Abstract
Dystonia is a neurological condition characterized by abnormal involuntary movements or postures owing to sustained or intermittent muscle contractions. Dystonia can be the manifesting neurological sign of many disorders, either in isolation (isolated dystonia) or with additional signs (combined dystonia). The main focus of this Primer is forms of isolated dystonia of idiopathic or genetic aetiology. These disorders differ in manifestations and severity but can affect all age groups and lead to substantial disability and impaired quality of life. The discovery of genes underlying the mendelian forms of isolated or combined dystonia has led to a better understanding of its pathophysiology. In some of the most common genetic dystonias, such as those caused by TOR1A, THAP1, GCH1 and KMT2B mutations, and idiopathic dystonia, these mechanisms include abnormalities in transcriptional regulation, striatal dopaminergic signalling and synaptic plasticity and a loss of inhibition at neuronal circuits. The diagnosis of dystonia is largely based on clinical signs, and the diagnosis and aetiological definition of this disorder remain a challenge. Effective symptomatic treatments with pharmacological therapy (anticholinergics), intramuscular botulinum toxin injection and deep brain stimulation are available; however, future research will hopefully lead to reliable biomarkers, better treatments and cure of this disorder.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 1 digital issues and online access to articles
$99.00 per year
only $99.00 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
19 October 2018
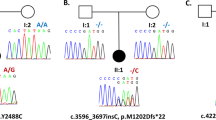
The affiliation for Enza Maria Valente and Antonio Pisani at IRCCS Santa Lucia Foundation, Rome, Italy, has been amended to remove the laboratory designation. Additionally, in Figure 3a, the subthalamic nucleus was incorrectly included in the anatomy of the brain shown and this has been amended.
References
Fahn, S., Bressman, S. B. & Marsden, C. D. Classification of dystonia. Adv. Neurol. 78, 1–10 (1998).
Albanese, A. et al. Phenomenology and classification of dystonia: a consensus update. Mov. Disord. 28, 863–873 (2013). This is a consensus update that contains the definition of dystonia and its clinical manifestations and the new classification of dystonia.
Jankovic, J. Treatment of hyperkinetic movement disorders. Lancet Neurol. 8, 844–856 (2009).
Steeves, T. D., Day, L., Dykeman, J., Jette, N. & Pringsheim, T. The prevalence of primary dystonia: a systematic review and meta-analysis. Mov. Disord. 27, 1789–1796 (2012). This is a systematic review and meta-analysis of the prevalence of isolated dystonia.
Das, S. K. et al. Community survey of primary dystonia in the city of Kolkata, India. Mov. Disord. 22, 2031–2036 (2007).
Muller, J. et al. The prevalence of primary dystonia in the general community. Neurology 59, 941–943 (2002).
Ichinose, H. et al. Hereditary progressive dystonia with marked diurnal fluctuation caused by mutations in the GTP cyclohydrolase I gene. Nat. Genet. 8, 236–242 (1994).
Williams, L. et al. Epidemiological, clinical and genetic aspects of adult onset isolated focal dystonia in Ireland. Eur. J. Neurol. 24, 73–81 (2017).
Asgeirsson, H., Jakobsson, F., Hjaltason, H., Jonsdottir, H. & Sveinbjornsdottir, S. Prevalence study of primary dystonia in Iceland. Mov. Disord. 21, 293–298 (2006).
Soland, V. L., Bhatia, K. P. & Marsden, C. D. Sex prevalence of focal dystonias. J. Neurol. Neurosurg. Psychiatry 60, 204–205 (1996).
Ozelius, L. J. et al. The early-onset torsion dystonia gene (DYT1) encodes an ATP-binding protein. Nat. Genet. 17, 40–48 (1997). This paper describes the identification of a single GAG in-frame deletion in TOR1A as the genetic cause of DYT1 dystonia, which is the most frequent genetic cause of childhood-onset generalized dystonia in the Ashkenazi Jewish population.
Risch, N. et al. Genetic analysis of idiopathic torsion dystonia in Ashkenazi Jews and their recent descent from a small founder population. Nat. Genet. 9, 152–159 (1995).
Valente, E. M. et al. The role of DYT1 in primary torsion dystonia in Europe. Brain 121, 2335–2339 (1998). This work establishes the relevance and the high mutational frequency of the common TOR1A pathogenic mutation in the European, non-Ashkenazi Jewish, population, extending the importance of screening for this mutation to all patients with generalized dystonia.
Lee, W. W., Ahn, T. B., Chung, S. J. & Jeon, B. S. Phenotypic differences in Dyt1 between ethnic groups. Curr. Neurol. Neurosci. Rep. 12, 341–347 (2012).
Fasano, A. et al. Non-DYT1 early-onset primary torsion dystonia: comparison with DYT1 phenotype and review of the literature. Mov. Disord. 21, 1411–1418 (2006).
Grundmann, K. et al. Frequency and phenotypic variability of the GAG deletion of the DYT1 gene in an unselected group of patients with dystonia. Arch. Neurol. 60, 1266–1270 (2003).
Bressman, S. B. et al. Diagnostic criteria for dystonia in DYT1 families. Neurology 59, 1780–1782 (2002).
Frederic, M. et al. First determination of the incidence of the unique TOR1A gene mutation, c.907delGAG, in a Mediterranean population. Mov. Disord. 22, 884–888 (2007).
Fuchs, T. et al. Mutations in the THAP1 gene are responsible for DYT6 primary torsion dystonia. Nat. Genet. 41, 286–288 (2009).
Bressman, S. B. et al. Mutations in THAP1 (DYT6) in early-onset dystonia: a genetic screening study. Lancet Neurol. 8, 441–446 (2009).
Djarmati, A. et al. Mutations in THAP1 (DYT6) and generalised dystonia with prominent spasmodic dysphonia: a genetic screening study. Lancet Neurol. 8, 447–452 (2009).
Blanchard, A. et al. DYT6 dystonia: review of the literature and creation of the UMD Locus-Specific Database (LSDB) for mutations in the THAP1 gene. Hum. Mutat. 32, 1213–1224 (2011).
LeDoux, M. S. et al. Genotype-phenotype correlations in THAP1 dystonia: molecular foundations and description of new cases. Parkinsonism Relat. Disord. 18, 414–425 (2012).
Fuchs, T. et al. Mutations in GNAL cause primary torsion dystonia. Nat. Genet. 45, 88–92 (2013).
Vemula, S. R. et al. Role of Gα(olf) in familial and sporadic adult-onset primary dystonia. Hum. Mol. Genet. 22, 2510–2519 (2013).
Charlesworth, G. et al. Mutations in ANO3 cause dominant craniocervical dystonia: ion channel implicated in pathogenesis. Am. J. Hum. Genet. 91, 1041–1050 (2012).
Miltgen, M. et al. Novel heterozygous mutation in ANO3 responsible for craniocervical dystonia. Mov. Disord. 31, 1251–1252 (2016).
Xiao, J. et al. Mutations in CIZ1 cause adult onset primary cervical dystonia. Ann. Neurol. 71, 458–469 (2012).
Dufke, C. et al. Mutations in CIZ1 are not a major cause for dystonia in Germany. Mov. Disord. 30, 740–743 (2015).
Martino, D. et al. Extragenetic factors and clinical penetrance of DYT1 dystonia: an exploratory study. J. Neurol. 260, 1081–1086 (2013).
Bressman, S. B. et al. Idiopathic dystonia among Ashkenazi Jews: evidence for autosomal dominant inheritance. Ann. Neurol. 26, 612–620 (1989).
Kramer, P. L. et al. The DYT1 gene on 9q34 is responsible for most cases of early limb-onset idiopathic torsion dystonia in non-Jews. Am. J. Hum. Genet. 55, 468–475 (1994).
Risch, N. J., Bressman, S. B., Senthil, G. & Ozelius, L. J. Intragenic Cis and Trans modification of genetic susceptibility in DYT1 torsion dystonia. Am. J. Hum. Genet. 80, 1188–1193 (2007).
Argyelan, M. et al. Cerebellothalamocortical connectivity regulates penetrance in dystonia. J. Neurosci. 29, 9740–9747 (2009).
Charlesworth, G. et al. Mutations in HPCA cause autosomal-recessive primary isolated dystonia. Am. J. Hum. Genet. 96, 657–665 (2015).
Cai, X. et al. Homozygous mutation of VPS16 gene is responsible for an autosomal recessive adolescent-onset primary dystonia. Sci. Rep. 6, 25834 (2016).
Zech, M. et al. Recessive mutations in the α3 (VI) collagen gene COL6A3 cause early-onset isolated dystonia. Am. J. Hum. Genet. 96, 883–893 (2015).
Carecchio, M. et al. DYT2 screening in early-onset isolated dystonia. Eur. J. Paediatr. Neurol. 21, 269–271 (2017).
Lohmann, K. et al. The role of mutations in COL6A3 in isolated dystonia. J. Neurol. 263, 730–734 (2016).
Masuho, I. et al. Homozygous GNAL mutation associated with familial childhood-onset generalized dystonia. Neurol. Genet. 2, e78 (2016).
Schneider, S. A. et al. Homozygous THAP1 mutations as cause of early-onset generalized dystonia. Mov. Disord. 26, 858–861 (2011).
Houlden, H. et al. THAP1 mutations (DYT6) are an additional cause of early-onset dystonia. Neurology 74, 846–850 (2010).
Balint, B. & Bhatia, K. P. Isolated and combined dystonia syndromes - an update on new genes and their phenotypes. Eur. J. Neurol. 22, 610–617 (2015). This review outlines the genetic forms of isolated and combined dystonia.
Fung, V. S., Jinnah, H. A., Bhatia, K. & Vidailhet, M. Assessment of patients with isolated or combined dystonia: an update on dystonia syndromes. Mov. Disord. 28, 889–898 (2013). This is an excellent paper on the approach of isolated and combined dystonia in view of the new classification of dystonia.
Paudel, R., Hardy, J., Revesz, T., Holton, J. L. & Houlden, H. Review: genetics and neuropathology of primary pure dystonia. Neuropathol. Appl. Neurobiol. 38, 520–534 (2012).
Paudel, R. et al. DYT6 dystonia: a neuropathological study. Neurodegener. Dis. 16, 273–278 (2016).
Rostasy, K. et al. TorsinA protein and neuropathology in early onset generalized dystonia with GAG deletion. Neurobiol. Dis. 12, 11–24 (2003).
McNaught, K. S. et al. Brainstem pathology in DYT1 primary torsion dystonia. Ann. Neurol. 56, 540–547 (2004).
Standaert, D. G. Update on the pathology of dystonia. Neurobiol. Dis. 42, 148–151 (2011).
Prudente, C. N. et al. Neuropathology of cervical dystonia. Exp. Neurol. 241, 95–104 (2013).
Batla, A. et al. The role of cerebellum in patients with late onset cervical/segmental dystonia? — evidence from the clinic. Parkinsonism Relat. Disord. 21, 1317–1322 (2015).
Bhatia, K. P. & Marsden, C. D. The behavioural and motor consequences of focal lesions of the basal ganglia in man. Brain: J. Neurol. 117, 859–876 (1994).
Jinnah, H. A., Neychev, V. & Hess, E. J. The anatomical basis for dystonia: the motor network model. Tremor Other Hyperkinet. Mov. 7, 506 (2017).
Egger, K. et al. Voxel based morphometry reveals specific gray matter changes in primary dystonia. Mov. Disord. 22, 1538–1542 (2007).
Pantano, P. et al. A transverse and longitudinal MR imaging voxel-based morphometry study in patients with primary cervical dystonia. AJNR Am. J. Neuroradiol. 32, 81–84 (2011).
Granert, O. et al. Manual activity shapes structure and function in contralateral human motor hand area. Neuroimage 54, 32–41 (2011).
Poston, K. L. & Eidelberg, D. Functional brain networks and abnormal connectivity in the movement disorders. Neuorimage 62, 2261–2270 (2012).
Neumann, W. J. et al. A localized pallidal physiomarker in cervical dystonia. Ann. Neurol. 82, 912–924 (2017).
Vitek, J. L. Deep brain stimulation: how does it work? Cleve. Clin. J. Med. 75 (Suppl. 2), 59–65 (2008).
Connolly, A. T. et al. Local field potential recordings in a non-human primate model of Parkinsons disease using the Activa PC + S neurostimulator. J. Neural Eng. 12, 066012 (2015).
Brittain, J. S. & Brown, P. Oscillations and the basal ganglia: motor control and beyond. Neuroimage 85, 637–647 (2014).
Berardelli, A. et al. The pathophysiology of primary dystonia. Brain 121, 1195–1212 (1998).
Berardelli, A. et al. Consensus paper on short-interval intracortical inhibition and other transcranial magnetic stimulation intracortical paradigms in movement disorders. Brain Stimul. 1, 183–191 (2008).
Lozeron, P. et al. Contribution of TMS and rTMS in the understanding of the pathophysiology and in the treatment of dystonia. Front. Neural Circuits 10, 90 (2016).
Calabresi, P. et al. Hyperkinetic disorders and loss of synaptic downscaling. Nat. Neurosci. 19, 868–875 (2016).
Martella, G. et al. Impairment of bidirectional synaptic plasticity in the striatum of a mouse model of DYT1 dystonia: role of endogenous acetylcholine. Brain 132, 2336–2349 (2009). This is the first demonstration of an impaired synaptic plasticity in a dystonia model, which recapitulates the alterations found in patients.
Martella, G. et al. Regional specificity of synaptic plasticity deficits in a knock-in mouse model of DYT1 dystonia. Neurobiol. Dis. 65, 124–132 (2014).
Calabresi, P. & Tambasco, N. Movement disorders in 2016: from genes to phenotypes. Lancet Neurol. 16, 9–10 (2017).
Quartarone, A. et al. Abnormal associative plasticity of the human motor cortex in writer’s cramp. Brain 126, 2586–2596 (2003).
Sadnicka, A., Hamada, M., Bhatia, K. P., Rothwell, J. C. & Edwards, M. J. A reflection on plasticity research in writing dystonia. Mov. Disord. 29, 980–987 (2014).
Quartarone, A. et al. Abnormal sensorimotor plasticity in organic but not in psychogenic dystonia. Brain 132, 2871–2877 (2009).
Kang, J. S., Terranova, C., Hilker, R., Quartarone, A. & Ziemann, U. Deficient homeostatic regulation of practice-dependent plasticity in writer’s cramp. Cereb. Cortex 21, 1203–1212 (2011).
Ashkan, K., Rogers, P., Bergman, H. & Ughratdar, I. Insights into the mechanisms of deep brain stimulation. Nat. Rev. Neurol. 13, 548–554 (2017). This is an excellent review on possible mechanisms of action of DBS in dystonia and Parkinson disease.
Ruge, D. et al. Deep brain stimulation effects in dystonia: time course of electrophysiological changes in early treatment. Mov. Disord. 26, 1913–1921 (2011).
Ruge, D. et al. Shaping reversibility? Long-term deep brain stimulation in dystonia: the relationship between effects on electrophysiology and clinical symptoms. Brain 134, 2106–2115 (2011).
Tinazzi, M. et al. Aristotle’s illusion reveals interdigit functional somatosensory alterations in focal hand dystonia. Brain 136, 782–789 (2013).
Nelson, A. J., Blake, D. T. & Chen, R. Digit-specific aberrations in the primary somatosensory cortex in writer’s cramp. Ann. Neurol. 66, 146–154 (2009).
Ganos, C. et al. Cortical inhibitory function in cervical dystonia. Clin. Neurophysiol. 129, 466–472 (2017).
Kojovic, M. et al. Secondary and primary dystonia: pathophysiological differences. Brain 136, 2038–2049 (2013).
Macerollo, A. et al. Abnormal movement-related suppression of sensory evoked potentials in upper limb dystonia. Eur. J. Neurol. 23, 562–568 (2016).
Langbour, N. et al. The cortical processing of sensorimotor sequences is disrupted in writer’s cramp. Cereb. Cortex 27, 2544–2559 (2017).
Naumann, M., Magyar-Lehmann, S., Reiners, K., Erbguth, F. & Leenders, K. L. Sensory tricks in cervical dystonia: perceptual dysbalance of parietal cortex modulates frontal motor programming. Ann. Neurol. 47, 322–328 (2000).
Gomez-Wong, E. et al. The ‘geste antagonistique’ induces transient modulation of the blink reflex in human patients with blepharospasm. Neurosci. Lett. 251, 125–128 (1998).
Brighina, F. et al. Effects of cerebellar TMS on motor cortex of patients with focal dystonia: a preliminary report. Exp. Brain Res. 192, 651–656 (2009).
Kaji, R., Bhatia, K. & Graybiel, A. M. Pathogenesis of dystonia: is it of cerebellar or basal ganglia origin? J. Neurol. Neurosurg. Psychiatry 89, 488–492 (2017).
Sadnicka, A. et al. All in the blink of an eye: new insight into cerebellar and brainstem function in DYT1 and DYT6 dystonia. Eur. J. Neurol. 22, 762–767 (2015).
Carbon, M. et al. Increased cerebellar activation during sequence learning in DYT1 carriers: an equiperformance study. Brain 131, 146–154 (2008).
Carbon, M. et al. Abnormal striatal and thalamic dopamine neurotransmission: genotype-related features of dystonia. Neurology 72, 2097–2103 (2009).
Katschnig-Winter, P. et al. Motor sequence learning and motor adaptation in primary cervical dystonia. J. Clin. Neurosci. 21, 934–938 (2014).
Sadnicka, A. et al. Normal motor adaptation in cervical dystonia: a fundamental cerebellar computation is intact. Cerebellum 13, 558–567 (2014).
Perlmutter, J. S. & Mink, J. W. Dysfunction of dopaminergic pathways in dystonia. Adv. Neurol. 94, 163–170 (2004).
Wichmann, T. Commentary: dopaminergic dysfunction in DYT1 dystonia. Exp. Neurol. 212, 242–246 (2008).
Ichinose, H., Inagaki, H., Suzuki, T., Ohye, T. & Nagatsu, T. Molecular mechanisms of hereditary progressive dystonia with marked diurnal fluctuation, Segawa’s disease. Brain Dev. 22 (Suppl. 1), 107–110 (2000).
Knappskog, P. M., Flatmark, T., Mallet, J., Ludecke, B. & Bartholome, K. Recessively inherited L-DOPA-responsive dystonia caused by a point mutation (Q381K) in the tyrosine hydroxylase gene. Hum. Mol. Genet. 4, 1209–1212 (1995).
van den Heuvel, L. P. et al. A common point mutation in the tyrosine hydroxylase gene in autosomal recessive L-DOPA-responsive dystonia in the Dutch population. Hum. Genet. 102, 644–646 (1998).
Thony, B. & Blau, N. Mutations in the BH4-metabolizing genes GTP cyclohydrolase I, 6-pyruvoyl-tetrahydropterin synthase, sepiapterin reductase, carbinolamine-4a-dehydratase, and dihydropteridine reductase. Hum. Mut. 27, 870–878 (2006).
Teo, J. T., Edwards, M. J. & Bhatia, K. Tardive dyskinesia is caused by maladaptive synaptic plasticity: a hypothesis. Mov. Disord. 27, 1205–1215 (2012).
Asanuma, K. et al. Decreased striatal D2 receptor binding in non-manifesting carriers of the DYT1 dystonia mutation. Neurology 64, 347–349 (2005).
Berman, B. D., Hallett, M., Herscovitch, P. & Simonyan, K. Striatal dopaminergic dysfunction at rest and during task performance in writer’s cramp. Brain 136, 3645–3658 (2013).
Black, K. J. et al. Spatial reorganization of putaminal dopamine D2-like receptors in cranial and hand dystonia. PLOS ONE 9, e88121 (2014).
Karimi, M. et al. Striatal dopamine D1-like receptor binding is unchanged in primary focal dystonia. Mov. Disord. 28, 2002–2006 (2013).
Simonyan, K., Cho, H., Hamzehei Sichani, A., Rubien-Thomas, E. & Hallett, M. The direct basal ganglia pathway is hyperfunctional in focal dystonia. Brain 140, 3179–3190 (2017).
Goodchild, R. E., Grundmann, K. & Pisani, A. New genetic insights highlight ‘old’ ideas on motor dysfunction in dystonia. Trends Neurosci. 36, 717–725 (2013).
Mencacci, N. E. et al. De novo mutations in PDE10A cause childhood-onset chorea with bilateral striatal lesions. Am. J. Hum. Genet. 98, 763–771 (2016).
Diggle, C. P. et al. Biallelic mutations in PDE10A lead to loss of striatal PDE10A and a hyperkinetic movement disorder with onset in infancy. Am. J. Hum. Genet. 98, 735–743 (2016).
Herve, D. et al. G(olf) and Gs in rat basal ganglia: possible involvement of G(olf) in the coupling of dopamine D1 receptor with adenylyl cyclase. J. Neurosci. 13, 2237–2248 (1993).
Herve, D. et al. Galpha(olf) levels are regulated by receptor usage and control dopamine and adenosine action in the striatum. J. Neurosci. 21, 4390–4399 (2001).
Chen, Y. Z. et al. Gain-of-function ADCY5 mutations in familial dyskinesia with facial myokymia. Ann. Neurol. 75, 542–549 (2014).
Herve, D. Identification of a specific assembly of the g protein golf as a critical and regulated module of dopamine and adenosine-activated cAMP pathways in the striatum. Front. Neuroanat. 5, 48 (2011).
Corvol, J. C., Studler, J. M., Schonn, J. S., Girault, J. A. & Herve, D. Galpha(olf) is necessary for coupling D1 and A2a receptors to adenylyl cyclase in the striatum. J. Neurochem. 76, 1585–1588 (2001).
Corvol, J. C. et al. Quantitative changes in Galphaolf protein levels, but not D1 receptor, alter specifically acute responses to psychostimulants. Neuropsychopharmacology 32, 1109–1121 (2007).
Alcacer, C. et al. Gα(olf) mutation allows parsing the role of cAMP-dependent and extracellular signal-regulated kinase-dependent signaling in L-3,4-dihydroxyphenylalanine-induced dyskinesia. J. Neurosci. 32, 5900–5910 (2012).
Li, Q. et al. Conserved properties of Drosophila insomniac link sleep regulation and synaptic function. PLOS Genet. 13, e1006815 (2017).
Tzingounis, A. V., Kobayashi, M., Takamatsu, K. & Nicoll, R. A. Hippocalcin gates the calcium activation of the slow afterhyperpolarization in hippocampal pyramidal cells. Neuron 53, 487–493 (2007).
Pfeiffenberger, C. & Allada, R. Cul3 and the BTB adaptor insomniac are key regulators of sleep homeostasis and a dopamine arousal pathway in Drosophila. PLOS Genet. 8, e1003003 (2012).
Mencacci, N. E. et al. A missense mutation in KCTD17 causes autosomal dominant myoclonus-dystonia. Am. J. Hum. Genet. 96, 938–947 (2015).
Chen, K.-F., Lamaze, A., Kratschmer, P. & Jepson, J. E. C. Neurocalcin acts in a clock- and light-modulated dopaminergic pathway to promote night sleep in Drosophila. Preprint at https://www.biorxiv.org/content/early/2017/11/13/159772 (2017).
Meyer, E. et al. Mutations in the histone methyltransferase gene KMT2B cause complex early-onset dystonia. Nat. Genet. 49, 223–237 (2017).
Napolitano, F. et al. Dopamine D2 receptor dysfunction is rescued by adenosine A2A receptor antagonism in a model of DYT1 dystonia. Neurobiol. Dis. 38, 434–445 (2010).
Zhang, L., Yokoi, F., Parsons, D. S., Standaert, D. G. & Li, Y. Alteration of striatal dopaminergic neurotransmission in a mouse model of DYT11 myoclonus-dystonia. PLOS ONE 7, e33669 (2012).
Yokoi, F., Dang, M. T., Li, J., Standaert, D. G. & Li, Y. Motor deficits and decreased striatal dopamine receptor 2 binding activity in the striatum-specific Dyt1 conditional knockout mice. PLOS ONE 6, e24539 (2011).
Maltese, M. et al. Abnormal striatal plasticity in a DYT11/SGCE myoclonus dystonia mouse model is reversed by adenosine A2A receptor inhibition. Neurobiol. Dis. 108, 128–139 (2017).
Eskow Jaunarajs, K. L., Bonsi, P., Chesselet, M. F., Standaert, D. G. & Pisani, A. Striatal cholinergic dysfunction as a unifying theme in the pathophysiology of dystonia. Prog. Neurobiol. 127–128, 91–107 (2015).
Maltese, M. et al. Anticholinergic drugs rescue synaptic plasticity in DYT1 dystonia: role of M1 muscarinic receptors. Mov. Disord. 29, 1655–1665 (2014).
Dang, M. T. et al. An anticholinergic reverses motor control and corticostriatal LTD deficits in Dyt1 DeltaGAG knock-in mice. Behav. Brain Res. 226, 465–472 (2012).
Helassa, N., Antonyuk, S. V., Lian, L. Y., Haynes, L. P. & Burgoyne, R. D. Biophysical and functional characterization of hippocalcin mutants responsible for human dystonia. Hum. Mol. Genet. 26, 2426–2435 (2017).
Goodchild, R. E. & Dauer, W. T. The AAA+ protein torsinA interacts with a conserved domain present in LAP1 and a novel ER protein. J. Cell Biol. 168, 855–862 (2005).
Kustedjo, K., Bracey, M. H. & Cravatt, B. F. Torsin A and its torsion dystonia-associated mutant forms are lumenal glycoproteins that exhibit distinct subcellular localizations. J. Biol. Chem. 275, 27933–27939 (2000).
Goodchild, R. E. & Dauer, W. T. Mislocalization to the nuclear envelope: an effect of the dystonia-causing torsinA mutation. Proc. Natl Acad. Sci. USA 101, 847–852 (2004). This paper reports the finding that the mutant torsin A1 protein relocalizes from the endoplasmic reticulum to the nuclear envelope, suggesting that abnormal function of these subcellular structures is critical in DYT1 dystonia pathogenesis.
Hewett, J. W. et al. Mutant torsinA interferes with protein processing through the secretory pathway in DYT1 dystonia cells. Proc. Natl Acad. Sci. USA 104, 7271–7276 (2007).
Chen, P. et al. The early-onset torsion dystonia-associated protein, torsinA, is a homeostatic regulator of endoplasmic reticulum stress response. Hum. Mol. Genet. 19, 3502–3515 (2010).
Nery, F. C. et al. TorsinA participates in endoplasmic reticulum-associated degradation. Nat. Commun. 2, 393 (2011).
Liang, C. C., Tanabe, L. M., Jou, S., Chi, F. & Dauer, W. T. TorsinA hypofunction causes abnormal twisting movements and sensorimotor circuit neurodegeneration. J. Clin. Invest. 124, 3080–3092 (2014).
Grillet, M. et al. Torsins are essential regulators of cellular lipid metabolism. Dev. Cell 38, 235–247 (2016).
Rittiner, J. E. et al. Functional genomic analyses of mendelian and sporadic disease identify impaired eIF2alpha signaling as a generalizable mechanism for dystonia. Neuron 92, 1238–1251 (2016).
Di Prisco, G. V. et al. Translational control of mGluR-dependent long-term depression and object-place learning by eIF2alpha. Nat. Neurosci. 17, 1073–1082 (2014).
Trinh, M. A. et al. The eIF2alpha kinase PERK limits the expression of hippocampal metabotropic glutamate receptor-dependent long-term depression. Learn. Mem. 21, 298–304 (2014).
Vaughn, L. S. et al. Altered activation of protein kinase PKR and enhanced apoptosis in dystonia cells carrying a mutation in PKR activator protein PACT. J. Biol. Chem. 290, 22543–22557 (2015).
Bessiere, D. et al. Structure-function analysis of the THAP zinc finger of THAP1, a large C2CH DNA-binding module linked to Rb/E2F pathways. J. Biol. Chem. 283, 4352–4363 (2008).
Santos-Rosa, H. et al. Active genes are tri-methylated at K4 of histone H3. Nature 419, 407–411 (2002).
Kaiser, F. J. et al. The dystonia gene DYT1 is repressed by the transcription factor THAP1 (DYT6). Ann. Neurol. 68, 554–559 (2010).
Ruiz, M. et al. Abnormalities of motor function, transcription and cerebellar structure in mouse models of THAP1 dystonia. Hum. Mol. Genet. 24, 7159–7170 (2015).
Zhao, Y., Xiao, J., Gong, S., Clara, J. A. & Ledoux, M. S. Neural expression of the transcription factor THAP1 during development in rat. Neuroscience 231, 282–295 (2013).
Aguilo, F. et al. THAP1: role in mouse embryonic stem cell survival and differentiation. Stem Cell Rep 9, 92–107 (2017).
Sitburana, O., Wu, L. J., Sheffield, J. K., Davidson, A. & Jankovic, J. Motor overflow and mirror dystonia. Parkinsonism Relat. Disord. 15, 758–761 (2009).
Stamelou, M., Edwards, M. J., Hallett, M. & Bhatia, K. P. The non-motor syndrome of primary dystonia: clinical and pathophysiological implications. Brain 135, 1668–1681 (2012).
Patel, N., Hanfelt, J., Marsh, L. & Jankovic, J. Alleviating manoeuvres (sensory tricks) in cervical dystonia. J. Neurol. Neurosurg. Psychiatry 85, 882–884 (2014).
Rubio-Agusti, I. et al. Tremulous cervical dystonia is likely to be familial: clinical characteristics of a large cohort. Parkinsonism Relat. Disord. 19, 634–638 (2013).
Bressman, S. B. et al. The DYT1 phenotype and guidelines for diagnostic testing. Neurology 54, 1746–1752 (2000).
Stamelou, M. et al. The phenotypic spectrum of DYT24 due to ANO3 mutations. Mov. Disord. 29, 928–934 (2014).
Wijemanne, S. & Jankovic, J. Dopa-responsive dystonia—clinical and genetic heterogeneity. Nat. Rev. Neurol. 11, 414–424 (2015).
Camargos, S. et al. DYT16, a novel young-onset dystonia-parkinsonism disorder: identification of a segregating mutation in the stress-response protein PRKRA. Lancet Neurol. 7, 207–215 (2008).
Dos Santos, C. O. et al. The prevalence of PRKRA mutations in idiopathic dystonia. Parkinsonism Relat. Disord. 48, 93–96 (2018).
Quadri, M. et al. PRKRA mutation causing early-onset generalized dystonia-parkinsonism (DYT16) in an Italian family. Mov. Disord. 31, 765–767 (2016).
Aneichyk, T. et al. Dissecting the causal mechanism of X-linked dystonia-parkinsonism by integrating genome and transcriptome assembly. Cell 172, 897–909 (2018).
de Carvalho Aguiar, P. et al. Mutations in the Na+/K+ -ATPase alpha3 gene ATP1A3 are associated with rapid-onset dystonia parkinsonism. Neuron 43, 169–175 (2004).
Brashear, A. et al. The phenotypic spectrum of rapid-onset dystonia-parkinsonism (RDP) and mutations in the ATP1A3 gene. Brain 130, 828–835 (2007).
Rosewich, H. et al. Heterozygous de-novo mutations in ATP1A3 in patients with alternating hemiplegia of childhood: a whole-exome sequencing gene-identification study. Lancet Neurol. 11, 764–773 (2012).
Heinzen, E. L. et al. De novo mutations in ATP1A3 cause alternating hemiplegia of childhood. Nat. Genet. 44, 1030–1034 (2012).
Heinzen, E. L. et al. Distinct neurological disorders with ATP1A3 mutations. Lancet Neurol. 13, 503–514 (2014).
Zech, M. et al. Haploinsufficiency of KMT2B, encoding the lysine-specific histone methyltransferase 2B, results in early-onset generalized dystonia. Am. J. Hum. Genet. 99, 1377–1387 (2016).
Zech, M. et al. KMT2B rare missense variants in generalized dystonia. Mov. Disord. 32, 1087–1091 (2017).
Zimprich, A. et al. Mutations in the gene encoding epsilon-sarcoglycan cause myoclonus-dystonia syndrome. Nat. Genet. 29, 66–69 (2001).
Chen, D. H. et al. ADCY5-related dyskinesia: broader spectrum and genotype-phenotype correlations. Neurology 85, 2026–2035 (2015).
Mencacci, N. E. et al. ADCY5 mutations are another cause of benign hereditary chorea. Neurology 85, 80–88 (2015).
Chang, F. C. et al. Phenotypic insights into ADCY5-associated disease. Mov. Disord. 31, 1033–1040 (2016).
Balint, B. & Bhatia, K. P. Dystonia: an update on phenomenology, classification, pathogenesis and treatment. Curr. Opin. Neurol. 27, 468–476 (2014).
Jinnah, H. A. et al. Treatable inherited rare movement disorders. Mov. Disord. 33, 21–35 (2017). This is a comprehensive review of specific therapeutic approaches in hereditary dystonias.
Edwards, M. J., Stamelou, M., Quinn, N. & Bhatia, K. P. Parkinson’s Disease and Other Movement Disorders (Oxford Univ. Press, 2016).
Erro, R. et al. The clinical syndrome of paroxysmal exercise-induced dystonia: diagnostic outcomes and an algorithm. Mov. Disord. Clin. Pract. 1, 57–61 (2014).
De Giorgis, V. & Veggiotti, P. GLUT1 deficiency syndrome 2013: current state of the art. Seizure 22, 803–811 (2013).
Frei, K., Truong, D. D., Fahn, S., Jankovic, J. & Hauser, R. A. The nosology of tardive syndromes. J. Neurol. Sci. 389, 10–16 (2018).
Vijayakumar, D. & Jankovic, J. Drug-induced dyskinesia, part 2: treatment of tardive dyskinesia. Drugs 76, 779–787 (2016).
Thenganatt, M. A. & Jankovic, J. Treatment of dystonia. Neurotherapeutics 11, 139–152 (2014).
Pietracupa, S. et al. Scales for hyperkinetic disorders: a systematic review. J. Neurol. Sci. 358, 9–21 (2015).
Jankovic, J. Botulinum toxin: state of the art. Mov. Disord. 32, 1131–1138 (2017). Botulinum toxin has revolutionized the treatment of focal dystonia. This paper summarizes the current knowledge and state of the art strategy for the use of botulinum toxin in dystonia and beyond.
Pirio Richardson, S., Wegele, A. R., Skipper, B., Deligtisch, A. & Jinnah, H. A. Dystonia treatment: patterns of medication use in an international cohort. Neurology 88, 543–550 (2017).
Pirazzini, M., Rossetto, O., Eleopra, R. & Montecucco, C. Botulinum neurotoxins: biology, pharmacology, and toxicology. Pharmacol. Rev. 69, 200–235 (2017).
Simpson, D. M. et al. Practice guideline update summary: botulinum neurotoxin for the treatment of blepharospasm, cervical dystonia, adult spasticity, and headache: report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology 86, 1818–1826 (2016).
Hallett, M. et al. Evidence-based review and assessment of botulinum neurotoxin for the treatment of movement disorders. Toxicon 67, 94–114 (2013).
Contarino, M. F. et al. Clinical practice: evidence-based recommendations for the treatment of cervical dystonia with botulinum toxin. Front. Neurol. 8, 35 (2017).
Comella, C. et al. A phase 2, open-label, dose-escalating study to evaluate the safety and preliminary efficacy of daxibotulinumtoxina for injection (RT002) in isolated cervical dystonia. Neurology 88 (Suppl. 16), P3.021 (2017).
Contarino, M. F., Smit, M., van den Dool, J., Volkmann, J. & Tijssen, M. A. Unmet needs in the management of cervical dystonia. Front. Neurol. 7, 165 (2016).
De Pauw, J. et al. The effectiveness of physiotherapy for cervical dystonia: a systematic literature review. J. Neurol. 261, 1857–1865 (2014).
Counsell, C. et al. A randomized trial of specialized versus standard neck physiotherapy in cervical dystonia. Parkinsonism Relat. Disord. 23, 72–79 (2016).
Boyce, M. J. et al. Active exercise for individuals with cervical dystonia: a pilot randomized controlled trial. Clin. Rehabil. 27, 226–235 (2013).
Tassorelli, C. et al. Botulinum toxin and neuromotor rehabilitation: an integrated approach to idiopathic cervical dystonia. Mov. Disord. 21, 2240–2243 (2006).
Bleton, J. P. et al. Baseline Features Influencing the Effectiveness of Retraining Therapy for Writer’s Cramp. Mov. Disord. Clin. Pract. 2, 232–236 (2015).
Priori, A., Pesenti, A., Cappellari, A., Scarlato, G. & Barbieri, S. Limb immobilization for the treatment of focal occupational dystonia. Neurology 57, 405–409 (2001).
Pirio Richardson, S. et al. Research priorities in limb and task-specific dystonias. Front. Neurol. 8, 170 (2017).
Cho, H. J. & Hallett, M. Non-invasive brain stimulation for treatment of focal hand dystonia: update and future direction. J. Mov. Disord. 9, 55–62 (2016).
Erro, R., Tinazzi, M., Morgante, F. & Bhatia, K. P. Non-invasive brain stimulation for dystonia: therapeutic implications. Eur. J. Neurol. 24, 1228 (2017).
Burke, R. E., Fahn, S. & Marsden, C. D. Torsion dystonia: a double-blind, prospective trial of high-dosage trihexyphenidyl. Neurology 36, 160–164 (1986).
Lumsden, D. E., Kaminska, M., Tomlin, S. & Lin, J. P. Medication use in childhood dystonia. Eur. J. Paediatr. Neurol. 20, 625–629 (2016).
Alfradique-Dunham, I. & Jankovic, J. Available treatment options for dystonia. Expert Opin. Orphan Drugs 5, 707–716 (2017).
Vijayakumar, D. & Jankovic, J. Drug-induced dyskinesia, part 1: treatment of levodopa-induced dyskinesia. Drugs 76, 759–777 (2016).
Jankovic, J. Dopamine depleters in the treatment of hyperkinetic movement disorders. Expert Opin. Pharmacother. 17, 2461–2470 (2016).
Solmi, M., Pigato, G., Kane, J. M. & Correll, C. U. Treatment of tardive dyskinesia with VMAT-2 inhibitors: a systematic review and meta-analysis of randomized controlled trials. Drug Des. Devel. Ther. 12, 1215–1238 (2018).
Monbaliu, E. et al. Clinical presentation and management of dyskinetic cerebral palsy. Lancet Neurol. 16, 741–749 (2017).
Motta, F. & Antonello, C. E. Analysis of complications in 430 consecutive pediatric patients treated with intrathecal baclofen therapy: 14-year experience. J. Neurosurg. Pediatr. 13, 301–306 (2014).
Turner, M., Nguyen, H. S. & Cohen-Gadol, A. A. Intraventricular baclofen as an alternative to intrathecal baclofen for intractable spasticity or dystonia: outcomes and technical considerations. J. Neurosurg. Pediatr. 10, 315–319 (2012).
Hainque, E. et al. A randomized, controlled, double-blind, crossover trial of zonisamide in myoclonus-dystonia. Neurology 86, 1729–1735 (2016).
Bomalaski, M. N., Claflin, E. S., Townsend, W. & Peterson, M. D. Zolpidem for the treatment of neurologic disorders: a systematic review. JAMA Neurol. 74, 1130–1139 (2017).
Liow, N. Y. et al. Gabapentin can significantly improve dystonia severity and quality of life in children. Eur. J. Paediatr. Neurol. 20, 100–107 (2016).
Vijayakumar, D., Wijemanne, S. & Jankovic, J. Treatment of blepharospasm with apraclonidine. J. Neurol. Sci. 372, 57–59 (2017).
Moro, E. et al. Efficacy of pallidal stimulation in isolated dystonia: a systematic review and meta-analysis. Eur. J. Neurol. 24, 552–560 (2017). This is a comprehensive meta-analysis with a systematic review of the literature on DBS in dystonia. This paper provides strong clinical evidence of the efficacy of DBS of the GPi in isolated inherited or idiopathic dystonia.
Vidailhet, M. et al. Bilateral, pallidal, deep-brain stimulation in primary generalised dystonia: a prospective 3 year follow-up study. Lancet Neurol. 6, 223–229 (2007).
Volkmann, J. et al. Pallidal deep brain stimulation in patients with primary generalised or segmental dystonia: 5-year follow-up of a randomised trial. Lancet Neurol. 11, 1029–1038 (2012).
Volkmann, J. et al. Pallidal neurostimulation in patients with medication-refractory cervical dystonia: a randomised, sham-controlled trial. Lancet Neurol. 13, 875–884 (2014).
Reese, R. et al. Long-term clinical outcome in meige syndrome treated with internal pallidum deep brain stimulation. Mov. Disord. 26, 691–698 (2011).
Schrader, C. et al. GPi-DBS may induce a hypokinetic gait disorder with freezing of gait in patients with dystonia. Neurology 77, 483–488 (2011).
Ostrem, J. L. et al. Subthalamic nucleus deep brain stimulation in isolated dystonia: a 3-year follow-up study. Neurology 88, 25–35 (2017).
Mills, K. A., Scherzer, R., Starr, P. A. & Ostrem, J. L. Weight change after globus pallidus internus or subthalamic nucleus deep brain stimulation in Parkinson’s disease and dystonia. Stereotact. Funct. Neurosurg. 90, 386–393 (2012).
Baizabal Carvallo, J. F. et al. Deep brain stimulation hardware complications in patients with movement disorders: risk factors and clinical correlations. Stereotact. Funct. Neurosurg. 90, 300–306 (2012).
Pouclet-Courtemanche, H. et al. Long-term efficacy and tolerability of bilateral pallidal stimulation to treat tardive dyskinesia. Neurology 86, 651–659 (2016).
Azoulay-Zyss, J. et al. Bilateral deep brain stimulation of the pallidum for myoclonus-dystonia due to epsilon-sarcoglycan mutations: a pilot study. Arch. Neurol. 68, 94–98 (2011).
Panov, F. et al. Deep brain stimulation in DYT1 dystonia: a 10-year experience. Neurosurgery 73, 86–93 (2013).
Krause, P. et al. Long-term effect on dystonia after pallidal deep brain stimulation (DBS) in three members of a family with a THAP1 mutation. J. Neurol. 262, 2739–2744 (2015).
Panov, F. et al. Pallidal deep brain stimulation for DYT6 dystonia. J. Neurol. Neurosurg. Psychiatry 83, 182–187 (2012).
Ziegan, J. et al. Novel GNAL mutations in two German patients with sporadic dystonia. Mov Disord. 29, 1833–1834 (2014).
Carecchio, M. et al. Novel GNAL mutation with intra-familial clinical heterogeneity: expanding the phenotype. Parkinsonism Relat. Disord. 23, 66–71 (2016).
Horisawa, S., Taira, T., Goto, S., Ochiai, T. & Nakajima, T. Long-term improvement of musician’s dystonia after stereotactic ventro-oral thalamotomy. Ann. Neurol. 74, 648–654 (2013).
Dallapiazza, R. F. et al. Noninvasive neuromodulation and thalamic mapping with low-intensity focused ultrasound. J. Neurosurg. 128, 875–884 (2017).
Allen, N. M., Lin, J. P., Lynch, T. & King, M. D. Status dystonicus: a practice guide. Dev. Med. Child Neurol. 56, 105–112 (2014).
Termsarasab, P. & Frucht, S. J. Dystonic storm: a practical clinical and video review. J. Clin. Mov. Disord. 4, 10 (2017).
Ben-Shlomo, Y., Camfield, L. & Warner, T. What are the determinants of quality of life in people with cervical dystonia? J. Neurol. Neurosurg. Psychiatry 72, 608–614 (2002).
Marsden, C. D., Marion, M. H. & Quinn, N. The treatment of severe dystonia in children and adults. J. Neurol. Neurosurg. Psychiatry 47, 1166–1173 (1984).
Fezza, J. et al. A cross-sectional structured survey of patients receiving botulinum toxin type A treatment for blepharospasm. J. Neurol. Sci. 367, 56–62 (2016).
Benninger, M. S., Gardner, G. & Grywalski, C. Outcomes of botulinum toxin treatment for patients with spasmodic dysphonia. Arch. Otolaryngol. Head Neck Surg. 127, 1083–1085 (2001).
Sheehy, M. P. & Marsden, C. D. Writers’ cramp-a focal dystonia. Brain 105, 461–480 (1982).
Conti, A. M., Pullman, S. & Frucht, S. J. The hand that has forgotten its cunning — lessons from musicians’ hand dystonia. Mov. Disord. 23, 1398–1406 (2008).
Hwang, W. J. & Tsai, C. F. Motor vehicle accidents and injuries in patients with idiopathic blepharospasm. J. Neurol. Sci. 339, 217–219 (2014).
van den Dool, J., Tijssen, M. A., Koelman, J. H., Engelbert, R. H. & Visser, B. Determinants of disability in cervical dystonia. Parkinsonism Relat. Disord. 32, 48–53 (2016).
Muller, J. et al. The impact of blepharospasm and cervical dystonia on health-related quality of life and depression. J. Neurol. 249, 842–846 (2002).
Smit, M. et al. Psychiatric co-morbidity is highly prevalent in idiopathic cervical dystonia and significantly influences health-related quality of life: results of a controlled study. Parkinsonism Relat. Disord. 30, 7–12 (2016).
Gundel, H. et al. High psychiatric comorbidity in spasmodic torticollis: a controlled study. J. Nerv. Ment. Dis. 191, 465–473 (2003).
Berman, B. D. et al. Psychiatric associations of adult-onset focal dystonia phenotypes. J. Neurol. Neurosurg. Psychiatry 88, 595–602 (2017).
Heiman, G. A. et al. Increased risk for recurrent major depression in DYT1 dystonia mutation carriers. Neurology 63, 631–637 (2004).
Zoons, E., Dijkgraaf, M. G., Dijk, J. M., van Schaik, I. N. & Tijssen, M. A. Botulinum toxin as treatment for focal dystonia: a systematic review of the pharmaco-therapeutic and pharmaco-economic value. J. Neurol. 259, 2519–2526 (2012).
Antelmi, E. et al. Modulation of the muscle activity during sleep in cervical dystonia. Sleep 40, zsx088 (2017).
Paus, S. et al. Impaired sleep quality and restless legs syndrome in idiopathic focal dystonia: a controlled study. J. Neurol. 258, 1835–1840 (2011).
Molho, E. S. et al. Impact of cervical dystonia on work productivity: an analysis from a patient registry. Mov Disord. Clin. Pract. 3, 130–138 (2016).
Molho, E. S., Agarwal, N., Regan, K., Higgins, D. S. & Factor, S. A. Effect of cervical dystonia on employment: a retrospective analysis of the ability of treatment to restore premorbid employment status. Mov. Disord. 24, 1384–1387 (2009).
Skogseid, I. M., Roislien, J., Claussen, B. & Kerty, E. Long-term botulinum toxin treatment increases employment rate in patients with cervical dystonia. Mov. Disord. 20, 1604–1609 (2005).
Martikainen, K. K., Luukkaala, T. H. & Marttila, R. J. Working capacity and cervical dystonia. Parkinsonism Relat. Disord. 16, 215–217 (2010).
Zech, M. et al. Rare sequence variants in ANO3 and GNAL in a primary torsion dystonia series and controls. Mov. Disord. 29, 143–147 (2014).
Zech, M. et al. Systematic TOR1A non-c.907_909delGAG variant analysis in isolated dystonia and controls. Parkinsonism Relat. Disord. 31, 119–123 (2016).
Kuhn, A. A. & Volkmann, J. Innovations in deep brain stimulation methodology. Mov. Disord. 32, 11–19 (2017).
Munts, A. G. & Koehler, P. J. How psychogenic is dystonia? Views from past to present. Brain 133, 1552–1564 (2010).
Marsden, C. D. The problem of adult-onset idiopathic torsion dystonia and other isolated dyskinesias in adult life (including blepharospasm, oromandibular dystonia, dystonic writer’s cramp, and torticollis, or axial dystonia). Adv. Neurol. 14, 259–276 (1976).
Robertson, M. M. et al. Gilles de la Tourette syndrome. Nat. Rev. Dis. Primers 3, 16097 (2017).
Charlesworth, G., Bhatia, K. P. & Wood, N. W. The genetics of dystonia: new twists in an old tale. Brain 136, 2017–2037 (2013).
Marras, C., Lohmann, K., Lang, A. & Klein, C. Fixing the broken system of genetic locus symbols: Parkinson disease and dystonia as examples. Neurology 78, 1016–1024 (2012).
Acknowledgements
B.B. is supported by a European Academy of Neurology fellowship and the Robert Bosch Foundation.
Reviewer information
Nature Reviews Disease Primers thanks G. Abbruzzese, H. Jinnah, R. Kaji and the other, anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
Introduction (B.B. and K.P.B.); Epidemiology (B.B., E.M.V., N.E.M. and K.P.B.); Mechanisms/pathophysiology (N.E.M., E.M.V., J.R. and A.P.); Diagnosis, screening and prevention (B.B. and K.P.B.); Management (J.J. and M.V.); Quality of life (B.B. and K.P.B.); Outlook (B.B., M.V. and K.P.B.); Overview of Primer (B.B. and K.P.B.).
Corresponding author
Ethics declarations
Competing interests
E.M.V. holds research grants from the European Research Council (ERC StG260888), Telethon Foundation Italy (GGP13146), the Italian Ministry of Health (Ricerca Finalizzata 2013 NET-2013-02356160) and the University of Pavia (BlueSky Research Grants), receives a stipend from The BMJ as Associate Editor of the Journal of Medical Genetics and has received financial support to speak and/or attend meetings from Zambon. A.P. receives grants from the Italian Ministry of Education, Universities and Research (MIUR, ref PRIN 2015, 2015FNWP34-002) and the Dystonia Medical Research Foundation and received honoraria and financial support to speak or attend meetings from AbbVie, Teva and Union Chimique Belge. M.V. has received travel grants as faculty from The International Parkinson and Movement Disorder Society and the European Academy of Neurology and holds an unrestricted research grant from Merz. J.J. has received research and/or training grants from Adamas Pharmaceuticals, Allergan, Biotie Therapies, CHDI Foundation, Civitas/Acorda Therapeutics, Dystonia Coalition, Dystonia Medical Research Foundation, F. Hoffmann-La Roche, Huntington Study Group, Kyowa Hakko Kirin Pharma, Medtronic Neuromodulation, Merz Pharmaceuticals, Michael J. Fox Foundation for Parkinson Research, National Institutes of Health, Neurocrine Biosciences, NeuroDerm, Nuvelution, Parkinson Disease Foundation, Parkinson Study Group, Pfizer, Prothena Biosciences, Psyadon Pharmaceuticals, Revance Therapeutics, Sangamo BioSciences, St. Jude Medical and Teva. J.J. has served as a consultant or as an advisory committee member for Adamas Pharmaceuticals, Allergan, Merz Pharmaceuticals, Pfizer, Prothena Biosciences, Revance Therapeutics and Teva and has received royalties or other payments from Cambridge, Elsevier, Future Science Group, Hodder Arnold, Lippincott Williams and Wilkins, MedLink, Neurology and Wiley-Blackwell. K.P.B. holds research grants from the National Institute for Health Research, Research for Patient Benefit (NIHR RfPB), a Medical Research Council Wellcome Strategic grant (WT089698) and a grant from Parkinson’s UK (G-1009) and has received honoraria and/or financial support to speak and/or attend meetings from Allergan, Boehringer-Ingelheim, GSK, Ipsen, Lundbeck, Merz, Orion, Sun Pharma and Teva. K.P.B. receives royalties from the Oxford University Press and a stipend for Movement Disorders Clinical Practice editorship. All other authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
41572_2018_23_MOESM1_ESM.mp4
Supplementary Video 1 Patient with dystonia due to TOR1A mutation. Abnormal, dystonic posturing of the left more than the right hand
41572_2018_23_MOESM2_ESM.mp4
Supplementary Video 2 Patient with cervical dystonia and geste antagoniste. Touching the side of the face, the patient can correct her torticollis
Rights and permissions
About this article
Cite this article
Balint, B., Mencacci, N.E., Valente, E.M. et al. Dystonia. Nat Rev Dis Primers 4, 25 (2018). https://doi.org/10.1038/s41572-018-0023-6
Published:
DOI: https://doi.org/10.1038/s41572-018-0023-6
This article is cited by
-
Quality of life outcomes after deep brain stimulation in acquired dystonia: a systematic review and meta-analysis
Neurological Sciences (2024)
-
Cranial geometry in patients with dystonia and Parkinson’s disease
Scientific Reports (2023)
-
Mapping the landscape of genetic dependencies in chordoma
Nature Communications (2023)
-
Rare variants in IMPDH2 cause autosomal dominant dystonia in Chinese population
Journal of Neurology (2023)
-
Mutation screening of AOPEP variants in a large dystonia cohort
Journal of Neurology (2023)