Abstract
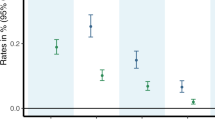
Cannabis use disorder (CUD) is an underappreciated risk of using cannabis that affects ~10% of the 193 million cannabis users worldwide. The individual and public health burdens are less than those of other forms of drug use, but CUD accounts for a substantial proportion of persons seeking treatment for drug use disorders owing to the high global prevalence of cannabis use. Cognitive behavioural therapy, motivational enhancement therapy and contingency management can substantially reduce cannabis use and cannabis-related problems, but enduring abstinence is not a common outcome. No pharmacotherapies have been approved for cannabis use or CUD, although a number of drug classes (such as cannabinoid agonists) have shown promise and require more rigorous evaluation. Treatment of cannabis use and CUD is often complicated by comorbid mental health and other substance use disorders. The legalization of non-medical cannabis use in some high-income countries may increase the prevalence of CUD by making more potent cannabis products more readily available at a lower price. States that legalize medical and non-medical cannabis use should inform users about the risks of CUD and provide information on how to obtain assistance if they develop cannabis-related mental and/or physical health problems.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 1 digital issues and online access to articles
$99.00 per year
only $99.00 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
United Nations. World Drug Report 2020 (2020). This work provides the most recent global estimates of the prevalence of cannabis use.
Peacock, A. et al. Global statistics on alcohol, tobacco and illicit drug use: 2017 status report. Addiction 113, 1905–1926 (2018).
Carvalho, A. F. et al. Cannabis use and suicide attempts among 86,254 adolescents aged 12–15 years from 21 low- and middle-income countries. Eur. Psychiatry 56, 8–13 (2019).
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5 (American Psychiatric Association, 2013).
World Health Organization. International Classification of Diseases for Mortality and Morbidity Statistics (11th Revision) (WHO, version 09/2020). https://icd.who.int/browse11/l-m/en
Degenhardt, L. et al. The global burden of disease attributable to alcohol and drug use in 195 countries and territories, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Psychiatry 5, 987–1012 (2018). This analysis provides global estimates of the burden of cannabis use and CUD.
Bloomfield, M. A. P., Ashok, A. H., Volkow, N. D. & Howes, O. D. The effects of Δ9-tetrahydrocannabinol on the dopamine system. Nature 539, 369–377 (2016).
Zehra, A. et al. Cannabis addiction and the brain: a review. J. Neuroimmune Pharmacol. 13, 438–452 (2018).
Vandrey, R. & Haney, M. Pharmacotherapy for cannabis dependence: how close are we? CNS Drugs 23, 543–553 (2009).
Hall, W., Renström, M. & Poznyak, V. The Health and Social Effects of Nonmedical Cannabis Use (World Health Organization, 2016). This is a seminal review of the short-term and long-term effects of cannabis use and CUD on health.
Simpson, A. K. & Magid, V. Cannabis use disorder in adolescence. Child. Adolesc. Psychiatr. Clin. North. Am. 25, 431–443 (2016).
Tanner-Smith, E. E., Wilson, S. J. & Lipsey, M. W. The comparative effectiveness of outpatient treatment for adolescent substance abuse: a meta-analysis. J. Subst. Abuse Treat. 44, 145–158 (2013).
Winters, K., Tanner-Smith, E., Bresani, E. & Meyers, K. Current advances in the treatment of adolescent drug use. Adolesc. Health Med. Ther. 5, 199 (2014).
Stanger, C. & Budney, A. J. Contingency management: using incentives to improve outcomes for adolescent substance use disorders. Pediatr. Clin. North. Am. 66, 1183–1192 (2019).
Budney, A. J., Sofis, M. J. & Borodovsky, J. T. An update on cannabis use disorder with comment on the impact of policy related to therapeutic and recreational cannabis use. Eur. Arch. Psychiatry Clin. Neurosci. 269, 73–86 (2019). This review and commentary convey what is known about cannabis use and the development of CUD, and predict what impact medicinal and recreational cannabis policy changes may have on these relationships.
Ferri, M., Allara, E., Bo, A., Gasparrini, A. & Faggiano, F. Media campaigns for the prevention of illicit drug use in young people. Cochrane Database Syst. Rev. 6, CD009287 (2013).
Hornik, R., Jacobsohn, L., Orwin, R., Piesse, A. & Kalton, G. Effects of the national youth anti-drug media campaign on youths. Am. J. Public Health 98, 2229–2236 (2008).
MacArthur, G. et al. Individual-, family-, and school-level interventions targeting multiple risk behaviours in young people. Cochrane Database Syst. Rev. 10, CD009927 (2018).
Bühler, A., Thrul, J. & European Monitoring Centre for Drugs and Drug Addiction. Prevention of Addictive Behaviours (Publications Office, 2015).
Stockings, E. et al. Whole-of-community interventions to reduce population-level harms arising from alcohol and other drug use: a systematic review and meta-analysis. Addiction 113, 1984–2018 (2018).
Hall, W. et al. Public health implications of legalising the production and sale of cannabis for medicinal and recreational use. Lancet 394, 1580–1590 (2019). This review discusses the potential public health impacts of changes to the legal status of production, sale and use in the Americas.
Freeman, T. P. et al. Cannabidiol for the treatment of cannabis use disorder: a phase 2a, double-blind, placebo-controlled, randomised, adaptive Bayesian trial. Lancet Psychiatry 7, 865–874 (2020).
National Academies of Sciences, Engineering, and Medicine, Health and Medicine Division, Board on Population Health and Public Health Practice, & Committee on the Health Effects of Marijuana: An Evidence Review and Research Agenda. The Health Effects of Cannabis and Cannabinoids: The Current State of Evidence and Recommendations for Research (National Academies Press, 2017).
ElSohly, M. A. & Slade, D. Chemical constituents of marijuana: the complex mixture of natural cannabinoids. Life Sci. 78, 539–548 (2005).
Expert Committee on Drug Dependence (ECDD). Cannabidiol (CBD) - Critical Review Report. WHO. https://www.who.int/medicines/access/controlled-substances/WHOCBDReportMay2018-2.pdf?ua=1 (2018).
Johnston, L. D., Miech, R. A., O’Malley, P. M., Bachman, J. G. & Schulenberg, J. E. Demographic subgroup trends among adolescents in the use of various licit and illicit drugs 1975–2017. Monitoring the Future http://www.monitoringthefuture.org/pubs/occpapers/mtf-occ90.pdf (2018).
Compton, W. M. & Han, B. in The Complex Connection between Cannabis and Schizophrenia (eds Compton, M. & Manseau, M.) (Academic Press, 2017).
Caulkins, J. P. Recognizing and regulating cannabis as a temptation good. Int. J. Drug Policy 42, 50–56 (2017).
Hasin, D. S. US epidemiology of cannabis use and associated problems. Neuropsychopharmacology 43, 195–212 (2018). This review provides an overview of US epidemiology of cannabis use and CUD, providing trends over time by reviewing results of multiple nationally representative studies. Additionally, it provides a view of how epidemiological estimates are affected by a change from DSM-IV-TR to DSM-5 diagnostic criteria.
Anthony, J. C., Warner, L. A. & Kessler, R. C. Comparative epidemiology of dependence on tobacco, alcohol, controlled substances, and inhalants: basic findings from the national comorbidity survey. Exp. Clin. Psychopharmacol. 2, 244–268 (1994).
Coffey, C. & Patton, G. C. Cannabis use in adolescence and young adulthood: a review of findings from the Victorian Adolescent Health Cohort Study. Can. J. Psychiatry 61, 318–327 (2016).
Hasin, D. S. et al. Prevalence of marijuana use disorders in the United States between 2001–2002 and 2012–2013. JAMA Psychiatry 72, 1235 (2015).
Compton, W. M., Han, B., Jones, C. M., Blanco, C. & Hughes, A. Marijuana use and use disorders in adults in the USA, 2002–14: analysis of annual cross-sectional surveys. Lancet Psychiatry 3, 954–964 (2016).
Dilley, J. A. et al. Prevalence of cannabis use in youths after legalization in Washington state. JAMA Pediatrics 173, 192 (2019).
Miech, R. A., Patrick, M. E., O’Malley, P. M., Johnston, L. D. & Bachman, J. G. Trends in reported marijuana vaping among US adolescents, 2017–2019. JAMA 323, 475–476 (2020).
Smart, R., Caulkins, J. P., Kilmer, B., Davenport, S. & Midgette, G. Variation in cannabis potency and prices in a newly legal market: evidence from 30 million cannabis sales in Washington state: legal cannabis potency and price variation. Addiction 112, 2167–2177 (2017).
Kelly, A. B., Weier, M. & Hall, W. D. in Age of Onset of Mental Disorders (eds de Girolamo, G., McGorry, P. D. & Sartorius, N.) 149–167 (Springer, 2019).
Degenhardt, L. et al. Toward a global view of alcohol, tobacco, cannabis, and cocaine use: findings from the WHO world mental health surveys. PLoS Med. 5, e141 (2008).
Butterworth, P., Slade, T. & Degenhardt, L. Factors associated with the timing and onset of cannabis use and cannabis use disorder: results from the 2007 Australian National Survey of Mental Health and Well-Being: predictors of cannabis use and CUD. Drug Alcohol. Rev. 33, 555–564 (2014).
Le Strat, Y., Dubertret, C. & Le Foll, B. Impact of age at onset of cannabis use on cannabis dependence and driving under the influence in the United States. Accid. Anal. Prev. 76, 1–5 (2015).
Behrendt, S., Wittchen, H.-U., Höfler, M., Lieb, R. & Beesdo, K. Transitions from first substance use to substance use disorders in adolescence: is early onset associated with a rapid escalation? Drug Alcohol. Depend. 99, 68–78 (2009).
Agrawal, A. & Lynskey, M. T. Cannabis controversies: how genetics can inform the study of comorbidity. Addiction 109, 360 370 (2014).
Verweij, K. J. et al. Genetic and environmental influences on cannabis use initiation and problematic use: a meta-analysis of twin studies. Addiction 105, 417–430 (2010).
Hasin, D. S. et al. Prevalence and correlates of DSM-5 cannabis use disorder, 2012–2013: findings from the National Epidemiologic Survey on Alcohol and Related Conditions–III. Am. J. Psychiatry 173, 588–599 (2016).
Cougle, J. R., Hakes, J. K., Macatee, R. J., Zvolensky, M. J. & Chavarria, J. Probability and correlates of dependence among regular users of alcohol, nicotine, cannabis, and cocaine: concurrent and prospective analyses of the National Epidemiologic Survey on Alcohol and Related Conditions. J. Clin. Psychiatry 77, e444–e450 (2016).
Peters, E. N., Budney, A. J. & Carroll, K. M. Clinical correlates of co-occurring cannabis and tobacco use: a systematic review: cannabis-tobacco clinical correlates. Addiction 107, 1404–1417 (2012).
Teesson, M., Hall, W., Lynskey, M. & Degenhardt, L. Alcohol- and drug-use disorders in Australia: implications of the National Survey of Mental Health and Wellbeing. Aust. N. Z. J. Psychiatry 34, 206–213 (2000).
Teesson, M. et al. Prevalence, correlates and comorbidity of DSM-IV cannabis use and cannabis use disorders in Australia. Aust. N. Z. J. Psychiatry 46, 1182–1192 (2012).
Feingold, D., Weiser, M., Rehm, J. & Lev-Ran, S. The association between cannabis use and anxiety disorders: results from a population-based representative sample. Eur. Neuropsychopharmacol. 26, 493–505 (2016).
Hunt, G. E., Malhi, G. S., Lai, H. M. X. & Cleary, M. Prevalence of comorbid substance use in major depressive disorder in community and clinical settings, 1990–2019: systematic review and meta-analysis. J. Affect. Disord. 266, 288–304 (2020).
Pinto, J. V. et al. The prevalence and clinical correlates of cannabis use and cannabis use disorder among patients with bipolar disorder: a systematic review with meta-analysis and meta-regression. Neurosci. Biobehav. Rev. 101, 78–84 (2019).
Hunt, G. E., Large, M. M., Cleary, M., Lai, H. M. X. & Saunders, J. B. Prevalence of comorbid substance use in schizophrenia spectrum disorders in community and clinical settings, 1990–2017: systematic review and meta-analysis. Drug Alcohol Depend. 191, 234–258 (2018).
Toftdahl, N. G., Nordentoft, M. & Hjorthøj, C. Prevalence of substance use disorders in psychiatric patients: a nationwide Danish population-based study. Soc. Psychiatry Psychiatr. Epidemiol. 51, 129–140 (2016).
Kerridge, B. T., Pickering, R., Chou, P., Saha, T. D. & Hasin, D. S. DSM-5 cannabis use disorder in the National Epidemiologic Survey on Alcohol and Related Conditions-III: gender-specific profiles. Addict. Behav. 76, 52–60 (2018).
Connor, J. P., Feeney, G. F. X., Kelly, A. B. & Saunders, J. B. in The SAGE Handbook of Drug & Alcohol Studies. Volume 2, 283–305 (SAGE Publications, 2016).
Liu, Y., Williamson, V., Setlow, B., Cottler, L. B. & Knackstedt, L. A. The importance of considering polysubstance use: lessons from cocaine research. Drug Alcohol Depend. 192, 16–28 (2018).
Connor, J. P. et al. Polysubstance use in cannabis users referred for treatment: drug use profiles, psychiatric comorbidity and cannabis-related beliefs. Front. Psychiatry 4, 1–7 (2013).
Fergusson, D. M., Boden, J. M. & Horwood, L. J. Cannabis use and other illicit drug use: testing the cannabis gateway hypothesis. Addiction 101, 556–569 (2006). This longitudinal study tracked a birth cohort annually for up to 25 years, providing a unique test of the cannabis gateway hypothesis.
Hall, W. D. & Lynskey, M. Is cannabis a gateway drug? Testing hypotheses about the relationship between cannabis use and the use of other illicit drugs. Drug Alcohol Rev. 24, 39–48 (2005).
Field, M. & Kersbergen, I. Are animal models of addiction useful? Addiction 115, 6–12 (2020).
Le Foll, B. & Goldberg, S. R. Control of the reinforcing effects of nicotine by associated environmental stimuli in animals and humans. Trends Pharmacol. Sci. 26, 287–293 (2005).
Munro, S., Thomas, K. L. & Abu-Shaar, M. Molecular characterization of a peripheral receptor for cannabinoids. Nature 365, 61–65 (1993).
Pertwee, R. G. The diverse CB1 and CB2 receptor pharmacology of three plant cannabinoids: Δ9-tetrahydrocannabinol, cannabidiol and Δ9-tetrahydrocannabivarin. Br. J. Pharmacol. 153, 199–215 (2008).
Devane, W. et al. Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 258, 1946–1949 (1992). This seminal work is the first description of the endogenous cannabinoid anandamide.
Mechoulam, R. et al. Identification of an endogenous 2-monoglyceride, present in canine gut, that binds to cannabinoid receptors. Biochem. Pharmacol. 50, 83–90 (1995).
Sugiura, T. et al. 2-Arachidonoylgylcerol: a possible endogenous cannabinoid receptor ligand in brain. Biochem. Biophys. Res. Commun. 215, 89–97 (1995).
Bisogno, T. et al. Cloning of the first sn1-DAG lipases points to the spatial and temporal regulation of endocannabinoid signaling in the brain. J. Cell Biol. 163, 463–468 (2003).
Stella, N., Schweitzer, P. & Piomelli, D. A second endogenous cannabinoid that modulates long-term potentiation. Nature 388, 773–778 (1997).
Cravatt, B. F. et al. Molecular characterization of an enzyme that degrades neuromodulatory fatty-acid amides. Nature 384, 83–87 (1996). This seminal work characterizes fatty-acid amide hydrolase, which acts to break down the endogenous cannabinoid anandamide.
Deutsch, D. G. & Chin, S. A. Enzymatic synthesis and degradation of anandamide, a cannabinoid receptor agonist. Biochem. Pharmacol. 46, 791–796 (1993).
Di Marzo, V. et al. Formation and inactivation of endogenous cannabinoid anandamide in central neurons. Nature 372, 686–691 (1994).
Dinh, T. P. et al. Brain monoglyceride lipase participating in endocannabinoid inactivation. Proc. Natl Acad. Sci. USA 99, 10819–10824 (2002).
Howlett, A. C. in Cannabinoids. Handbook of Experimental Psychology, vol 168 (ed. Pertwee, R. G.) 53–79. (Springer, 2005).
Busquets-Garcia, A., Bains, J. & Marsicano, G. CB1 receptor signaling in the brain: extracting specificity from ubiquity. Neuropsychopharmacology 43, 4–20 (2018).
Herkenham, M. et al. Characterization and localization of cannabinoid receptors in rat brain: a quantitative in vitro autoradiographic study. J. Neurosci. 11, 563–583 (1991).
Herkenham, M. et al. Cannabinoid receptor localization in brain. Proc. Natl Acad. Sci. USA 87, 1932–1936 (1990).
Katona, I. et al. Presynaptically located CB1 cannabinoid receptors regulate GABA release from axon terminals of specific hippocampal interneurons. J. Neurosci. 19, 4544–4558 (1999).
Lupica, C. R. & Riegel, A. C. Endocannabinoid release from midbrain dopamine neurons: a potential substrate for cannabinoid receptor antagonist treatment of addiction. Neuropharmacology 48, 1105–1116 (2005).
Bossong, M. G. et al. Further human evidence for striatal dopamine release induced by administration of ∆9-tetrahydrocannabinol (THC): selectivity to limbic striatum. Psychopharmacology 232, 2723–2729 (2015).
Trick, L. et al. in PET and SPECT in Psychiatry 2nd edn (eds Dierckx, R. A. J. O., Otte, A., de Vries, E. F. J., van Waarde, A. & Sommer, I. E.) 653–712 (Springer, 2020).
Wiley, J. L., Lowe, J. A., Balster, R. L. & Martin, B. R. Antagonism of the discriminative stimulus effects of delta 9-tetrahydrocannabinol in rats and rhesus monkeys. J. Pharmacol. Exp. Ther. 275, 1–6 (1995).
Huestis, M. A. et al. Blockade of effects of smoked marijuana by the CB1-selective cannabinoid receptor antagonist SR141716. Arch. Gen. Psychiatry 58, 322 (2001).
Kubilius, R. A., Kaplick, P. M. & Wotjak, C. T. Highway to hell or magic smoke? The dose-dependence of Δ9-THC in place conditioning paradigms. Learn. Mem. 25, 446–454 (2018).
Le Foll, B. & Goldberg, S. R. Cannabinoid CB1 receptor antagonists as promising new medications for drug dependence. J. Pharmacol. Exp. Ther. 312, 875–883 (2005).
Panlilio, L. V., Justinova, Z., Trigo, J. M. & Le Foll, B. Screening medications for the treatment of cannabis use disorder. Int. Rev. Neurobiol. 126, 87–120 (2016).
Schindler, C. W. et al. Blockade of nicotine and cannabinoid reinforcement and relapse by a cannabinoid CB1-receptor neutral antagonist AM4113 and inverse agonist rimonabant in squirrel monkeys. Neuropsychopharmacol 41, 2283–2293 (2016). This work compares the attenuation of nicotine and THC reinforcement and reinstatement using the CB1 receptor inverse agonist rimonabant and the CB1 receptor neutral antagonist AM4113, highlighting the potential utility of neutral antagonists in treating tobacco and cannabis dependence.
Justinova, Z., Tanda, G., Munzar, P. & Goldberg, S. R. The opioid antagonist naltrexone reduces the reinforcing effects of Δ9-tetrahydrocannabinol (THC) in squirrel monkeys. Psychopharmacology 173, 186–194 (2004). This work demonstrates for the first time the modulation of the reinforcing effects of THC self-administration of the opioid antagonist naltrexone.
Bossert, J. M., Marchant, N. J., Calu, D. J. & Shaham, Y. The reinstatement model of drug relapse: recent neurobiological findings, emerging research topics, and translational research. Psychopharmacology 229, 453–476 (2013).
Freels, T. G. et al. Vaporized cannabis extracts have reinforcing properties and support conditioned drug-seeking behavior in rats. J. Neurosci. 40, 1897–1908 (2020).
Tanda, G., Pontieri, F. E. & Di Chiara, G. Cannabinoid and heroin activation of mesolimbic dopamine transmission by a common mu1 opioid receptor mechanism. Science 276, 2048–2050 (1997).
Ibrahim, C. et al. The insula: a brain stimulation target for the treatment of addiction. Front. Pharmacol. 10, 720 (2019).
Kodas, E., Cohen, C., Louis, C. & Griebel, G. Cortico-limbic circuitry for conditioned nicotine-seeking behavior in rats involves endocannabinoid signaling. Psychopharmacology 194, 161–171 (2007).
Haney, M. et al. Nabilone decreases marijuana withdrawal and a laboratory measure of marijuana relapse. Neuropsychopharmacology 38, 1557–1565 (2013).
D’Souza, D. C. et al. Rapid changes in cannabinoid 1 receptor availability in cannabis-dependent male subjects after abstinence from cannabis. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 1, 60–67 (2016).
Haney, M. et al. Marijuana withdrawal in humans: effects of oral THC or Divalproex. Neuropsychopharmacology 29, 158–170 (2004).
Trigo, J. M. et al. Effects of fixed or self-titrated dosages of Sativex on cannabis withdrawal and cravings. Drug Alcohol. Depend. 161, 298–306 (2016).
D’Souza, D. C. et al. Efficacy and safety of a fatty acid amide hydrolase inhibitor (PF-04457845) in the treatment of cannabis withdrawal and dependence in men: a double-blind, placebo-controlled, parallel group, phase 2a single-site randomised controlled trial. Lancet Psychiatry 6, 35–45 (2019).
Sloan, M. E., Grant, C. W., Gowin, J. L., Ramchandani, V. A. & Le Foll, B. Endocannabinoid signaling in psychiatric disorders: a review of positron emission tomography studies. Acta Pharmacol. Sin. 40, 342–350 (2019). This review examines differences in cannabinoid signalling in individuals with psychiatric disorders and controls, demonstrating reduced CB1 binding in cannabis users.
Hirvonen, J. et al. Reversible and regionally selective downregulation of brain cannabinoid CB1 receptors in chronic daily cannabis smokers. Mol. Psychiatry 17, 642–649 (2012).
Di Marzo, V. et al. Enhancement of anandamide formation in the limbic forebrain and reduction of endocannabinoid contents in the striatum of Δ9-tetrahydrocannabinol-tolerant rats. J. Neurochem. 74, 1627–1635 (2002).
Morgan, C. J. A. et al. Cerebrospinal fluid anandamide levels, cannabis use and psychotic-like symptoms. Br. J. Psychiatry 202, 381–382 (2013).
Boileau, I. et al. Fatty acid amide hydrolase binding in brain of cannabis users: imaging with the novel radiotracer [11C]CURB. Biol. Psychiatry 80, 691–701 (2016).
Koob, G. F. & Volkow, N. D. Neurocircuitry of addiction. Neuropsychopharmacology 35, 217–238 (2010). This review delineates the neural circuits underlying drug use, abuse and addiction, drawing on animal and human imaging work.
Sevy, S. et al. Cerebral glucose metabolism and D2/D3 receptor availability in young adults with cannabis dependence measured with positron emission tomography. Psychopharmacology 197, 549–556 (2008).
Stokes, P. R. et al. History of cannabis use is not associated with alterations in striatal dopamine D2/D3 receptor availability. J. Psychopharmacol. 26, 144–149 (2012).
Albrecht, D. S. et al. Striatal D2/D3 receptor availability is inversely correlated with cannabis consumption in chronic marijuana users. Drug Alcohol. Depend. 128, 52–57 (2013).
Volkow, N. D. et al. Decreased dopamine brain reactivity in marijuana abusers is associated with negative emotionality and addiction severity. Proc. Natl Acad. Sci. USA 111, E3149–E3156 (2014).
John, W. S. et al. Chronic Δ9-THC in rhesus monkeys: effects on cognitive performance and dopamine D2/D3 receptor availability. J. Pharmacol. Exp. Ther. 364, 300–310 (2018).
Bloomfield, M. A. P. et al. Dopaminergic function in cannabis users and its relationship to cannabis-induced psychotic symptoms. Biol. Psychiatry 75, 470–478 (2014).
van de Giessen, E. et al. Deficits in striatal dopamine release in cannabis dependence. Mol. Psychiatry 22, 68–75 (2017).
Urban, N. B. L. et al. Dopamine release in chronic cannabis users: a [11C]raclopride positron emission tomography study. Biol. Psychiatry 71, 677–683 (2012).
Leroy, C. et al. Striatal and extrastriatal dopamine transporter in cannabis and tobacco addiction: a high-resolution PET study: DAT availability in addictions. Addiction Biol. 17, 981–990 (2012).
Bloomfield, M. A. P. et al. The neuropsychopharmacology of cannabis: a review of human imaging studies. Pharmacol. Ther. 195, 132–161 (2019).
Yanes, J. A. et al. Neuroimaging meta-analysis of cannabis use studies reveals convergent functional alterations in brain regions supporting cognitive control and reward processing. J. Psychopharmacol. 32, 283–295 (2018).
Zhou, X. et al. Cue reactivity in the ventral striatum characterizes heavy cannabis use, whereas reactivity in the dorsal striatum mediates dependent use. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 4, 751–762 (2019).
Lorenzetti, V., Chye, Y., Silva, P., Solowij, N. & Roberts, C. A. Does regular cannabis use affect neuroanatomy? An updated systematic review and meta-analysis of structural neuroimaging studies. Eur. Arch. Psychiatry Clin. Neurosci. 269, 59–71 (2019).
Chye, Y., Christensen, E. & Yücel, M. Cannabis use in adolescence: a review of neuroimaging findings. J. Dual Diagn. 16, 83–105 (2020).
Zahr, N. M. & Pfefferbaum, A. Alcohol’s effects on the brain: neuroimaging results in humans and animal models. Alcohol. Res. 38, 183–206 (2017).
Figueiredo, P. R., Tolomeo, S., Steele, J. D. & Baldacchino, A. Neurocognitive consequences of chronic cannabis use: a systematic review and meta-analysis. Neurosci. Biobehav. Rev. 108, 358–369 (2020).
Hall, W., Leung, J. & Lynskey, M. The effects of cannabis use on the development of adolescents and young adults. Annu. Rev. Dev. Psychol. 2, 461–369 (2020).
Broyd, S. J., van Hell, H. H., Beale, C., Yücel, M. & Solowij, N. Acute and chronic effects of cannabinoids on human cognition—a systematic review. Biol. Psychiatry 79, 557–567 (2016).
Mulligan, M. K. in Recent Advances in Cannabinoid Physiology and Pathology. (ed. Bukiya, A. N.) 129–150 (Springer, 2019). [Advances in Experimental Medicine and Biology vol. 1162].
Hines, L. A. et al. Overlap of heritable influences between cannabis use disorder, frequency of use and opportunity to use cannabis: trivariate twin modelling and implications for genetic design. Psychol. Med. 48, 2786–2793 (2018).
Lee, S.-Y., Chang, Y.-H. & Lu, R.-B. in Alcohol Dependence in Neuropathology of Drug Addictions and Substance Misuse Volume 1 (ed. Preedy, V. R.) 543–551 (Elsevier, 2016).
Benyamina, A., Kebir, O., Blecha, L., Reynaud, M. & Krebs, M.-O. CNR1 gene polymorphisms in addictive disorders: a systematic review and a meta-analysis. Addict. Biol. 16, 1–6 (2011).
Bidwell, L. C. et al. Impulsivity, variation in the cannabinoid receptor (CNR1) and fatty acid amide hydrolase (FAAH) genes, and marijuana-related problems. J. Stud. Alcohol. Drugs 74, 867–878 (2013).
Hindocha, C. et al. Acute effects of cannabinoids on addiction endophenotypes are moderated by genes encoding the CB1 receptor and FAAH enzyme. Addict. Biol. 25, e12762 (2020).
Saffroy, R. et al. Several clock genes polymorphisms are meaningful risk factors in the development and severity of cannabis addiction. Chronobiol. Int. 36, 122–134 (2019).
Pasman, J. A. et al. GWAS of lifetime cannabis use reveals new risk loci, genetic overlap with psychiatric traits, and a causal effect of schizophrenia liability. Nat. Neurosci. 21, 1161–1170 (2018). This work is the largest GWAS of lifetime cannabis use completed to date. Additionally, Mendelian randomization provides a test of the direction of causality between cannabis use and schizophrenia risk.
Smith, A., Kaufman, F., Sandy, M. S. & Cardenas, A. Cannabis exposure during critical windows of development: epigenetic and molecular pathways implicated in neuropsychiatric disease. Curr. Environ. Health Rep. 7, 325–342 (2020).
Heinbockel, T. & Csoka, A. Epigenetic effects of drugs of abuse. Int. J. Environ. Res. Public. Health 15, 2098 (2018).
Szutorisz, H. & Hurd, Y. L. High times for cannabis: epigenetic imprint and its legacy on brain and behavior. Neurosci. Biobehav. Rev. 85, 93–101 (2018).
McMurran, M. The Psychology of Addiction (Routledge, 1994).
Hull, J. G. & Bond, C. F. Social and behavioral consequences of alcohol consumption and expectancy: a meta-analysis. Psychol. Bull. 99, 347–360 (1986).
Bandura, A. Social Learning Theory (Prentice-Hall, 2002).
Connor, J. P., Gullo, M. J., Feeney, G. F. X., Kavanagh, D. J. & Young, R. McD. The relationship between cannabis outcome expectancies and cannabis refusal self-efficacy in a treatment population: cannabis expectancies and self-efficacy. Addiction 109, 111–119 (2014). This is the first social learning study to examine the role of cannabis outcome expectancies and self-efficacy in a cannabis-treatment-seeking sample.
Connor, J. P., Gullo, M. J., Feeney, G. F. X. & Young, R. McD. Validation of the Cannabis Expectancy Questionnaire (CEQ) in adult cannabis users in treatment. Drug Alcohol Depend. 115, 167–174 (2011).
Young, R. McD., Gullo, M. J., Feeney, G. F. X. & Connor, J. P. Development and validation of the Cannabis Refusal Self-Efficacy Questionnaire (CRSEQ) in adult cannabis users in treatment. Drug Alcohol Depend. 125, 244–251 (2012).
Bandura, A. A sociocognitive analysis of substance abuse: an agentic perspective. Psychol. Sci. 10, 214–217 (1999).
Bandura, A. Social Foundations of Thought and Action: A Social Cognitive Theory (Prentice-Hall, 1986).
Papinczak, Z. E., Connor, J. P., Feeney, G. F. X., Young, R. Mc. D. & Gullo, M. J. Treatment seeking in cannabis dependence: the role of social cognition. Drug Alcohol Depend. 170, 142–146 (2017).
Papinczak, Z. E., Connor, J. P., Harnett, P. & Gullo, M. J. A biosocial cognitive model of cannabis use in emerging adulthood. Addict. Behav. 76, 229–235 (2018).
Lewis, M. J. Alcohol: mechanisms of addiction and reinforcement. Adv. Alcohol. Subst. Abuse 9, 47–66 (1990).
Schulteis, G. & Koob, G. F. Reinforcement processes in opiate addiction: a homeostatic model. Neurochem. Res. 21, 1437–1454 (1996).
Hoch, E. & Rohrbacher, H. in Handbook of Cannabis and Related Pathologies (ed. Preedy, V.R.) e193–e201 (Elsevier, 2017).
von der Goltz, C. & Kiefer, F. Learning and memory in the aetiopathogenesis of addiction: future implications for therapy? Eur. Arch. Psychiatry Clin. Neurosci. 259, 183–187 (2009).
Hyman, S. E., Malenka, R. C. & Nestler, E. J. Neural mechanisms of addiction: the role of reward-related learning and memory. Annu. Rev. Neurosci. 29, 565–598 (2006).
White, N. M. Addictive drugs as reinforcers: multiple partial actions on memory systems. Addiction 91, 921–950 (1996).
Lisman, J. E. & Grace, A. A. The hippocampal-VTA loop: controlling the entry of information into long-term memory. Neuron 46, 703–713 (2005).
Pontieri, F. E., Tanda, G. & Di Chiara, G. Intravenous cocaine, morphine, and amphetamine preferentially increase extracellular dopamine in the ‘shell’ as compared with the ‘core’ of the rat nucleus accumbens. Proc. Natl Acad. Sci. USA 92, 12304–12308 (1995).
Ito, R., Robbins, T. W. & Everitt, B. J. Differential control over cocaine-seeking behavior by nucleus accumbens core and shell. Nat. Neurosci. 7, 389–397 (2004).
Robinson, T. The neural basis of drug craving: an incentive-sensitization theory of addiction. Brain Res. Rev. 18, 247–291 (1993).
Berridge, K. C. & Robinson, T. E. What is the role of dopamine in reward: hedonic impact, reward learning, or incentive salience? Brain Res. Rev. 28, 309–369 (1998).
Kalivas, P. W. & Volkow, N. D. The neural basis of addiction: a pathology of motivation and choice. AJP 162, 1403–1413 (2005).
Gates, P. J., Sabioni, P., Copeland, J., Le Foll, B. & Gowing, L. Psychosocial interventions for cannabis use disorder. Cochrane Database Syst. Rev. 5, CD005336 (2016). This work provides an updated systematic review of the efficacy of psychosocial interventions in treating CUD.
Schuster, R. M. et al. A contingency management method for 30-days abstinence in non-treatment seeking young adult cannabis users. Drug Alcohol Depend. 167, 199–206 (2016).
Hoch, E. et al. CANDIS-Curriculum: A Marijuana Treatment Program for Youth and Adults (Hazelden Publishing, 2017).
Fergus, S. & Zimmerman, M. A. Adolescent resilience: a framework for understanding healthy development in the face of risk. Annu. Rev. Public Health 26, 399–419 (2005).
von Sydow, K., Lieb, R., Pfister, H., Höfler, M. & Wittchen, H.-U. What predicts incident use of cannabis and progression to abuse and dependence? Drug Alcohol Depend. 68, 49–64 (2002).
Antecol, H. & Bedard, K. Does single parenthood increase the probability of teenage promiscuity, substance use, and crime? J. Popul. Econ. 20, 55–71 (2007).
Cleveland, M. J., Feinberg, M. E., Bontempo, D. E. & Greenberg, M. T. The role of risk and protective factors in substance use across adolescence. J. Adolesc. Health 43, 157–164 (2008).
Agrawal, A., Lynskey, M. T., Bucholz, K. K., Madden, P. A. F. & Heath, A. C. Correlates of cannabis initiation in a longitudinal sample of young women: the importance of peer influences. Prev. Med. 45, 31–34 (2007).
Fergusson, D. M. & Horwood, L. J. Early onset cannabis use and psychosocial adjustment in young adults. Addiction 92, 279–296 (1997). This seminal work examines the influence that early onset cannabis use has on a variety of later psychosocial outcomes.
van der Pol, P. et al. Predicting the transition from frequent cannabis use to cannabis dependence: a three-year prospective study. Drug Alcohol Depend. 133, 352–359 (2013).
Boden, J. M., Fergusson, D. M. & John Horwood, L. Illicit drug use and dependence in a New Zealand birth cohort. Aust. N. Z. J. Psychiatry 40, 156–163 (2006).
United Nations. Cannabis and Hallucinogens in World Drug Report 2019 (United Nations, 2019).
Hall, W. The adverse health effects of cannabis use: what are they, and what are their implications for policy? Int. J. Drug Policy 20, 458–466 (2009).
Coffey, C., Carlin, J. B., Lynskey, M., Li, N. & Patton, G. C. Adolescent precursors of cannabis dependence: findings from the Victorian Adolescent Health Cohort Study. Br. J. Psychiatry 182, 330–336 (2003).
Papinczak, Z. E. et al. Testing the biosocial cognitive model of substance use in cannabis users referred to treatment. Drug Alcohol Depend. 194, 216–224 (2019).
West, R. & Brown, J. A Theory of Addiction (John Wiley & Sons, 2013).
Wittchen, H.-U., Lieber, R. & Perkonig, A. in Basic and Clinical Science of Substance Related Disorders (ed. Ladewig, D.) 7–22 (Karger, 1999). [Bibliotecha Psychiatrica vol. 168].
Hasin, D. S. et al. DSM-5 criteria for substance use disorders: recommendations and rationale. Am. J. Psychiatry 170, 834–851 (2013). This article provides the main motivations and evidence in the reconceptualization of CUD from two main disorders (dependence and abuse) in DSM-IV-TR to a single disorder in DSM-5.
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-IV-TR (American Psychiatric Association, 2000).
Reed, G. M. et al. Innovations and changes in the ICD-11 classification of mental, behavioural and neurodevelopmental disorders: innovations and changes in the ICD-11 classification of mental, behavioural and neurodevelopmental disorders. World Psychiatry 18, 3–19 (2019).
Schlienz, N. J., Budney, A. J., Lee, D. C. & Vandrey, R. Cannabis withdrawal: a review of neurobiological mechanisms and sex differences. Curr. Addict. Rep. 4, 75–81 (2017). This review examines the clinical and pre-clinical research on the aetiology and neurological underpinnings of cannabis withdrawal.
Budney, A. J., Moore, B. A., Vandrey, R. G. & Hughes, J. R. The time course and significance of cannabis withdrawal. J. Abnorm. Psychol. 112, 393–402 (2003).
Livne, O., Shmulewitz, D., Lev-Ran, S. & Hasin, D. S. DSM-5 cannabis withdrawal syndrome: demographic and clinical correlates in US adults. Drug Alcohol Depend. 195, 170–177 (2019).
Lévesque, A. & Le Foll, B. When and how to treat possible cannabis use disorder. Med. Clin. North. Am. 102, 667–681 (2018).
US Preventive Services Task Force. Screening for unhealthy drug use: US Preventive Services Task Force recommendation statement. JAMA 323, 2301–2309 (2020).
Newton, A. S. et al. Instruments to detect alcohol and other drug misuse in the emergency department: a systematic review. Pediatrics 128, e180–e192 (2011).
Sobell, L. C. & Sobell, M. B. in Measuring Alcohol Consumption (eds Litten, R. Z. & Allen, J. P.) 41–72 (Humana Press, 1992).
Martin, G., Copeland, J., Gates, P. & Gilmour, S. The Severity of Dependence Scale (SDS) in an adolescent population of cannabis users: reliability, validity and diagnostic cut-off. Drug Alcohol Depend. 83, 90–93 (2006).
Swift, W., Copeland, J. & Hall, W. Choosing a diagnostic cut-off for cannabis dependence. Addiction 93, 1681–1692 (1998). This seminal work provides guidelines for choosing cut-offs for cannabis dependence diagnosis across three diagnostic instruments.
Hjorthøj, C. R., Hjorthøj, A. R. & Nordentoft, M. Validity of Timeline Follow-Back for self-reported use of cannabis and other illicit substances — systematic review and meta-analysis. Addict. Behav. 37, 225–233 (2012). This study establishes the validity of the Timeline Follow-Back instrument as a self-reported measure of cannabis and other drug use by examining agreement with biological measures of substance use.
Hamilton, I. Cannabis, psychosis and schizophrenia: unravelling a complex interaction: cannabis, psychosis and schizophrenia. Addiction 112, 1653–1657 (2017).
Ksir, C. & Hart, C. L. Cannabis and psychosis: a critical overview of the relationship. Curr. Psychiatry Rep. 18, 12 (2016).
Wilson, L., Szigeti, A., Kearney, A. & Clarke, M. Clinical characteristics of primary psychotic disorders with concurrent substance abuse and substance-induced psychotic disorders: a systematic review. Schizophr. Res. 197, 78–86 (2018).
Simonetto, D. A., Oxentenko, A. S., Herman, M. L. & Szostek, J. H. Cannabinoid hyperemesis: a case series of 98 patients. Mayo Clin. Proc. 87, 114–119 (2012).
Chocron, Y., Zuber, J.-P. & Vaucher, J. Cannabinoid hyperemesis syndrome. Br. Med.J. 366, l4336 (2019).
Sorensen, C. J., DeSanto, K., Borgelt, L., Phillips, K. T. & Monte, A. A. Cannabinoid hyperemesis syndrome: diagnosis, pathophysiology, and treatment—a systematic review. J. Med. Toxicol. 13, 71–87 (2017).
Hall, W. & Degenhardt, L. The adverse health effects of chronic cannabis use: adverse effects of chronic cannabis use. Drug Test. Anal. 6, 39–45 (2014).
Tashkin, D. P., Baldwin, G. C., Sarafian, T., Dubinett, S. & Roth, M. D. Respiratory and immunologic consequences of marijuana smoking. J. Clin. Pharmacol. 42, 71S–81S (2002).
American Psychiatric Association. Substance-related and addictive disorders. in Diagnostic and Statistical Manual of Mental Disorders (American Psychiatric Association, 2013).
Stockings, E. et al. Prevention, early intervention, harm reduction, and treatment of substance use in young people. Lancet Psychiatry 3, 280–296 (2016).
Connor, J. P. & Hall, W. Alcohol burden in low-income and middle-income countries. Lancet 386, 1922–1924 (2015).
Norberg, M. M., Kezelman, S. & Lim-Howe, N. Primary prevention of cannabis use: a systematic review of randomized controlled trials. PLoS ONE 8, e53187 (2013).
Porath-Waller, A. J., Beasley, E. & Beirness, D. J. A meta-analytic review of school-based prevention for cannabis use. Health Educ. Behav. 37, 709–723 (2010).
Carney, T., Myers, B. J., Louw, J. & Okwundu, C. I. Brief school-based interventions and behavioural outcomes for substance-using adolescents. Cochrane Database Syst. Rev. 4, CD008069 (2014). This Cochrane review evaluates the effectiveness of brief school-based interventions used to reduce substance use in adolescents.
Flynn, A. B., Falco, M. & Hocini, S. Independent evaluation of middle school-based drug prevention curricula: a systematic review. JAMA Pediatrics 169, 1046–1052 (2015).
Fischer, B. et al. Lower-risk cannabis use guidelines: a comprehensive update of evidence and recommendations. Am. J. Public Health 107, e1–e12 (2017).
Public Health Agency of Canada. Canada’s lower-risk cannabis use guidelines. Government of Canada. https://www.canada.ca/en/health-canada/services/drugs-medication/cannabis/resources/lower-risk-cannabis-use-guidelines.html (2019).
Davis, J. P., Smith, D. C., Morphew, J. W., Lei, X. & Zhang, S. Cannabis withdrawal, posttreatment abstinence, and days to first cannabis use among emerging adults in substance use treatment: a prospective study. J. Drug Issues 46, 64–83 (2016).
Bahji, A., Stephenson, C., Tyo, R., Hawken, E. R. & Seitz, D. P. Prevalence of cannabis withdrawal symptoms among people with regular or dependent use of cannabinoids: a systematic review and meta-analysis. JAMA Netw. Open 3, e202370 (2020).
Stephens, R. S., Babor, T. F., Kadden, R., Miller, M. & The Marijuana Treatment Project Research Group. The Marijuana Treatment Project: rationale, design and participant characteristics. Addiction 97, 109–124 (2002).
Montgomery, L. et al. Blunts versus joints: cannabis use characteristics and consequences among treatment-seeking adults. Drug Alcohol Depend. 198, 105–111 (2019).
Steinberg, K. L. et al. Brief counseling for marijuana dependence: a manual for treating adults. NCJRS. https://www.ncjrs.gov/app/publications/abstractDBDetails.aspx?ID=232790 (2005).
Brezing, C. A. & Levin, F. R. The current state of pharmacological treatments for cannabis use disorder and withdrawal. Neuropsychopharmacology 43, 173–194 (2018).
Zhand, N. & Milin, R. What do we know about the pharmacotheraputic management of insomnia in cannabis withdrawal: a systematic review. Am. J. Addict. 27, 453–464 (2018).
Nielsen, S., Sabioni, P., Gowing, L. & Le Foll, B. Pharmacotherapies for cannabis use disorders: clinical challenges and promising therapeutic agents. Handb. Exp. Pharmacol. 258, 355–372 (2020). This review examines the effectiveness of pharmacotherapies — compared against each other, placebo or no pharmacotherapy — in reducing symptoms of cannabis withdrawal and promoting abstinence from or reduction in cannabis use.
Davis, M. L. et al. Behavioral therapies for treatment-seeking cannabis users: a meta-analysis of randomized controlled trials. Eval. Health Prof. 38, 94–114 (2015).
Dutra, L. et al. A meta-analytic review of psychosocial interventions for substance use disorders. Am. J. Psychiatry 165, 179–187 (2008).
Magill, M. & Ray, L. A. Cognitive-behavioral treatment with adult alcohol and illicit drug users: a meta-analysis of randomized controlled trials. J. Stud. Alcohol Drugs 70, 516–527 (2009).
Martin, G. W. & Rehm, J. The effectiveness of psychosocial modalities in the treatment of alcohol problems in adults: a review of the evidence. Can. J. Psychiatry 57, 350–358 (2012).
Cuijpers, P., Karyotaki, E., de Wit, L. & Ebert, D. D. The effects of fifteen evidence-supported therapies for adult depression: a meta-analytic review. Psychotherapy Res. 30, 279–293 (2020).
Cooper, K., Chatters, R., Kaltenthaler, E. & Wong, R. Psychological and psychosocial interventions for cannabis cessation in adults: a systematic review short report. Health Technol. Assess. 19, 1–130 (2015).
Sabioni, P. & Le Foll, B. Psychosocial and pharmacological interventions for the treatment of cannabis use disorder. Focus 17, 163–168 (2019).
Danovitch, I. & Gorelick, D. A. State of the art treatments for cannabis dependence. Psychiatr. Clin. North. Am. 35, 309–326 (2012).
Kadden, R. M., Litt, M. D., Kabela-Cormier, E. & Petry, N. M. Abstinence rates following behavioral treatments for marijuana dependence. Addict. Behav. 32, 1220–1236 (2007).
Carroll, K. M. et al. The use of contingency management and motivational/skills-building therapy to treat young adults with marijuana dependence. J. Consult. Clin. Psychol. 74, 955–966 (2006).
Budney, A. J., Moore, B. A., Rocha, H. L. & Higgins, S. T. Clinical trial of abstinence-based vouchers and cognitive-behavioral therapy for cannabis dependence. J. Consult. Clin. Psychol. 74, 307–316 (2006).
Imtiaz, S. et al. Brief interventions for cannabis use in healthcare settings: systematic review and meta-analyses of randomized trials. J. Addict. Med. 14, 78–88 (2020).
Hogue, A., Henderson, C. E., Becker, S. J. & Knight, D. K. Evidence base on outpatient behavioral treatments for adolescent substance use, 2014–2017: outcomes, treatment delivery, and promising horizons. J. Clin. Child. Adolesc. Psychol. 47, 499–526 (2018).
Parmar, A. & Sarkar, S. Brief interventions for cannabis use disorders: a review. Addict. Disord. Treat. 16, 80–93 (2017).
Marsch, L. A. & Borodovsky, J. T. Technology-based interventions for preventing and treating substance use among youth. Child. Adolesc. Psychiatr. Clin. North. Am. 25, 755–768 (2016).
Nielsen, S., Gowing, L., Sabioni, P. & Le Foll, B. Pharmacotherapies for cannabis dependence. Cochrane Database Syst. Rev. 1, CD008940 (2019). This Cochrane review covers the studies testing pharmacological interventions for CUD treatment.
Connor, J. P., Saunders, J. B. & Feeney, G. F. X. in Quality of Life in Mental Disorders (eds Katschnig, H., Freeman, H. L. & Sartorius, N.) 199–208 (John Wiley & Sons, 2006).
Goldenberg, M., IsHak, W. W. & Danovitch, I. Quality of life and recreational cannabis use. Am. J. Addict. 26, 8–25 (2017).
Lev-Ran, S. et al. Gender differences in health-related quality of life among cannabis users: results from the national epidemiologic survey on alcohol and related conditions. Drug Alcohol Depend. 123, 190–200 (2012).
Rubio, J. M. et al. Quality of life following remission of mental disorders: findings from the national epidemiologic survey on alcohol and related conditions. J. Clin. Psychiatry 74, e445–e450 (2013).
Liao, J.-Y. et al. Relationships between marijuana use, severity of marijuana-related problems, and health-related quality of life. Psychiatry Res. 279, 237–243 (2019).
Hser, Y.-I. et al. Reductions in cannabis use are associated with improvements in anxiety, depression, and sleep quality, but not quality of life. J. Substance Abuse Treat. 81, 53–58 (2017).
Brezing, C. A. et al. Abstinence and reduced frequency of use are associated with improvements in quality of life among treatment-seekers with cannabis use disorder. Am. J. Addict. 27, 101–107 (2018).
Eisen, S. A. et al. Does marijuana use have residual adverse effects on self-reported health measures, socio-demographics and quality of life? A monozygotic co-twin control study in men. Addiction 97, 1137–1144 (2002).
Lorenzetti, V., Hoch, E. & Hall, W. Adolescent cannabis use, cognition, brain health and educational outcomes: a review of the evidence. Eur. Neuropsychopharmacol. 36, 169–180 (2020).
Matheson, J. et al. Acute and residual mood and cognitive performance of young adults following smoked cannabis. Pharmacol. Biochem. Behav. 194, 172937 (2020).
Kroon, E., Kuhns, L., Hoch, E. & Cousijn, J. Heavy cannabis use, dependence and the brain: a clinical perspective. Addiction 115, 559–572 (2020).
Rogeberg, O. & Elvik, R. The effects of cannabis intoxication on motor vehicle collision revisited and revised: cannabis and motor vehicle collision risk. Addiction 111, 1348–1359 (2016).
Bhandari, S., Jha, P., Lisdahl, K. M., Hillard, C. J. & Venkatesan, T. Recent trends in cyclic vomiting syndrome-associated hospitalizations with liberalization of cannabis use in the state of Colorado. Intern. Med. J. 49, 649–655 (2019).
Caulkins, J. P., Kilmer, B. & Kleiman, M. Marijuana Legalization: What Everyone Needs to Know (Oxford University Press, 2016).
Budney, A. J. & Borodovsky, J. T. The potential impact of cannabis legalization on the development of cannabis use disorders. Prev. Med. 104, 31–36 (2017). This essay illustrates several ways through which public policy and regulation of cannabis use can influence the risk of cannabis use and development of CUD.
Babor, T. F. et al. Alcohol: No Ordinary Commodity: Research and Public Policy (Oxford University Press, 2010).
Silver, L. D., Naprawa, A. Z. & Padon, A. A. Assessment of incorporation of lessons from tobacco control in city and county laws regulating legal marijuana in California. JAMA Netw. Open 3, e208393 (2020).
Borodovsky, J. T., Sofis, M. J., Grucza, R. A. & Budney, A. J. The importance of psychology for shaping legal cannabis regulation. Exp. Clin. Psychopharmacol. https://doi.org/10.1037/pha0000362 (2020).
Ayers, J. W., Caputi, T. L. & Leas, E. C. The need for federal regulation of marijuana marketing. JAMA 321, 2163 (2019).
Chan, G. C. K. et al. User characteristics and effect profile of butane hash oil: an extremely high-potency cannabis concentrate. Drug Alcohol Depend. 178, 32–38 (2017).
Freeman, T. P. et al. Changes in cannabis potency and first-time admissions to drug treatment: a 16-year study in the Netherlands. Psychol. Med. 48, 2346–2352.
Di Forti, M. et al. The contribution of cannabis use to variation in the incidence of psychotic disorder across Europe (EU-GEI): a multicentre case-control study. Lancet Psychiatry 6, 427–436 (2019).
Matheson, J. & Le Foll, B. Cannabis legalization and acute harm from high potency cannabis products: a narrative review and recommendations for public health. Front. Psychiatry 11, 591979 (2020).
Loflin, M. J. E. et al. The state of clinical outcome assessments for cannabis use disorder clinical trials: a review and research agenda. Drug Alcohol Depend. 212, 107993 (2020). This review paper, first, provides an overview of the issues related to measuring cannabis use; second, reviews the strengths and limitations of current assessment tools; third, reports on best practices for current clinical outcome assessments and end points for CUD trials; and fourth, provides recommendations for improving CUD.
Lee, D. C. et al. Systematic review of outcome domains and measures used in psychosocial and pharmacological treatment trials for cannabis use disorder. Drug Alcohol Depend. 194, 500–517 (2019).
National Advisory Council on Drug Abuse Workgroup. Recommendations for NIDA’s Cannabis Policy Research Agenda: Report from the Cannabis Policy Research Workgroup. National Institute on Drug Abuse. https://d14rmgtrwzf5a.cloudfront.net/sites/default/files/nacda_cannabis_policy_research_workgroup_report_feb_2018.pdf (2018).
Zeisser, C. et al. A ‘standard joint’? The role of quantity in predicting cannabis-related problems. Addict. Res. Theory 20, 82–92 (2012).
Freeman, T. P. & Lorenzetti, V. ‘Standard THC units’: a proposal to standardize dose across all cannabis products and methods of administration. Addiction 115, 1207–1216 (2019).
Huestis, M. A. et al. Single and multiple doses of rimonabant antagonize acute effects of smoked cannabis in male cannabis users. Psychopharmacology 194, 505–515 (2007). This work establishes the ability of rimonabant, a CB1 receptor antagonist, to attenuate the physiological effects of smoked cannabis in human participants.
Longabaugh, R., Magill, M., Morgenstern, J. & Huebner, R. in Addictions: A Comprehensive Guidebook (eds McCrady, B. S. & Epstein, E. E.) 572–596 (Oxford University Press, 2013).
McLoughlin, B. C. et al. Cannabis and schizophrenia. Cochrane Database Syst. Rev. 10, CD004837 (2014).
van der Pol, P. et al. Facilitators and barriers in treatment seeking for cannabis dependence. Drug Alcohol Depend. 133, 776–780 (2013).
Tuliao, A. P. & Holyoak, D. Psychometric properties of the perceived stigma towards substance users scale: factor structure, internal consistency, and associations with help-seeking variables. Am. J. Drug Alcohol Abuse 46, 158–166 (2020).
Boumparis, N. et al. Short- and long-term effects of digital prevention and treatment interventions for cannabis use reduction: a systematic review and meta-analysis. Drug Alcohol Depend. 200, 82–94 (2019).
Rogers, M. A., Lemmen, K., Kramer, R., Mann, J. & Chopra, V. Internet-delivered health interventions that work: systematic review of meta-analyses and evaluation of website availability. J. Med. Internet Res. 19, e90 (2017).
Hoch, E. et al. How effective and safe is medical cannabis as a treatment of mental disorders? A systematic review. Eur. Arch. Psychiatry Clin. Neurosci. 269, 87–105 (2019).
Parsons, L. H. & Hurd, Y. L. Endocannabinoid signalling in reward and addiction. Nat. Rev. Neurosci. 16, 579–594 (2015).
Agrawal, A. et al. Genome-wide association study identifies a novel locus for cannabis dependence. Mol. Psychiatry 23, 1293–1302 (2018). This meta-analysis combines data from a number of GWAS to identify genetic loci involved in the development of cannabis dependence.
Agrawal, A. et al. A genome-wide association study of DSM-IV cannabis dependence. Addict. Biol. 16, 514–518 (2011).
Sherva, R. et al. Genome-wide association study of cannabis dependence severity, novel risk variants, and shared genetic risks. JAMA Psychiatry 73, 472–480 (2016).
Gizer, I. R., Bizon, C., Gilder, D. A., Ehlers, C. L. & Wilhelmsen, K. C. Whole genome sequence study of cannabis dependence in two independent cohorts. Addict. Biol. 23, 461–473 (2018).
Stringer, S. et al. Genome-wide association study of lifetime cannabis use based on a large meta-analytic sample of 32 330 subjects from the International Cannabis Consortium. Transl. Psychiatry 6, e769 (2016).
Blecha, L., Lafaye, G. & Benyamina, A. in Cannabis Use Disorders (eds Montoya, I. D. & Weiss, S. R. B.) 13–20 (Springer, 2019).
Barr, P. B. et al. Using polygenic scores for identifying individuals at increased risk of substance use disorders in clinical and population samples. Transl. Psychiatry 10, 196 (2020).
Pasman, J. A. et al. Substance use: interplay between polygenic risk and neighborhood environment. Drug Alcohol Depend. 209, 107948 (2020).
Meyers, J. L. et al. Psychosocial moderation of polygenic risk for cannabis involvement: the role of trauma exposure and frequency of religious service attendance. Transl. Psychiatry 9, 269 (2019).
Marlatt, G. A., & Donovan, D. M. (eds) Relapse Prevention: Maintenance Strategies in the Treatment of Addictive Behaviors (Guilford Press, 1985).
Marlatt, G. A. & Gordon, J. R. in Behavioral Medicine: Changing health lifestyles (eds Davidson, P. O. & Davidson, S. M.) 410–452 (Brunner/Mazel, 1980).
Hill, K. P. et al. Nabilone pharmacotherapy for cannabis dependence: a randomized, controlled pilot study. Am. J. Addict. 26, 795–801 (2017).
Levin, F. R. et al. Dronabinol for the treatment of cannabis dependence: a randomized, double-blind, placebo-controlled trial. Drug Alcohol Depend. 116, 142–150 (2011).
Levin, F. R. et al. Dronabinol and lofexidine for cannabis use disorder: a randomized, double-blind, placebo-controlled trial. Drug Alcohol Depend. 159, 53–60 (2016).
Allsop, D. J. et al. Nabiximols as an agonist replacement therapy during cannabis withdrawal: a randomized clinical trial. JAMA Psychiatry 71, 281 (2014).
Gueye, A. B. et al. The CB1 neutral antagonist AM4113 retains the therapeutic efficacy of the inverse agonist rimonabant for nicotine dependence and weight loss with better psychiatric tolerability. Int. J. Neuropsychopharmacol. 19, pyw068 (2016).
Cooper, Z. D. & Haney, M. Opioid antagonism enhances marijuana’s effects in heavy marijuana smokers. Psychopharmacology 211, 141–148 (2010).
Ranganathan, M. et al. Naltrexone does not attenuate the effects of intravenous Δ9-tetrahydrocannabinol in healthy humans. Int. J. Neuropsychopharmacol. 15, 1251–1264 (2012).
Haney, M. et al. Naltrexone maintenance decreases cannabis self-administration and subjective effects in daily cannabis smokers. Neuropsychopharmacology 40, 2489–2498 (2015).
Notzon, D. P. et al. Open-label pilot study of injectable naltrexone for cannabis dependence. Am. J. Drug Alcohol Abuse 44, 619–627 (2018).
Gray, J. C., Treloar Padovano, H., Wemm, S. E. & Miranda, R. Predictors of topiramate tolerability in heavy cannabis–using adolescents and young adults: a secondary analysis of a randomized, double-blind, placebo-controlled trial. J. Clin. Psychopharmacol. 38, 134–137 (2018).
Miranda, R. et al. Topiramate and motivational enhancement therapy for cannabis use among youth: a randomized placebo-controlled pilot study: topiramate and cannabis use. Addict. Biol. 22, 779–790 (2017).
Mason, B. J. et al. A proof-of-concept randomized controlled study of gabapentin: effects on cannabis use, withdrawal and executive function deficits in cannabis-dependent adults. Neuropsychopharmacology 37, 1689–1698 (2012).
Gray, K. M. et al. A double-blind randomized controlled trial of N-acetylcysteine in cannabis-dependent adolescents. Am. J. Psychiatry 169, 805–812 (2012).
Gray, K. M. et al. A randomized placebo-controlled trial of N-acetylcysteine for cannabis use disorder in adults. Drug Alcohol Depend. 177, 249–257 (2017).
McRae-Clark, A. L., Baker, N. L., Maria, M. M.-S. & Brady, K. T. Effect of oxytocin on craving and stress response in marijuana-dependent individuals: a pilot study. Psychopharmacology 228, 623–631 (2013).
Adams, T. R., Arnsten, J. H., Ning, Y. & Nahvi, S. Feasibility and preliminary effectiveness of varenicline for treating co-occurring cannabis and tobacco use. J. Psychoact. Drugs 50, 12–18 (2018).
Salami, S. A. et al. It is our turn to get cannabis high: put cannabinoids in food and health baskets. Molecules 25, 4036 (2020).
Weiss, S. R. B., Howlett, K. D. & Baler, R. D. Building smart cannabis policy from the science up. Int. J. Drug Policy 42, 39–49 (2017).
Curran, H. V. et al. Keep off the grass? Cannabis, cognition and addiction. Nat. Rev. Neurosci. 17, 293–306 (2016).
Varlow, C., Boileau, I., Wey, H.-Y., Liang, S. H. & Vasdev, N. Classics in neuroimaging: imaging the endocannabinoid pathway with PET. ACS Chem. Neurosci. 11, 1855–1862 (2020).
Saunders, J. B. et al. (eds) Addiction Medicine (Oxford University Press, 2016).
Conboy, J. R. Smoke screen: America’s drug policy and medical marijuana. Food Drug Law J. 55, 601–617 (2000).
Hall, W. et al. It is premature to expand access to medicinal cannabis in hopes of solving the US opioid crisis. Addiction 113, 987–988 (2018).
Ries, N. M. Prescribe with caution: the response of canada’s medical regulatory authorities to the therapeutic use of cannabis. McGill J. Law Health 9, 215–254 (2016).
Ablin, J., Ste-Marie, P. A., Schäfer, M., Häuser, W. & Fitzcharles, M. A. Medical use of cannabis products: lessons to be learned from Israel and Canada. Schmerz 30, 3–13 (2016).
Whiting, P. F. et al. Cannabinoids for medical use: a systematic review and meta-analysis. JAMA 313, 2456–2473 (2015). This systematic review seeks to establish the effectiveness of and adverse events associated with using cannabinoids to treat disease and alleviate symptoms.
Kilmer, B. & MacCoun, R. J. How medical marijuana smoothed the transition to marijuana legalization in the United States. Annu. Rev. Law Soc. Sci. 13, 181–202 (2017).
Felson, J., Adamczyk, A. & Thomas, C. How and why have attitudes about cannabis legalization changed so much? Soc. Sci. Res. 78, 12–27 (2019).
Garvey, T. & Yeh, B. T. State legalization of recreational marijuana: selected legal issues. Congressional Research Service. https://fas.org/sgp/crs/misc/R43034.pdf (2014).
Klieger, S. B. et al. Mapping medical marijuana: state laws regulating patients, product safety, supply chains and dispensaries. Addiction 112, 2206–2216 (2017).
Leung, J., Chiu, C. Y. V., Stjepanović, D. & Hall, W. Has the legalisation of medical and recreational cannabis use in the USA affected the prevalence of cannabis use and cannabis use disorders? Curr. Addict. Rep. 5, 403–417 (2018).
Cox, C. The Canadian Cannabis Act legalizes and regulates recreational cannabis use in 2018. Health Policy 122, 205–209 (2018).
Kandel, D. B. (ed.) Stages and Pathways of Drug Involvement: Examining the Gateway Hypothesis (Cambridge University Press, 2002).
Hall, W. and Pacula, R. Cannabis Use and Dependence: Public Health and Public Policy (Cambridge University Press, 2004).
Friedman, A. L., Meurice, C. & Jutkiewicz, E. M. Effects of adolescent Δ9-tetrahydrocannabinol exposure on the behavioral effects of cocaine in adult Sprague–Dawley rats. Exp. Clin. Psychopharmacol. 27, 326–337 (2019).
Lecca, D. et al. Adolescent cannabis exposure increases heroin reinforcement in rats genetically vulnerable to addiction. Neuropharmacol. 166, 107974 (2020).
Wagner, F. A. Into the world of illegal drug use: exposure opportunity and other mechanisms linking the use of alcohol, tobacco, marijuana, and cocaine. Am. J. Epidemiol. 155, 918–925 (2002).
Fergusson, D. M., Boden, J. M. & Horwood, L. J. The developmental antecedents of illicit drug use: evidence from a 25-year longitudinal study. Drug Alcohol Depend. 96, 165–177 (2008).
Morral, A. R., McCaffrey, D. F. & Paddock, S. M. Reassessing the marijuana gateway effect. Addiction 97, 1493–1504 (2002).
Lynskey, M. T. et al. Escalation of drug use in early-onset cannabis users vs co-twin controls. JAMA 289, 427 (2003).
Stephens, R. S., Roffman, R. A., & Curtin, L. Comparison of extended versus brief treatments for marijuana use. J. Consult. Clin. Psychol. 68, 898–908 (2000).
Stein, L. A. R. et al. Validation of a measure to assess alcohol- and marijuana-related risks and consequences among incarcerated adolescents. Drug Alcohol Depend. 109, 104–113 (2010).
WHO.– The World Health Organization World Mental Health Composite International Diagnostic Interview. WHO WMH-CIDI. https://www.hcp.med.harvard.edu/wmhcidi/.
Kessler, R. C. et al. Clinical calibration of DSM-IV diagnoses in the World Mental Health (WMH) version of the World Health Organization (WHO) Composite International Diagnostic Interview (WMH-CIDI) (2021).
Fernandez-Artamendi, S., Fernández-Hermida, J. R., Muñiz-Fernández, J., Secades-Villa, R. & García-Fernández, G. Screening of cannabis-related problems among youth: the CPQ-A-S and CAST questionnaires. Subst. Abuse Treat. Prev. Policy 7, 13 (2012).
Annaheim, B., Rehm, J. & Gmel, G. How to screen for problematic cannabis use in population surveys. Eur. Addict. Res. 14, 190–197 (2008).
Thake, J. & Davis, C. G. Assessing problematic cannabis use. Addict. Res. Theory 19, 448–458 (2011).
Copeland, J., Gilmour, S., Gates, P. & Swift, W. The Cannabis Problems Questionnaire: factor structure, reliability, and validity. Drug Alcohol Depend. 80, 313–319 (2005).
Alexander, D. & Leung, P. The Marijuana Screening Inventory (MSI-X): concurrent, convergent and discriminant validity with multiple measures. Am. J. Drug Alcohol Abuse 32, 351–378 (2006).
WHO ASSIST Working Group. The Alcohol, Smoking and Substance Involvement Screening Test (ASSIST): development, reliability and feasibility. Addiction 97, 1183–1194 (2002).
Allsop, D. J., Norberg, M. M., Copeland, J., Fu, S. & Budney, A. J. The cannabis withdrawal scale development: patterns and predictors of cannabis withdrawal and distress. Drug Alcohol Depend. 119, 123–129 (2011).
Budney, A. J., Novy, P. L. & Hughes, J. R. Marijuana withdrawal among adults seeking treatment for marijuana dependence. Addiction 94, 1311–1322 (1999).
López-Pelayo, H., Batalla, A., Balcells, M. M., Colom, J. & Gual, A. Assessment of cannabis use disorders: a systematic review of screening and diagnostic instruments. Psychol. Med. 45, 1121–1133 (2015). This systematic review examines the utility of existing tools used to screen for and identify harmful cannabis use.
Acknowledgements
The Australian National Centre for Youth Substance Use Research (J.P.C., W.D.H., D.S.) is supported by funding from the Australian Government provided under the Commonwealth Drug and Alcohol Program grant. B.L.F. is supported by a clinician-scientist award from the Department of Family and Community Medicine at the University of Toronto and by the Centre for Addiction and Mental Health. E.H. has received research grants from the German Federal Ministry of Health and the European Monitoring Centre for Drugs and Drug Addiction. A.J.B. is supported in part by US National Institute on Drug Abuse (NIDA) grants P30DA029926, T32DA037202, R01DA050032. The funding bodies had no role in the study design, collection, analysis or interpretation of the data, writing the manuscript or the decision to submit the paper for publication.
Author information
Authors and Affiliations
Contributions
Introduction (J.P.C. and W.D.H.); Epidemiology (J.P.C., W.D.H., E.H. and D.S.); Mechanisms/pathophysiology (B.L.F., E.H. and J.P.C.); Diagnosis, screening and prevention (J.P.C., A.J.B., E.H. and B.L.F.); Management (J.P.C., A.J.B. and B.L.F.); Quality of life (D.S.); Outlook (J.P.C., B.L.F., W.H., A.J.B., E.H. and D.S.). All authors contributed substantially to the review and editing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
B.L.F. has obtained funding from Pfizer (GRAND Awards, including salary support) for investigator-initiated projects. B.L.F. has received in-kind donation of cannabis product from Aurora and medication donation from Pfizer and Bioprojet and was provided a coil for TMS study from Brainsway. B.L.F. has also obtained industry funding from Canopy (through research grants handled by CAMH or University of Toronto), Bioprojet, ACS and Alkermes, and has received in kind donations of nabiximols from GW Pharma for past studies funded by CIHR and NIH. E.H. receives fees for trainings on treatment of CUD from clinics and public health organizations and royalties for a treatment manual for cannabis use disorders from Hogrefe Publishing (Germany). All other authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Disease Primers thanks L. Greenwood, who co-reviewed with N. Solowij, T. Freeman, R. Gonzalez, K. Gray and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Connor, J.P., Stjepanović, D., Le Foll, B. et al. Cannabis use and cannabis use disorder. Nat Rev Dis Primers 7, 16 (2021). https://doi.org/10.1038/s41572-021-00247-4
Accepted:
Published:
DOI: https://doi.org/10.1038/s41572-021-00247-4
This article is cited by
-
Genetic influences and causal pathways shared between cannabis use disorder and other substance use traits
Molecular Psychiatry (2024)
-
Wirksamkeit eines Cannabispräventionsprogramms in der Schule: Ergebnisse einer randomisiert-prospektiven Studie
Bundesgesundheitsblatt - Gesundheitsforschung - Gesundheitsschutz (2024)
-
Predictors of effective therapy among individuals with Cannabis Use Disorder: a review of the literature
European Archives of Psychiatry and Clinical Neuroscience (2024)
-
Investigating Cannabis-Use Among Students Attending High Schools Within the Cherokee Nation Reservation 2017 and 2019
Journal of Community Health (2024)
-
Cannabis use and cannabis use disorders and their treatment in the Europe
European Archives of Psychiatry and Clinical Neuroscience (2024)