. 31, 653–664 (2015).
Zhang, W. C. et al. Elevated sodium chloride drives type I interferon signaling in macrophages and increases antiviral resistance. J. Biol. Chem. 293, 1030–1039 (2018).
McNab, F., Mayer-Barber, K., Sher, A., Wack, A. & O’Garra, A. Type I interferons in infectious disease. Nat. Rev. Immunol. 15, 87–103 (2015).
Koepsell, H., Nicholson, W. A., Kriz, W. & Hohling, H. J. Measurements of exponential gradients of sodium and chlorine in the rat kidney medulla using the electron microprobe. Pflugers Archiv 350, 167–184 (1974).
Guzik, T. J. et al. Role of the T cell in the genesis of angiotensin II induced hypertension and vascular dysfunction. J. Exp. Med. 204, 2449–2460 (2007). This is one of the first studies to describe a role for the adaptive immune system in the development of hypertension.
Mattson, D. L. et al. Genetic mutation of recombination activating gene 1 in Dahl salt-sensitive rats attenuates hypertension and renal damage. Am. J. Physiol. Regul. Integr. Comp. Physiol. 304, R407–R414 (2013).
Barbaro, N. R. et al. Dendritic cell amiloride-sensitive channels mediate sodium-induced inflammation and hypertension. Cell Rep. 21, 1009–1020 (2017).
Shah, K. H. et al. Myeloid suppressor cells accumulate and regulate blood pressure in hypertension. Circ. Res. 117, 858–869 (2015).
Itani, H. A. et al. CD70 exacerbates blood pressure elevation and renal damage in response to repeated hypertensive stimuli. Circ. Res. 118, 1233–1243 (2016).
Becattini, S., Taur, Y. & Pamer, E. G. Antibiotic-induced changes in the intestinal microbiota and disease. Trends Mol. Med. 22, 458–478 (2016).
David, L. A. et al. Host lifestyle affects human microbiota on daily timescales. Genome Biol. 15, R89 (2014).
David, L. A. et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 505, 559–563 (2014).
Turnbaugh, P. J. et al. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444, 1027–1031 (2006).
Haghikia, A. et al. Dietary fatty acids directly impact central nervous system autoimmunity via the small intestine. Immunity 43, 817–829 (2015).
Backhed, F., Manchester, J. K., Semenkovich, C. F. & Gordon, J. I. Mechanisms underlying the resistance to diet-induced obesity in germ-free mice. Proc. Natl Acad. Sci. USA 104, 979–984 (2007).
Tremaroli, V. & Backhed, F. Functional interactions between the gut microbiota and host metabolism. Nature 489, 242–249 (2012). This review discusses possible links between the human gut microbiome and the development of obesity, cardiovascular disease and metabolic syndromes.
Koeth, R. A. et al. Intestinal microbiota metabolism of l-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat. Med. 19, 576–585 (2013).
Honda, K. & Littman, D. R. The microbiota in adaptive immune homeostasis and disease. Nature 535, 75–84 (2016).
Ivanov, I. I. et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell 139, 485–498 (2009). This is a pivotal study showing that a non-pathogenic gut commensal can influence T H 17 cell-mediated responses.
Tan, T. G. et al. Identifying species of symbiont bacteria from the human gut that, alone, can induce intestinal Th17 cells in mice. Proc. Natl Acad. Sci. USA 113, E8141–E8150 (2016).
Manfredo Vieira, S. et al. Translocation of a gut pathobiont drives autoimmunity in mice and humans. Science 359, 1156–1161 (2018). This study shows that, on a susceptible genetic background, the systemic translocation of gut pathobionts as a result of intestinal barrier dysfunction can trigger autoimmunity.
Monteleone, I. et al. Sodium chloride-enriched diet enhanced inflammatory cytokine production and exacerbated experimental colitis in mice. J. Crohns Colitis 11, 237–245 (2017).
Wei, Y. et al. High salt diet stimulates gut Th17 response and exacerbates TNBS-induced colitis in mice. Oncotarget 8, 70–823 (2017).
Aguiar, S. L. F. et al. High-salt diet induces IL-17-dependent gut inflammation and exacerbates colitis in mice. Front. Immunol. 8, 1969 (2017).
Miranda, P. M. et al. High salt diet exacerbates colitis in mice by decreasing Lactobacillus levels and butyrate production. Microbiome 6, 57 (2018).
Wang, C. et al. High-salt diet has a certain impact on protein digestion and gut microbiota: a sequencing and proteome combined study. Front. Microbiol. 8, 1838 (2017).
Libbey, J. E. et al. Variations in diet cause alterations in microbiota and metabolites that follow changes in disease severity in a multiple sclerosis model. Benef. Microbes 9, 495–513 (2018).
Stanisavljevic, S. et al. Correlation of gut microbiota composition with resistance to experimental autoimmune encephalomyelitis in rats. Front. Microbiol. 7, 2005 (2016).
Maassen, C. B. et al. Reduced experimental autoimmune encephalomyelitis after intranasal and oral administration of recombinant lactobacilli expressing myelin antigens. Vaccine 21, 4685–4693 (2003).
Toral, M. et al. The probiotic Lactobacillus coryniformis CECT5711 reduces the vascular pro-oxidant and pro-inflammatory status in obese mice. Clin. Sci. 127, 33–45 (2014).
Harrison, D. G., Marvar, P. J. & Titze, J. M. Vascular inflammatory cells in hypertension. Front. Physiol. 3, 128 (2012).
Karbach, S. et al. Interleukin 17 drives vascular inflammation, endothelial dysfunction, and arterial hypertension in psoriasis-like skin disease. Arterioscler. Thromb. Vasc. Biol. 34, 2658–2668 (2014).
Madhur, M. S. et al. Interleukin 17 promotes angiotensin II-induced hypertension and vascular dysfunction. Hypertension 55, 500–507 (2010).
Nguyen, H. et al. Interleukin-17 causes Rho-kinase-mediated endothelial dysfunction and hypertension. Cardiovasc. Res. 97, 696–704 (2013).
von Vietinghoff, S. & Ley, K. Interleukin 17 in vascular inflammation. Cytokine Growth Factor Rev. 21, 463–469 (2010).
Wu, J. et al. Immune activation caused by vascular oxidation promotes fibrosis and hypertension. J. Clin. Invest. 126, 1607 (2015).
Khalesi, S., Sun, J., Buys, N. & Jayasinghe, R. Effect of probiotics on blood pressure: a systematic review and meta-analysis of randomized, controlled trials. Hypertension 64, 897–903 (2014).
Chen, J. et al. Multiple sclerosis patients have a distinct gut microbiota compared to healthy controls. Sci. Rep. 6, 28484 (2016).
Xing, J. et al. Hypoxia induces senescence of bone marrow mesenchymal stem cells via altered gut microbiota. Nat. Commun. 9, 2020 (2018).
Tankou, S. K. et al. Investigation of probiotics in multiple sclerosis. Mult. Scler. 24, 58–63 (2018).
Cox, L. M. et al. Altering the intestinal microbiota during a critical developmental window has lasting metabolic consequences. Cell 158, 705–721 (2014).
Tamburini, S., Shen, N., Wu, H. C. & Clemente, J. C. The microbiome in early life: implications for health outcomes. Nat. Med. 22, 713–722 (2016).
Martinez, I. et al. The gut microbiota of rural papua new guineans: composition, diversity patterns, and ecological processes. Cell Rep. 11, 527–538 (2015).
Zelante, T. et al. Tryptophan catabolites from microbiota engage aryl hydrocarbon receptor and balance mucosal reactivity via interleukin-22. Immunity 39, 372–385 (2013).
Lamas, B. et al. CARD9 impacts colitis by altering gut microbiota metabolism of tryptophan into aryl hydrocarbon receptor ligands. Nat. Med. 22, 598–605 (2016).
Laurans, L. et al. Genetic deficiency of indoleamine 2,3-dioxygenase promotes gut microbiota-mediated metabolic health. Nat. Med. 24, 1113–1120 (2018).
Karczewski, J. et al. Regulation of human epithelial tight junction proteins by Lactobacillus plantarum in vivo and protective effects on the epithelial barrier. Am. J. Physiol. Gastrointest. Liver Physiol. 298, G851–G859 (2010).
Maldonado Galdeano, C., Novotny Nunez, I., Carmuega, E., de Moreno de LeBlanc, A. & Perdigon, G. Role of probiotics and functional foods in health: gut immune stimulation by two probiotic strains and a potential probiotic yoghurt. Endocr. Metab. Immune Disord. Drug Targets 15, 37–45 (2015).
van Baarlen, P., Wells, J. M. & Kleerebezem, M. Regulation of intestinal homeostasis and immunity with probiotic lactobacilli. Trends Immunol. 34, 208–215 (2013).
Faraco, G. et al. Dietary salt promotes neurovascular and cognitive dysfunction through a gut-initiated TH17 response. Nat. Neurosci. 21, 240–249 (2018).
Gomez-Guzman, M. et al. Antihypertensive effects of probiotics Lactobacillus strains in spontaneously hypertensive rats. Mol. Nutr. Food Res. 59, 2326–2336 (2015).
Mu, Q. et al. Control of lupus nephritis by changes of gut microbiota. Microbiome 5, 73 (2017).
Lavasani, S. et al. A novel probiotic mixture exerts a therapeutic effect on experimental autoimmune encephalomyelitis mediated by IL-10 producing regulatory T cells. PLOS ONE 5, e9009 (2010).
Mohle, L. et al. Ly6C(hi) monocytes provide a link between antibiotic-induced changes in gut microbiota and adult hippocampal neurogenesis. Cell Rep. 15, 1945–1956 (2016).
Miyara, M., Ito, Y. & Sakaguchi, S. TREG-cell therapies for autoimmune rheumatic diseases. Nat. Rev. Rheumatol. 10, 543–551 (2014).
Wiig, H. & Swartz, M. A. Interstitial fluid and lymph formation and transport: physiological regulation and roles in inflammation and cancer. Physiol. Rev. 92, 1005–1060 (2012).
Mobasheri, A. Correlation between [Na+], [glycosaminoglycan] and Na+/K+ pump density in the extracellular matrix of bovine articular cartilage. Physiol. Res. 47, 47–52 (1998).
Reed, R. K. & Rubin, K. Transcapillary exchange: role and importance of the interstitial fluid pressure and the extracellular matrix. Cardiovasc. Res. 87, 211–217 (2010).
Card, C. M., Yu, S. S. & Swartz, M. A. Emerging roles of lymphatic endothelium in regulating adaptive immunity. J. Clin. Invest. 124, 943–952 (2014).
Randolph, G. J., Angeli, V. & Swartz, M. A. Dendritic-cell trafficking to lymph nodes through lymphatic vessels. Nat. Rev. Immunol. 5, 617–628 (2005).
Olszewski, W. L. The lymphatic system in body homeostasis: physiological conditions. Lymphat. Res. Biol. 1, 11–21; discussion 21–14 (2003).
Wiig, H. Cornea fluid dynamics. I: measurement of hydrostatic and colloid osmotic pressure in rabbits. Exp. Eye Res. 49, 1015–1030 (1989).
Nedrebo, T., Reed, R. K., Jonsson, R., Berg, A. & Wiig, H. Differential cytokine response in interstitial fluid in skin and serum during experimental inflammation in rats. J. Physiol. 556, 193–202 (2004).
Olszewski, W. L. et al. Lymph draining from foot joints in rheumatoid arthritis provides insight into local cytokine and chemokine production and transport to lymph nodes. Arthritis Rheum. 44, 541–549 (2001).
Jorg, S. et al. High salt drives Th17 responses in experimental autoimmune encephalomyelitis without impacting myeloid dendritic cells. Exp. Neurol. 279, 212–222 (2016).
Tubbs, A. L., Liu, B., Rogers, T. D., Sartor, R. B. & Miao, E. A. Dietary salt exacerbates experimental colitis. J. Immunol. 199, 1051–1059 (2017).
Wu, H. et al. High salt promotes autoimmunity by TET2-induced DNA demethylation and driving the differentiation of Tfh cells. Sci. Rep. 6, 28065 (2016).
Acknowledgements
The authors acknowledge F. C. Luft, H. Wiig and J. Jantsch for their critical and helpful input. M.K. was supported by the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation program (640116), by a SALK-grant from the government of Flanders, Belgium, and by an Odysseus-grant of the Research Foundation Flanders (FWO), Belgium. N.W. and D.N.M. are members of and were supported by the DZHK (German Centre for Cardiovascular Research), Germany. N.W. is a participant in the Clinician Scientist Program funded by the Berlin Institute of Health (BIH), Germany.
Reviewer information
Nature Reviews Immunology thanks S. Z. Duan, D. Hafler and other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Isotonic
-
Two solutions are isotonic when they have the same effective osmole concentration and thus the same osmotic pressure.
- Nonosmotic
-
Not following the rules of osmosis, which is the spontaneous net movement of solvent molecules through a selectively permeable membrane into a region of higher solute concentration.
- MARS500 project
-
A psychosocial isolation experiment conducted between 2007 and 2011 by Russia, the European Space Agency and China in preparation for an unspecified future manned spaceflight to the planet Mars.
- Circaseptan
-
Relating to a 7-day cycle for biological processes of life.
- Infradian
-
A rhythm with a period longer than the period of a circadian rhythm.
- Pressure natriuresis
-
A mechanism for the long-term control of arterial pressure, whereby an increase in renal perfusion pressure leads to increased Na+ excretion.
- Sodium magnetic resonance imaging
-
(Na-MRI). A technique using a magnetic field with a special coil to allow for visualization of 23Na in living tissue.
- Primary aldosteronism
-
A disease that involves the excess production of aldosterone by the adrenal glands, resulting in high blood pressure.
- M1 and M2 macrophages
-
M1 and M2 are classifications historically used to define macrophages activated in vitro as pro-inflammatory (when classically activated with IFNγ and lipopolysaccharide) or anti-inflammatory (when alternatively activated with IL-4 or IL-10), respectively. However, in vivo, macrophages are highly specialized, transcriptomically dynamic and extremely heterogeneous with regard to their phenotypes and functions, which are continuously shaped by their tissue microenvironment. Therefore, the M1 or M2 classification is too simplistic to explain the true nature of in vivo macrophages, although these terms are still often used to indicate whether the macrophages in question are more pro-inflammatory or anti-inflammatory.
- Central osmoreceptors
-
Sensory receptors found primarily in the hypothalamus of most homeothermic organisms that detect changes in osmotic pressure.
- Vasopressin
-
A peptide hormone released from the posterior pituitary in response to extracellular fluid hypertonicity, which leads to re-absorption of solute-free water in the kidney tubules as well as vasoconstriction.
- Atrial natriuretic peptide
-
A peptide hormone secreted by the atria in the heart, which reduces an expanded extracellular fluid volume by increasing renal Na+ excretion.
- Renin–angiotensin–aldosterone system
-
A hormone system regulating blood pressure and fluid balance.
- Albuminuria
-
A pathological condition in which the plasma protein albumin is abnormally present in the urine.
- Diabetes insipidus
-
A pathological condition characterized by the production of large amounts of dilute urine and increased thirst.
- Segmented filamentous bacteria
-
(SFB). Members of the gut microbiota of rodents, fish and chickens, with morphological resemblance to bacterial filaments that were previously observed in the guts of insects.
- Operational taxonomic units
-
Clusters of (microbial) organisms grouped by DNA sequence similarity of a specific taxonomic marker gene.
Rights and permissions
About this article
Cite this article
Müller, D.N., Wilck, N., Haase, S. et al. Sodium in the microenvironment regulates immune responses and tissue homeostasis. Nat Rev Immunol 19, 243–254 (2019). https://doi.org/10.1038/s41577-018-0113-4
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41577-018-0113-4