Abstract
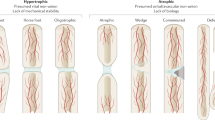
Bone stress injuries, including stress fractures, are overuse injuries that lead to substantial morbidity in active individuals. These injuries occur when excessive repetitive loads are introduced to a generally normal skeleton. Although the precise mechanisms for bone stress injuries are not completely understood, the prevailing theory is that an imbalance in bone metabolism favours microdamage accumulation over its removal and replacement with new bone via targeted remodelling. Diagnosis is achieved by a combination of patient history and physical examination, with imaging used for confirmation. Management of bone stress injuries is guided by their location and consequent risk of healing complications. Bone stress injuries at low-risk sites typically heal with activity modification followed by progressive loading and return to activity. Additional treatment approaches include non-weight-bearing immobilization, medications or surgery, but these approaches are usually limited to managing bone stress injuries that occur at high-risk sites. A comprehensive strategy that integrates anatomical, biomechanical and biological risk factors has the potential to improve the understanding of these injuries and aid in their prevention and management.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 1 digital issues and online access to articles
$99.00 per year
only $99.00 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Breithaupt, J. Zur Pathologie des menschlichen Fußes. Med. Ztg. 24, 169–177 (1855).
Stechow, S. Fußödem und Röntgenstrahlen. Dtsch. mil. ärztl. Z. 26, 465 (1897).
Burr, D. B. et al. Bone microdamage and skeletal fragility in osteoporotic and stress fractures. J. Bone Miner. Res. 12, 6–15 (1997).
Lee, D. Stress fractures, active component, U.S. Armed Forces, 2004–2010. MSMR 18, 8–11 (2011).
Waterman, B. R., Gun, B., Bader, J. O., Orr, J. D. & Belmont, P. J. Jr Epidemiology of lower extremity stress fractures in the United States military. Mil. Med. 181, 1308–1313 (2016).
Bulathsinhala, L. et al. Risk of stress fracture varies by race/ethnic origin in a cohort study of 1.3 million US Army soldiers. J. Bone Miner. Res. 32, 1546–1553 (2017).
Kardouni, J. R., McKinnon, C. J., Taylor, K. M. & Hughes, J. M. Timing of stress fracture in soldiers during the first 6 career months: a retrospective cohort study. J. Athl. Train. 56, 1278–1284 (2021).
Wentz, L., Liu, P. Y., Haymes, E. & Ilich, J. Z. Females have a greater incidence of stress fractures than males in both military and athletic populations: a systemic review. Mil. Med. 176, 420–430 (2011).
Shaffer, R. A., Rauh, M. J., Brodine, S. K., Trone, D. W. & Macera, C. A. Predictors of stress fracture susceptibility in young female recruits. Am. J. Sports Med. 34, 108–115 (2006).
Tenforde, A. S., Sayres, L. C., McCurdy, M. L., Sainani, K. L. & Fredericson, M. Identifying sex-specific risk factors for stress fractures in adolescent runners. Med. Sci. Sports Exerc. 45, 1843–1851 (2013).
Changstrom, B. G., Brou, L., Khodaee, M., Braund, C. & Comstock, R. D. Epidemiology of stress fracture injuries among US high school athletes, 2005-2006 through 2012-2013. Am. J. Sports Med. 43, 26–33 (2015).
Rizzone, K. H., Ackerman, K. E., Roos, K. G., Dompier, T. P. & Kerr, Z. Y. The epidemiology of stress fractures in collegiate student-athletes, 2004-2005 through 2013-2014 academic years. J. Athl. Train. 52, 966–975 (2017).
Patel, N. M. et al. Is the incidence of paediatric stress fractures on the rise? Trends in New York State from 2000 to 2015. J. Pediatr. Orthop. B 29, 499–504 (2020).
Kliethermes, S. A. et al. Defining a research agenda for youth sport specialisation in the USA: the AMSSM youth early sport specialization summit. Br. J. Sports Med. 55, 135–143 (2021).
Warden, S. J., Edwards, W. B. & Willy, R. W. Preventing bone stress injuries in runners with optimal workload. Curr. Osteoporos. Rep. 19, 298–307 (2021).
Kountouris, A. et al. MRI bone marrow oedema precedes lumbar bone stress injury diagnosis in junior elite cricket fast bowlers. Br. J. Sports Med. 53, 1236–1239 (2019).
Warden, S. J., Gutschlag, F. R., Wajswelner, H. & Crossley, K. M. Aetiology of rib stress fractures in rowers. Sports Med. 32, 819–836 (2002).
Branch, T., Partin, C., Chamberland, P., Emeterio, E. & Sabetelle, M. Spontaneous fractures of the humerus during pitching. A series of 12 cases. Am. J. Sports Med. 20, 468–470 (1992).
Warden, S. J. et al. Physical activity when young provides lifelong benefits to cortical bone size and strength in men. Proc. Natl Acad. Sci. USA 111, 5337–5342 (2014).
Caine, D., Meyers, R., Nguyen, J., Schöffl, V. & Maffulli, N. Primary periphyseal stress injuries in young athletes: a systematic review. Sports Med. 52, 741–772 (2021).
Tenforde, A. S. et al. Prevalence and anatomical distribution of bone stress injuries in the elite para athlete. Am. J. Phys. Med. Rehabil. 98, 1036–1040 (2019).
Weigl, K. & Amrami, B. Occupational stress fracture in an unusual location: report of a case in the distal end of the shaft of the radius. Clin. Orthop. Relat. Res. 147, 222–224 (1980).
Dellestable, F. & Gaucher, A. Clay-shoveler’s fracture. Stress fracture of the lower cervical and upper thoracic spinous processes. Rev. Rhum. Engl. Ed. 65, 575–582 (1998).
Peebles, C. R., Sulkin, T. & Sampson, M. A. ‘Cable-maker’s clavicle’: stress fracture of the medial clavicle. Skelet. Radiol. 29, 421–423 (2000).
Howard, R. S. & Conrad, G. R. Ice cream scooper’s hand. Report of an occupationally related stress fracture of the hand. Clin. Nucl. Med. 17, 721–723 (1992).
Wu, Y. F., Lu, K., Girgis, C., Preda, M. & Preda, V. Postpartum bilateral sacral stress fracture without osteoporosis—a case report and literature review. Osteoporos. Int. 32, 623–631 (2021).
Scott, R. D., Turoff, N. & Ewald, F. C. Stress fracture of the patella following duopatellar total knee arthroplasty with patellar resurfacing. Clin. Orthop. Relat. Res. 170, 147–151 (1982).
Venkatanarasimha, N., Kamath, S., Kambouroglou, G. & Ostlere, S. Proximal ulna stress fracture and stress reaction of the proximal radius associated with the use of crutches: a case report and literature review. J. Orthop. Traumatol. 10, 155–157 (2009).
Oren, V., Kozenitzky, I., Babiacki, A. & Stern, A. Unusual cough related stress injuries. Eur. J. Nucl. Med. 14, 108–111 (1988).
Hollander, K. et al. Sex-specific differences in running injuries: a systematic review with meta-analysis and meta-regression. Sports Med. 51, 1011–1039 (2021).
Wright, A. A., Taylor, J. B., Ford, K. R., Siska, L. & Smoliga, J. M. Risk factors associated with lower extremity stress fractures in runners: a systematic review with meta-analysis. Br. J. Sports Med. 49, 1517–1523 (2015).
Rauh, M. J., Barrack, M. & Nichols, J. F. Associations between the female athlete triad and injury among high school runners. Int. J. Sports Phys. Ther. 9, 948–958 (2014).
Tenforde, A. S. et al. Association of the female athlete triad risk assessment stratification to the development of bone stress injuries in collegiate athletes. Am. J. Sports Med. 45, 302–310 (2017).
Kraus, E. et al. Bone stress injuries in male distance runners: higher modified Female Athlete Triad Cumulative Risk Assessment scores predict increased rates of injury. Br. J. Sports Med. 53, 237–242 (2019).
Nieves, J. W. et al. Males have larger skeletal size and bone mass than females, despite comparable body size. J. Bone Miner. Res. 20, 529–535 (2005).
Tommasini, S. M., Nasser, P. & Jepsen, K. J. Sexual dimorphism affects tibia size and shape but not tissue-level mechanical properties. Bone 40, 498–505 (2007).
Hill, P. F., Chatterji, S., Chambers, D. & Keeling, J. D. Stress fracture of the pubic ramus in female recruits. J. Bone Jt. Surg. Br. 78, 383–386 (1996).
Kelly, E. W., Jonson, S. R., Cohen, M. E. & Shaffer, R. Stress fractures of the pelvis in female Navy recruits: an analysis of possible mechanisms of injury. Mil. Med. 165, 142–146 (2000).
Wang, H., Kia, M. & Dickin, D. C. Influences of load carriage and physical activity history on tibia bone strain. J. Sport. Health Sci. 8, 478–485 (2019).
Xu, C., Silder, A., Zhang, J., Reifman, J. & Unnikrishnan, G. A cross-sectional study of the effects of load carriage on running characteristics and tibial mechanical stress: implications for stress-fracture injuries in women. BMC Musculoskelet. Disord. 18, 1–12 (2017).
Knapik, J. et al. Stress fracture risk factors in basic combat training. Int. J. Sports Med. 33, 940–946 (2012).
Popp, K. L. et al. Bone mass, microarchitecture and strength are influenced by race/ethnicity in young adult men and women. Bone 103, 200–208 (2017).
Warden, S. J. et al. Racial differences in cortical bone and their relationship to biochemical variables in Black and White children in the early stages of puberty. Osteoporos. Int. 24, 1869–1879 (2013).
Seref-Ferlengez, Z., Kennedy, O. D. & Schaffler, M. B. Bone microdamage, remodeling and bone fragility: how much damage is too much damage? Bonekey Rep. 4, 644–644 (2015).
Giraud-Guille, M.-M. Twisted plywood architecture of collagen fibrils in human compact bone osteons. Calcif. Tissue Int. 42, 167–180 (1988).
Skedros, J. G., Mason, M. W., Nelson, M. C. & Bloebaum, R. D. Evidence of structural and material adaptation to specific strain features in cortical bone. Anat. Rec. 246, 47–63 (1996).
Wolff, J. Das Gesetz der Transformation der Knochen (Hirschwald, 1892).
Burr, D. B. Bone quality: understanding what matters. J. Musculoskelet. Neuronal Interact. 4, 184–186 (2004).
Pattin, C. A., Caler, W. E. & Carter, D. R. Cyclic mechanical property degradation during fatigue loading of cortical bone. J. Biomech. 29, 69–79 (1996).
Akkus, O., Knott, D. F., Jepsen, K. J., Davy, D. T. & Rimnac, C. M. Relationship between damage accumulation and mechanical property degradation in cortical bone: microcrack orientation is important. J. Biomed. Mater. Res. A 65, 482–488 (2003).
Burr, D. B. et al. Does microdamage accumulation affect the mechanical properties of bone? J. Biomech. 31, 337–345 (1998).
Lee, T. C. et al. Detecting microdamage in bone. J. Anat. 203, 161–172 (2003).
Burr, D. B. Why bones bend but don’t break. J. Musculoskelet. Neuronal Interact. 11, 270–285 (2011). This paper gives a broader view of why bones fail and what prevents failure. It also partly focuses on the role of muscle in reducing stresses in bone, and the role that gait has.
Wasserman, N., Yerramshetty, J. & Akkus, O. Microcracks colocalize within highly mineralized regions of cortical bone tissue. Eur. J. Morphol. 42, 43–51 (2005).
Schaffler, M. B., Choi, K. & Milgrom, C. Aging and matrix microdamage accumulation in human compact bone. Bone 17, 521–525 (1995).
Norman, T. L. & Wang, Z. Microdamage of human cortical bone: incidence and morphology in long bones. Bone 20, 375–379 (1997).
Seref-Ferlengez, Z., Basta-Pljakic, J., Kennedy, O. D., Philemon, C. J. & Schaffler, M. B. Structural and mechanical repair of diffuse damage in cortical bone in vivo. J. Bone Miner. Res. 29, 2537–2544 (2014).
Fantner, G. E. et al. Sacrificial bonds and hidden length dissipate energy as mineralized fibrils separate during bone fracture. Nat. Mater. 4, 612–616 (2005). This report gives insight into bone pathophysiology and guides better understanding of bone failure in response to repetitive loading.
Poundarik, A. A. et al. Dilatational band formation in bone. Proc. Natl Acad. Sci. USA 109, 19178–19183 (2012).
Burr, D. B., Schaffler, M. B. & Frederickson, R. G. Composition of the cement line and its possible mechanical role as a local interface in human compact bone. J. Biomech. 21, 939–945 (1988).
Bass, S. L. et al. The effect of mechanical loading on the size and shape of bone in pre-, peri-, and postpubertal girls: a study in tennis players. J. Bone Miner. Res. 17, 2274–2280 (2002).
Bradney, M. et al. Moderate exercise during growth in prepubertal boys: changes in bone mass, size, volumetric density, and bone strength: a controlled prospective study. J. bone Miner. Res. 13, 1814–1821 (1998).
Robling, A. G., Hinant, F. M., Burr, D. B. & Turner, C. H. Improved bone structure and strength after long-term mechanical loading is greatest if loading is separated into short bouts. J. Bone Miner. Res. 17, 1545–1554 (2002).
Warden, S. J. et al. Bone adaptation to a mechanical loading program significantly increases skeletal fatigue resistance. J. Bone Miner. Res. 20, 809–816 (2005).
Warden, S. J., Fuchs, R. K., Castillo, A. B., Nelson, I. R. & Turner, C. H. Exercise when young provides lifelong benefits to bone structure and strength. J. Bone Miner. Res. 22, 251–259 (2007).
Zaidi, M. Skeletal remodeling in health and disease. Nat. Med. 13, 791–801 (2007).
Slemenda, C. W., Peacock, M., Hui, S., Zhou, L. & Johnston, C. C. Reduced rates of skeletal remodeling are associated with increased bone mineral density during the development of peak skeletal mass. J. Bone Miner. Res. 12, 676–682 (1997).
Jandl, N. M. et al. Large osteocyte lacunae in iliac crest infantile bone are not associated with impaired mineral distribution or signs of osteocytic osteolysis. Bone 135, 115324 (2020).
Langdahl, B., Ferrari, S. & Dempster, D. W. Bone modeling and remodeling: potential as therapeutic targets for the treatment of osteoporosis. Ther. Adv. Musculoskelet. Dis. 8, 225–235 (2016).
Raggatt, L. J. & Partridge, N. C. Cellular and molecular mechanisms of bone remodeling. J. Biol. Chem. 285, 25103–25108 (2010).
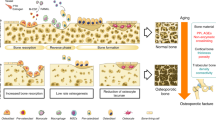
Parfitt, A. M. Targeted and nontargeted bone remodeling: relationship to basic multicellular unit origination and progression. Bone 30, 5–7 (2002). This paper is one of the first to define targeted remodelling as an entity and lead to studies to determine a mechanism for identification and repair of microdamage that could lead to a stress fracture.
Burr, D. B., Martin, R. B., Schaffler, M. B. & Radin, E. L. Bone remodeling in response to in vivo fatigue microdamage. J. Biomech. 18, 189–200 (1985).
Burr, D. B. & Martin, R. B. Calculating the probability that microcracks initiate resorption spaces. J. Biomech. 26, 613–616 (1993).
Verborgt, O., Gibson, G. J. & Schaffler, M. B. Loss of osteocyte integrity in association with microdamage and bone remodeling after fatigue in vivo. J. Bone Miner. Res. 15, 60–67 (2000).
Bonewald, L. F. The amazing osteocyte. J. Bone Miner. Res. 26, 229–238 (2011).
Nakashima, T. et al. Evidence for osteocyte regulation of bone homeostasis through RANKL expression. Nat. Med. 17, 1231–1234 (2011).
Kramer, I. et al. Osteocyte Wnt/beta-catenin signaling is required for normal bone homeostasis. Mol. Cell. Biol. 30, 3071–3085 (2010).
Lacey, D. L. et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 93, 165–176 (1998).
Simonet, W. et al. Osteoprotegerin: a novel secreted protein involved in the regulation of bone density. Cell 89, 309–319 (1997).
Yasuda, H. et al. Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc. Natl Acad. Sci. USA 95, 3597–3602 (1998).
Kennedy, O. D. et al. Activation of resorption in fatigue-loaded bone involves both apoptosis and active pro-osteoclastogenic signaling by distinct osteocyte populations. Bone 50, 1115–1122 (2012).
McCutcheon, S., Majeska, R. J., Spray, D. C., Schaffler, M. B. & Vazquez, M. Apoptotic osteocytes induce RANKL production in bystanders via purinergic signaling and activation of pannexin channels. J. Bone Miner. Res. 35, 966–977 (2020).
Bennell, K. L., Malcolm, S. A., Wark, J. D. & Brukner, P. D. Models for the pathogenesis of stress fractures in athletes. Br. J. Sports Med. 30, 200–204 (1996).
O’Brien, F. J., Taylor, D. & Clive Lee, T. The effect of bone microstructure on the initiation and growth of microcracks. J. Orthop. Res. Soc. 23, 475–480 (2005).
Schaffler, M. B., Radin, E. L. & Burr, D. B. Long-term fatigue behavior of compact bone at low strain magnitude and rate. Bone 11, 321–326 (1990).
Fyhrie, D. P. et al. Effect of fatiguing exercise on longitudinal bone strain as related to stress fracture in humans. Ann. Biomed. Eng. 26, 660–665 (1998). This paper demonstrates — in a real-life situation in which strains were measured in human bone — that muscle fatigue is associated with increased strains and altered strain distribution in the tibia. It shows the importance of muscle in regulating bone strain.
Warden, S. J., Edwards, W. B. & Willy, R. W. Optimal load for managing low-risk tibial and metatarsal bone stress injuries in runners: the science behind the clinical reasoning. J. Orthop. Sports Phys. Ther. 51, 322–330 (2021).
Kidd, L. J. et al. Temporal pattern of gene expression and histology of stress fracture healing. Bone 46, 369–378 (2010). This is one of the first papers to examine the histology of stress fractures and their healing.
Jamal, S. A., Dion, N. & Ste-Marie, L.-G. Atypical femoral fractures and bone turnover. N. Engl. J. Med. 365, 1261–1262 (2011).
Winters, M. et al. Microcrack-associated bone remodeling is rarely observed in biopsies from athletes with medial tibial stress syndrome. J. Bone Miner. Metab. 37, 496–502 (2019).
Schilcher, J., Bernhardsson, M. & Aspenberg, P. Chronic anterior tibial stress fractures in athletes: no crack but intense remodeling. Scand. J. Med. Sci. Sports 29, 1521–1528 (2019).
Avin, K. G., Bloomfield, S. A., Gross, T. S. & Warden, S. J. Biomechanical aspects of the muscle-bone interaction. Curr. Osteoporos. Rep. 13, 1–8 (2015).
Tenforde, A. S., Hayano, T., Jamison, S. T., Outerleys, J., & Davis, I. S. Tibial Acceleration Measured from Wearable Sensors Is Associated with Loading Rates in Injured Runners. P MR 12 678–684 (2020).
Bennell, K. L. et al. Risk factors for stress fractures in track and field athletes: a twelve-month prospective study. Am. J. Sports Med. 24, 810–818 (1996).
Cosman, F. et al. Determinants of stress fracture risk in United States Military Academy cadets. Bone 55, 359–366 (2013).
Beck, T. et al. Stress fracture in military recruits: gender differences in muscle and bone susceptibility factors. Bone 27, 437–444 (2000).
Beck, T. J. et al. Dual-energy X-ray absorptiometry derived structural geometry for stress fracture prediction in male US Marine Corps recruits. J. Bone Miner. Res. 11, 645–653 (1996).
Giladi, M. et al. Stress fractures and tibial bone width. A risk factor. J. Bone Jt. Surg. Br. 69, 326–329 (1987).
Field, A. E., Gordon, C. M., Pierce, L. M., Ramappa, A. & Kocher, M. S. Prospective study of physical activity and risk of developing a stress fracture among preadolescent and adolescent girls. Arch. Pediatr. Adolesc. Med. 165, 723–728 (2011).
Khan, M. et al. Epidemiology and impact on performance of lower extremity stress injuries in professional basketball players. Sports Health 10, 169–174 (2018).
Schaffler, M. B. in Musculoskeletal Fatigue and Stress Fractures (eds Burr, D. B. & Milgrom, C.) (CRC Press, 2001).
Dixon, S. J., Creaby, M. W. & Allsopp, A. J. Comparison of static and dynamic biomechanical measures in military recruits with and without a history of third metatarsal stress fracture. Clin. Biomech. 21, 412–419 (2006).
Milner, C. E., Hamill, J. & Davis, I. S. Distinct hip and rearfoot kinematics in female runners with a history of tibial stress fracture. J. Orthop. Sports Phys. Ther. 40, 59–66 (2010).
Pohl, M. B., Mullineaux, D. R., Milner, C. E., Hamill, J. & Davis, I. S. Biomechanical predictors of retrospective tibial stress fractures in runners. J. Biomech. 41, 1160–1165 (2008).
Milner, C. E., Ferber, R., Pollard, C. D., Hamill, J. & Davis, I. S. Biomechanical factors associated with tibial stress fracture in female runners. Med. Sci. Sports Exerc. 38, 323–328 (2006).
Finestone, A. et al. Risk factors for stress fractures among Israeli infantry recruits. Mil. Med. 156, 528–530 (1991).
Sullivan, D., Warren, R. F., Pavlov, H. & Kelman, G. Stress fractures in 51 runners. Clin. Orthop. Relat. Res. 187, 188–192 (1984).
Simkin, A., Leichter, I., Giladi, M., Stein, M. & Milgrom, C. Combined effect of foot arch structure and an orthotic device on stress fractures. Foot Ankle 10, 25–29 (1989).
Troy, K. L., Davis, I. S. & Tenforde, A. S. A narrative review of metatarsal bone stress injury in athletic populations: etiology, biomechanics, and management. PM R. 13, 1281–1290 (2021).
Kliethermes, S. A. et al. Lower step rate is associated with a higher risk of bone stress injury: a prospective study of collegiate cross country runners. Br. J. Sports Med. 55, 851–856 (2021).
Matijevich, E. S., Scott, L. R., Volgyesi, P., Derry, K. H. & Zelik, K. E. Combining wearable sensor signals, machine learning and biomechanics to estimate tibial bone force and damage during running. Hum. Mov. Sci. 74, 102690 (2020).
Scott, S. H. & Winter, D. A. Internal forces of chronic running injury sites. Med. Sci. Sports Exerc. 22, 357–369 (1990).
Armstrong, D. W. III, Rue, J.-P. H., Wilckens, J. H. & Frassica, F. J. Stress fracture injury in young military men and women. Bone 35, 806–816 (2004).
Hoffman, J. R., Chapnik, L., Shamis, A., Givon, U. & Davidson, B. The effect of leg strength on the incidence of lower extremity overuse injuries during military training. Mil. Med. 164, 153–156 (1999).
Yoshikawa, T. et al. The effects of muscle fatigue on bone strain. J. Exp. Biol. 188, 217–233 (1994).
Ferris, D. P., Louie, M. & Farley, C. T. Running in the real world: adjusting leg stiffness for different surfaces. Proc. R. Soc. Lon. Ser. B: Biol. Sci. 265, 989–994 (1998).
Milgrom, C., Finestone, A. S. & Voloshin, A. Differences in the principal strain angles during activities performed on natural hilly terrain versus engineered surfaces. Clin. Biomech. 80, 105146 (2020).
Davis, I. S. et al. Stepping back to minimal footwear: applications across the lifespan. Exerc. Sport Sci. Rev. 49, 228–243 (2021).
Giuliani, J., Masini, B., Alitz, C. & Owens, B. D. Barefoot-simulating footwear associated with metatarsal stress injury in 2 runners. Orthopedics 34, e320–e323 (2011).
Lieberman, D. E. et al. Foot strike patterns and collision forces in habitually barefoot versus shod runners. Nature 463, 531–535 (2010).
Finestone, A. & Milgrom, C. How stress fracture incidence was lowered in the Israeli army: a 25-yr struggle. Med. Sci. Sports Exerc. 40, S623–S629 (2008).
Tenforde, A. S., Sainani, K. L., Carter Sayres, L., Milgrom, C. & Fredericson, M. Participation in ball sports may represent a prehabilitation strategy to prevent future stress fractures and promote bone health in young athletes. PM R. 7, 222–225 (2015).
Tenforde, A. S., Parziale, A. L., Popp, K. L. & Ackerman, K. E. Low bone mineral density in male athletes is associated with bone stress injuries at anatomic sites with greater trabecular composition. Am. J. Sports Med. 46, 30–36 (2018).
Barrack, M. T. et al. Higher incidence of bone stress injuries with increasing female athlete triad-related risk factors: a prospective multisite study of exercising girls and women. Am. J. Sports Med. 42, 949–958 (2014).
Mountjoy, M. et al. The IOC consensus statement: beyond the female athlete triad — relative energy deficiency in sport (RED-S). Br. J. Sports Med. 48, 491–497 (2014).
Mountjoy, M. et al. IOC consensus statement on relative energy deficiency in sport (RED-S): 2018 update. Br. J. Sports Med. 52, 687–697 (2018). This International Olympic Committee consensus statement outlines terminology on low energy availability in athletes.
Elliott-Sale, K. J., Tenforde, A. S., Parziale, A. L., Holtzman, B. & Ackerman, K. E. Endocrine effects of relative energy deficiency in sport. Int. J. Sport Nutr. Exerc. Metab. 28, 335–349 (2018).
Papageorgiou, M. et al. Effects of reduced energy availability on bone metabolism in women and men. Bone 105, 191–199 (2017).
Hutson, M. J., O’Donnell, E., Brooke-Wavell, K., Sale, C. & Blagrove, R. C. Effects of low energy availability on bone health in endurance athletes and high-impact exercise as a potential countermeasure: a narrative review. Sports Med. 51, 391–403 (2021).
De Souza, M. J. et al. 2014 Female Athlete Triad Coalition Consensus Statement on Treatment and Return to Play of the Female Athlete Triad: 1st International Conference held in San Francisco, California, May 2012 and 2nd International Conference held in Indianapolis, Indiana, May 2013. Br. J. Sports Med. 48, 289 (2014).
Nattiv, A. et al. The Male Athlete Triad — a Consensus Statement From The Female and Male Athlete Triad Coalition Part 1: definition and scientific basis. Clin. J. Sport. Med. 31, 335–348 (2021).
Tenforde, A. S. et al. Sport and triad risk factors influence bone mineral density in collegiate athletes. Med. Sci. Sports Exerc. 50, 2536–2543 (2018).
Ackerman, K. E. et al. Bone microarchitecture is impaired in adolescent amenorrheic athletes compared with eumenorrheic athletes and nonathletic controls. J. Clin. Endocrinol. Metab. 96, 3123–3133 (2011).
Lappe, J. et al. Calcium and vitamin D supplementation decreases incidence of stress fractures in female Navy recruits. J. Bone Miner. Res. 23, 741–749 (2008).
Sonneville, K. R. et al. Vitamin D, calcium, and dairy intakes and stress fractures among female adolescents. Arch. Pediatr. Adolesc. Med. 166, 595–600 (2012).
Nieves, J. W. et al. Nutritional factors that influence change in bone density and stress fracture risk among young female cross-country runners. PM R. 2, 740–750 (2010). quiz 794.
Burgi, A. A. et al. High serum 25-hydroxyvitamin D is associated with a low incidence of stress fractures. J. Bone Miner. Res. 26, 2371–2377 (2011).
Putman, M. S. et al. Differences in skeletal microarchitecture and strength in African-American and white women. J. Bone Miner. Res. 28, 2177–2185 (2013).
Popp, K. L. et al. Trabecular microstructure is influenced by race and sex in Black and White young adults. Osteoporos. Int. 30, 201–209 (2019).
Varley, I. et al. Functional polymorphisms in the P2X7 receptor gene are associated with stress fracture injury. Purinergic Signal. 12, 103–113 (2016).
Lappe, J. M., Stegman, M. R. & Recker, R. R. The impact of lifestyle factors on stress fractures in female Army recruits. Osteoporos. Int. 12, 35–42 (2001).
Poonuru, S., Findling, J. W. & Shaker, J. L. Lower extremity insufficiency fractures: an underappreciated manifestation of endogenous Cushing’s syndrome. Osteoporos. Int. 27, 3645–3649 (2016).
Hughes, J. M. et al. Nonsteroidal anti-inflammatory drug prescriptions are associated with increased stress fracture diagnosis in the US Army population. J. Bone Miner. Res. 34, 429–436 (2019).
Apte, S. et al. Biomechanical response of the lower extremity to running-induced acute fatigue: a systematic review. Front. Physiol. 12, 646042 (2021).
Matheson, G. et al. Stress fractures in athletes: a study of 320 cases. Am. J. Sports Med. 15, 46–58 (1987).
Johnson, A. W., Weiss, C. B. Jr & Wheeler, D. L. Stress fractures of the femoral shaft in athletes — more common than expected: a new clinical test. Am. J. Sports Med. 22, 248–256 (1994).
Madden, C. & Mellion, M. Sever’s disease and other causes of heel pain in adolescents. Am. Fam. Phys. 54, 1995–2000 (1996).
Milgrom, C. et al. Medial tibial stress fracture diagnosis and treatment guidelines. J. Sci. Med. sport. 24, 526–530 (2021).
Schneiders, A. G. et al. The ability of clinical tests to diagnose stress fractures: a systematic review and meta-analysis. J. Orthopa. Sports Phys. Ther. 42, 760–771 (2012).
Shapiro, M., Zubkov, K. & Landau, R. Diagnosis of stress fractures in military trainees: a large-scale cohort. BMJ Mil. Health https://doi.org/10.1136/bmjmilitary-2020-001406 (2020).
Papalada, A. et al. Ultrasound as a primary evaluation tool of bone stress injuries in elite track and field athletes. Am. J. Sports Med. 40, 915–919 (2012).
Hoenig, T., Tenforde, A., Strahl, A., Rolvien, T. & Hollander, K. Does MRI grading correlate with return to sports following bone stress injuries? A systematic review and meta-analysis. Am. J. Sports Med. 50, 834–844 (2022).
Rolvien, T. et al. Clinical and radiological characterization of patients with immobilizing and progressive stress fractures in methotrexate osteopathy. Calcif. Tissue Int. 108, 219–230 (2021).
Kiuru, M. J., Pihlajamaki, H. K., Hietanen, H. J. & Ahovuo, J. A. MR imaging, bone scintigraphy, and radiography in bone stress injuries of the pelvis and the lower extremity. Acta Radiol. 43, 207–212 (2002).
Eustace, S. et al. MR imaging of bone oedema: mechanisms and interpretation: pictorial review. Clin. Radiol. 56, 4–12 (2001).
Bergman, A., Fredericson, M., Ho, C. & Matheson, G. O. Asymptomatic tibial stress reactions: MRI detection and clinical follow-up in distance runners. AJR 183, 635–638 (2004).
Kaeding, C. & Miller, T. The comprehensive description of stress fractures: a new classification system. J. Bone Jt Surg. Am. 95, 1214–1220 (2013).
Nattiv, A. et al. Correlation of MRI grading of bone stress injuries with clinical risk factors and return to play: a 5-year prospective study in collegiate track and field athletes. Am. J. Sports Med. 41, 1930–1941 (2013).
Rohena-Quinquilla, I. R., Rohena-Quinquilla, F. J., Scully, W. F. & Evanson, J. R. L. Femoral neck stress injuries: analysis of 156 cases in a U.S. Military population and proposal of a new MRI classification system. AJR 210, 601–607 (2018).
Arendt, E., Agel, J., Heikes, C. & Griffiths, H. Stress injuries to bone in college athletes - a retrospective review of experience at a single institution. Am. J. Sports Med. 31, 959–968 (2003).
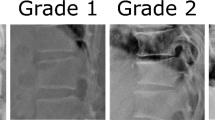
Fredericson, M., Bergman, A. G., Hoffman, K. L. & Dillingham, M. S. Tibial stress reaction in runners. Correlation of clinical symptoms and scintigraphy with a new magnetic resonance imaging grading system. Am. J. Sports Med. 23, 472–481 (1995). These researchers describe a new classification system to grade bone stress injuries at a time when MRI became the imaging modality of choice.
Beck, B. R. et al. Tibial stress injury: relationship of radiographic, nuclear medicine bone scanning, MR imaging, and CT severity grades to clinical severity and time to healing. Radiology 263, 811–818 (2012).
Shaffer, R., Brodine, S., Almeida, S., Williams, K. & Ronaghy, S. Use of simple measures of physical activity to predict stress fractures in young men undergoing a rigorous physical training program. Am. J. Epidemiol. 149, 236–242 (1999).
Ackerman, K. et al. Low energy availability surrogates correlate with health and performance consequences of relative energy deficiency in sport. Br. J. Sports Med. 53, 628–633 (2019).
Lima, F., De Falco, V., Baima, J., Carazzato, J. G. & Pereira, R. M. Effect of impact load and active load on bone metabolism and body composition of adolescent athletes. Med. Sci. Sports Exerc. 33, 1318–1323 (2001).
Duarte Sosa, D. & Fink Eriksen, E. Women with previous stress fractures show reduced bone material strength. Acta Orthop. 87, 626–631 (2016).
Warden, S. J., Liu, Z., Fuchs, R. K., van Rietbergen, B. & Moe, S. M. Reference data and calculators for second-generation HR-pQCT measures of the radius and tibia at anatomically standardized regions in white adults. Osteoporos. Int. 33, 791–806 (2021).
Schnackenburg, K. E., Macdonald, H. M., Ferber, R., Wiley, J. P. & Boyd, S. K. Bone quality and muscle strength in female athletes with lower limb stress fractures. Med. Sci. Sports Exerc. 43, 2110–2119 (2011).
Gama, E. et al. Low energy availability interferes with exercise-associated bone effects in female long-distance triathletes as detected by HR-pQCT. J. Clin. Densitom., https://doi.org/10.1016/j.jocd.2021.01.013.
Schanda, J. E. et al. Bone stress injuries are associated with differences in bone microarchitecture in male professional soldiers. J. Orthop. Res. 37, 2516–2523 (2019).
Jones, B. H., Thacker, S. B., Gilchrist, J., Kimsey, C. D. Jr & Sosin, D. M. Prevention of lower extremity stress fractures in athletes and soldiers: a systematic review. Epidemiol. Rev. 24, 228–247 (2002).
Gabbett, T. J. The training-injury prevention paradox: should athletes be training smarter and harder? Br. J. Sports Med. 50, 273–280 (2016).
Tenforde, A. S. et al. Overuse injuries in high school runners: lifetime prevalence and prevention strategies. PM R. 3, 125–131 (2011). quiz 131.
Curry, E. J., Logan, C., Ackerman, K., McInnis, K. C. & Matzkin, E. G. Female athlete triad awareness among multispecialty physicians. Sports Med. Open 1, 38 (2015).
Milgrom, C., Simkin, A., Eldad, A., Nyska, M. & Finestone, A. Using bone’s adaptation ability to lower the incidence of stress fractures. Am. J. Sports Med. 28, 245–251 (2000).
Boden, B. P. & Osbahr, D. C. High-risk stress fractures: evaluation and treatment. J. Am. Acad. Orthop. Surg. 8, 344–353 (2000). This paper highlights the importance of identifying ‘high-risk’ bone stress injuries and is a ‘must-read’ review for every sports medicine clinician.
Boden, B. P., Osbahr, D. C. & Jimenez, C. Low-risk stress fractures. Am. J. Sports Med. 29, 100–111 (2001).
Warden, S. J., Davis, I. S. & Fredericson, M. Management and prevention of bone stress injuries in long-distance runners. J. Orthop. Sports Phys. Ther. 44, 749–765 (2014).
Saxena, A. & Fullem, B. Navicular stress fractures: a prospective study on athletes. Foot Ankle Int. 27, 917–921 (2006).
Warden, S. J. Prophylactic use of NSAIDs by athletes: a risk/benefit assessment. Phys. Sportsmed. 38, 132–138 (2010).
Ackerman, K. E. et al. Effects of estrogen replacement on bone geometry and microarchitecture in adolescent and young adult oligoamenorrheic athletes: a randomized trial. J. Bone Miner. Res 35, 248–260 (2020).
Sloan, A. V., Martin, J. R., Li, S. & Li, J. Parathyroid hormone and bisphosphonate have opposite effects on stress fracture repair. Bone 47, 235–240 (2010).
Harvey, J. A., Zobitz, M. M. & Pak, C. Y. Dose dependency of calcium absorption: a comparison of calcium carbonate and calcium citrate. J. Bone Miner. Res. 3, 253–258 (1988).
Lawley, R., Syrop, I. P. & Fredericson, M. Vitamin D for improved bone health and prevention of stress fractures: a review of the literature. Curr. Sports Med. Rep. 19, 202–208 (2020).
Amrein, K. et al. Vitamin D deficiency 2.0: an update on the current status worldwide. Eur. J. Clin. Nutr. 74, 1498–1513 (2020).
Giustina, A. et al. Controversies in Vitamin D: a statement from the Third International Conference. JBMR Plus 4, e10417 (2020).
Bouillon, R. et al. The health effects of vitamin D supplementation: evidence from human studies. Nat. Rev. Endocrinol. 18, 96–110 (2022).
Larson-Meyer, D. E. & Willis, K. S. Vitamin D and athletes. Curr. Sports Med. Rep. 9, 220–226 (2010).
Gan, T. Y., Kuah, D. E., Graham, K. S. & Markson, G. Low-intensity pulsed ultrasound in lower limb bone stress injuries: a randomized controlled trial. Clin. J. Sport. Med. 24, 457–460 (2014).
Beck, B. R. et al. Do capacitively coupled electric fields accelerate tibial stress fracture healing? A randomized controlled trial. Am. J. Sports Med. 36, 545–553 (2008).
Daish, C., Blanchard, R., Fox, K., Pivonka, P. & Pirogova, E. The application of pulsed electromagnetic fields (PEMFs) for bone fracture repair: past and perspective findings. Ann. Biomed. Eng. 46, 525–542 (2018).
Taki, M., Iwata, O., Shiono, M., Kimura, M. & Takagishi, K. Extracorporeal shock wave therapy for resistant stress fracture in athletes: a report of 5 cases. Am. J. Sports Med. 35, 1188–1192 (2007).
Moretti, B. et al. Shock waves in the treatment of stress fractures. Ultrasound Med. Biol. 35, 1042–1049 (2009).
Mallee, W. H. et al. Surgical versus conservative treatment for high-risk stress fractures of the lower leg (anterior tibial cortex, navicular and fifth metatarsal base): a systematic review. Br. J. Sports Med. 49, 370–376 (2015).
Khan, K. M., Fuller, P. J., Brukner, P. D., Kearney, C. & Burry, H. C. Outcome of conservative and surgical management of navicular stress fracture in athletes. Eighty-six cases proven with computerized tomography. Am. J. Sports Med. 20, 657–666 (1992).
Torg, J. S., Moyer, J., Gaughan, J. P. & Boden, B. P. Management of tarsal navicular stress fractures: conservative versus surgical treatment: a meta-analysis. Am. J. Sports Med. 38, 1048–1053 (2010).
O’Malley, M. et al. Operative treatment of fifth metatarsal jones fractures (Zones II and III) in the NBA. Foot Ankle Int. 37, 488–500 (2016).
Varner, K. E., Younas, S. A., Lintner, D. M. & Marymont, J. V. Chronic anterior midtibial stress fractures in athletes treated with reamed intramedullary nailing. Am. J. Sports Med. 33, 1071–1076 (2005).
Chahla, J. et al. A call for standardization in platelet-rich plasma preparation protocols and composition reporting: a systematic review of the clinical orthopaedic literature. J. Bone Jt. Surg. Am. 99, 1769–1779 (2017).
Robertson, G. & Maffulli, N. in Stress Fractures in Athletes: Diagnosis and Management (eds Miller, T. L. & Kaeding, C. C.) 151–164 (Springer, 2020).
Harmon, K. G. Lower extremity stress fractures. Clin. J. Sport. Med. 13, 358–364 (2003).
Popp, K. L. et al. Changes in volumetric bone mineral density over 12 months after a tibial bone stress injury diagnosis: implications for return to sports and military duty. Am. J. Sports Med. 49, 226–235 (2021).
Ardern, C. L. et al. 2016 Consensus statement on return to sport from the First World Congress in Sports Physical Therapy, Bern. Br. J. Sports Med. 50, 853–864 (2016).
Millar, N. L. et al. Tendinopathy. Nat. Rev. Dis. Prim. 7, 1–21 (2021).
Valovich McLeod, T. C., Bay, R. C., Parsons, J. T., Sauers, E. L. & Snyder, A. R. Recent injury and health-related quality of life in adolescent athletes. J. Athl. Train. 44, 603–610 (2009).
Reis, J. P., Trone, D. W., Macera, C. A. & Rauh, M. J. Factors associated with discharge during marine corps basic training. Mil. Med. 172, 936–941 (2007).
Bull, F. C. et al. World Health Organization 2020 guidelines on physical activity and sedentary behaviour. Br. J. Sports Med. 54, 1451–1462 (2020).
Wildemann, B. et al. Non-union bone fractures. Nat. Rev. Dis. Prim. 7, 57 (2021).
Warden, S. in Bone Stress Injuries in Athletes: Diagnosis, Treatment, and Prevention (eds Tenforde, A. & Fredericson, M.) 3–12 (Springer, 2022).
Gao, C., Peng, S., Feng, P. & Shuai, C. Bone biomaterials and interactions with stem cells. Bone Res. 5, 17059 (2017).
Seeman, E. & Martin, T. J. Antiresorptive and anabolic agents in the prevention and reversal of bone fragility. Nat. Rev. Rheumatol. 15, 225–236 (2019).
Acknowledgements
S.J.W.: contribution was facilitated by support from the National Institutes of Health (NIH/NIAMS P30 AR072581) and National Basketball Association–General Electric Healthcare Orthopedics and Sports Medicine Collaboration. K.L.P.: the opinions or assertions contained herein are the private views of the authors and are not to be construed as official or reflecting the views of the U.S. Army or the Department of Defense. Any citations of commercial organizations and trade names in this report do not constitute an official U.S. Army or Department of Defense endorsement or approval of the products or services of these organizations.
Author information
Authors and Affiliations
Contributions
Introduction (T.H.); Epidemiology (K.H.); Risk factors (K.L.P., K.E.A. and T.R.); Pathophysiology (T.R., D.B.B. and M.L.B.); Diagnosis (B.R.B.); Treatment (S.J.W. and A.S.T.); Quality of life (T.H.); Outlook (T.H.). All authors critically revised all aspects of the article. T.H., A.S.T. and S.J.W. were responsible for overall handling of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Disease Primers thanks M. Baggaley, who co-reviewed with B. Edwards; G. Robertson, who co-reviewed with N. Maffulli; A. Kemper; M. Schaffler; and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Stress fractures
-
Bone stress injuries with radiologically visible sclerosis or fracture line.
- Stress reactions
-
Bone stress injuries without radiologically visible sclerosis or fracture line.
- Bone stress injury
-
Focal failure of bone tissue to repeated loading that results in localized pain and/or an increased risk of complete bone fracture to typically tolerable loads.
- Stress
-
The amount of force experienced per unit area of tissue when an external force is applied.
- Strain
-
The change in the dimensions of bone tissue when stress is applied.
- Material fatigue
-
The formation and propagation of cracks in a material due to repeated loading.
- Microdamage
-
Microscopic damage in bone tissue, often in the form of small cracks, in response to repeated stress, which stimulates targeted remodelling.
- Targeted remodelling
-
The coordinated and sequential action of bone-resorbing osteoclasts and bone-forming osteoblasts to remove bone microdamage and replace it with new bone.
- Insufficiency fracture
-
A bone injury due to loads within the normal range being applied to an abnormal bone or a bone with abnormal biology.
Rights and permissions
About this article
Cite this article
Hoenig, T., Ackerman, K.E., Beck, B.R. et al. Bone stress injuries. Nat Rev Dis Primers 8, 26 (2022). https://doi.org/10.1038/s41572-022-00352-y
Accepted:
Published:
DOI: https://doi.org/10.1038/s41572-022-00352-y
This article is cited by
-
The Middle Pleistocene human metatarsal from Sedia del Diavolo (Rome, Italy)
Scientific Reports (2024)
-
Appraising the Methodological Quality of Sports Injury Video Analysis Studies: The QA-SIVAS Scale
Sports Medicine (2024)
-
Lower Extremity Bone Stress Injuries in Athletes: an Update on Current Guidelines
Current Physical Medicine and Rehabilitation Reports (2024)
-
Changes in the incidence of stress reactions and fractures among intercollegiate athletes after the COVID-19 pandemic
Journal of Orthopaedic Surgery and Research (2023)
-
P1NP and β-CTX-1 Responses to a Prolonged, Continuous Running Bout in Young Healthy Adult Males: A Systematic Review with Individual Participant Data Meta-analysis
Sports Medicine - Open (2023)