Abstract
Liver cancer, more specifically hepatocellular carcinoma (HCC), is the second leading cause of cancer-related death and its incidence is increasing globally. Around 50% of patients with HCC receive systemic therapies, traditionally sorafenib or lenvatinib in the first line and regorafenib, cabozantinib or ramucirumab in the second line. In the past 5 years, immune-checkpoint inhibitors have revolutionized the management of HCC. The combination of atezolizumab and bevacizumab has been shown to improve overall survival relative to sorafenib, resulting in FDA approval of this regimen. More recently, durvalumab plus tremelimumab yielded superior overall survival versus sorafenib and atezolizumab plus cabozantinib yielded superior progression-free survival. In addition, pembrolizumab monotherapy and the combination of nivolumab plus ipilimumab have received FDA Accelerated Approval in the second-line setting based on early efficacy data. Despite these major advances, the molecular underpinnings governing immune responses and evasion remain unclear. The immune microenvironment has crucial roles in the development and progression of HCC and distinct aetiology-dependent immune features have been defined. Inflamed and non-inflamed classes of HCC and genomic signatures have been associated with response to immune-checkpoint inhibitors, yet no validated biomarker is available to guide clinical decision-making. This Review provides information on the immune microenvironments underlying the response or resistance of HCC to immunotherapies. In addition, current evidence from phase III trials on the efficacy, immune-related adverse events and aetiology-dependent mechanisms of response are described. Finally, we discuss emerging trials assessing immunotherapies across all stages of HCC that might change the management of this disease in the near future.
Key points
-
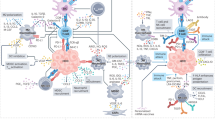
The composition of the hepatocellular carcinoma (HCC) immune microenvironment is the result of an interplay between immunosuppressive cells, immune effector cells, the cytokine milieu and tumour cell-intrinsic signalling pathways.
-
The aetiology of HCC influences the immune response and leads to unique microenvironmental features.
-
Combining anti-angiogenic drugs with immune-checkpoint inhibitors (ICIs) alters the tumour endothelium, thereby improving drug delivery and increasing the infiltration of effector immune cells.
-
Inhibiting vascular endothelial growth factor signalling also synergizes with ICIs by enhancing antitumour immune cell responses and inhibiting key immunosuppressive pathways.
-
A combination consisting of the ICI atezolizumab and the anti-angiogenic agent bevacizumab is the first treatment regimen that has been shown to improve overall survival in patients with advanced-stage HCC when compared with sorafenib.
-
Across all stages of HCC, almost 30 phase III trials testing immunotherapies (either alone or in combination) are currently ongoing and are likely to reshape the treatment landscape.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Sung, H. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Ca. Cancer J. Clin. 71, 209–249 (2021).
Llovet, J. M. et al. Hepatocellular carcinoma. Nat. Rev. Dis. Prim. 7, 6 (2021). This paper provides a comprehensive overview of the epidemiology, pathogenesis and management of HCC.
Villanueva, A. Hepatocellular Carcinoma. N. Engl. J. Med. 380, 1450–1462 (2019).
Llovet, J. M. et al. Locoregional therapies in the era of molecular and immune treatments for hepatocellular carcinoma. Nat. Rev. Gastroenterol. Hepatol. 18, 293–313 (2021).
Llovet, J. M., Montal, R., Sia, D. & Finn, R. S. Molecular therapies and precision medicine for hepatocellular carcinoma. Nat. Rev. Clin. Oncol. 15, 599–616 (2018).
Qin, S. et al. Donafenib versus sorafenib in first-line treatment of unresectable or metastatic hepatocellular carcinoma: a randomized, open-label, parallel-controlled phase II-III trial. J. Clin. Oncol. 39, 3002–3011 (2021).
Finn, R. S. et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N. Engl. J. Med. 382, 1894–1905 (2020). This trial is the first to demonstrate the superiority of an immunotherapy combination, atezolizumab–bevacizumab, over frontline sorafenib for advanced-stage HCC.
Ren, Z. et al. Sintilimab plus a bevacizumab biosimilar (IBI305) versus sorafenib in unresectable hepatocellular carcinoma (ORIENT-32): a randomised, open-label, phase 2–3 study. Lancet Oncol. 22, 977–990 (2021).
Bruni, D., Angell, H. K. & Galon, J. The immune contexture and Immunoscore in cancer prognosis and therapeutic efficacy. Nat. Rev. Cancer 20, 662–680 (2020).
Kalbasi, A. & Ribas, A. Tumour-intrinsic resistance to immune checkpoint blockade. Nat. Rev. Immunol. 20, 25–39 (2020).
Huinen, Z. R., Huijbers, E. J. M., van Beijnum, J. R., Nowak-Sliwinska, P. & Griffioen, A. W. Anti-angiogenic agents — overcoming tumour endothelial cell anergy and improving immunotherapy outcomes. Nat. Rev. Clin. Oncol. 18, 527–540 (2021). This Review discusses the main mechanisms supporting the synergism of anti-angiogenic drugs and immune-checkpoint inhibitors.
Havel, J. J., Chowell, D. & Chan, T. A. The evolving landscape of biomarkers for checkpoint inhibitor immunotherapy. Nat. Rev. Cancer 19, 133–150 (2019).
Zhu, A. X. et al. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): a non-randomised, open-label phase 2 trial. Lancet Oncol. 19, 940–952 (2018).
Yau, T. et al. Efficacy and safety of nivolumab plus ipilimumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib. JAMA Oncol. 6, e204564 (2020). This study was the first to provide evidence of the efficacy of combining two immune-checkpoint inhibitors targeting CTLA4 (ipilimumab) and PD-1 (nivolumab) in patients with HCC.
Ringelhan, M., Pfister, D., O’Connor, T., Pikarsky, E. & Heikenwalder, M. The immunology of hepatocellular carcinoma. Nat. Immunol. 19, 222–232 (2018).
Hou, J., Zhang, H., Sun, B. & Karin, M. The immunobiology of hepatocellular carcinoma in humans and mice: basic concepts and therapeutic implications. J. Hepatol. 72, 167–182 (2020).
Sia, D. et al. Identification of an immune-specific class of hepatocellular carcinoma, based on molecular features. Gastroenterology 153, 812–826 (2017). This study was the first to identify an immune-specific class of HCC with unique biological traits and potential implications for immunotherapy.
Sangro, B., Sarobe, P., Hervás-Stubbs, S. & Melero, I. Advances in immunotherapy for hepatocellular carcinoma. Nat. Rev. Gastroenterol. Hepatol. 2, 1–19 (2021).
Llovet, J. M. et al. Trial design and endpoints in hepatocellular carcinoma: AASLD consensus conference. Hepatology 73, 158–191 (2021).
Heymann, F. et al. Liver inflammation abrogates immunological tolerance induced by Kupffer cells. Hepatology 62, 279–291 (2015).
Cancer Genome Atlas Research Network.Comprehensive and integrative genomic characterization of hepatocellular carcinoma. Cell 169, 1327–1341.e23 (2017).
Zheng, C. et al. Landscape of Infiltrating T cells in liver cancer revealed by single-cell sequencing. Cell 169, 1342–1356.e16 (2017). This study was one of the first in-depth characterizations of the immune microenvironment of HCC at single-cell resolution.
Zhang, Q. et al. Landscape and dynamics of single immune cells in hepatocellular carcinoma. Cell 179, 829–845.e20 (2019).
Pfister, D. et al. NASH limits anti-tumour surveillance in immunotherapy-treated HCC. Nature 592, 450–456 (2021). This study demonstrates the existence of a CD8+PD-1+ subset of protumorigenic cells in non-alcoholic steatohepatitis that favour the development of HCC and hamper response to immune-checkpoint inhibitors.
Hoechst, B. et al. A new population of myeloid-derived suppressor cells in hepatocellular carcinoma patients induces CD4+CD25+Foxp3+ T cells. Gastroenterology 135, 234–243 (2008).
Kalathil, S., Lugade, A. A., Miller, A., Iyer, R. & Thanavala, Y. Higher frequencies of GARP+CTLA-4+Foxp3+ T regulatory cells and myeloid-derived suppressor cells in hepatocellular carcinoma patients are associated with impaired T-cell functionality. Cancer Res. 73, 2435–2444 (2013).
Han, Y. et al. Human CD14+CTLA-4+ regulatory dendritic cells suppress T-cell response by cytotoxic T-lymphocyte antigen-4-dependent IL-10 and indoleamine-2,3-dioxygenase production in hepatocellular carcinoma. Hepatology 59, 567–579 (2014).
Finkin, S. et al. Ectopic lymphoid structures function as microniches for tumor progenitor cells in hepatocellular carcinoma. Nat. Immunol. 16, 1235–1244 (2015).
Calderaro, J. et al. Intra-tumoral tertiary lymphoid structures are associated with a low risk of early recurrence of hepatocellular carcinoma. J. Hepatol. 70, 58–65 (2019).
Bruno, T. C. B cells to the forefront of immunotherapy. Nature 577, 474–476 (2020).
Dong, L. et al. Heterogeneous immunogenomic features and distinct escape mechanisms in multifocal hepatocellular carcinoma. J. Hepatol. 72, 896–908 (2020).
Jhunjhunwala, S., Hammer, C. & Delamarre, L. Antigen presentation in cancer: insights into tumour immunogenicity and immune evasion. Nat. Rev. Cancer 21, 298–312 (2021).
Amsen, D., van Gisbergen, K. P. J. M., Hombrink, P. & van Lier, R. A. W. Tissue-resident memory T cells at the center of immunity to solid tumors. Nat. Immunol. 19, 538–546 (2018).
Pallett, L. J. et al. Longevity and replenishment of human liver-resident memory T cells and mononuclear phagocytes. J. Exp. Med. 217, e20200050 (2020).
Tauber, C. et al. Inefficient induction of circulating TAA-specific CD8+ T-cell responses in hepatocellular carcinoma. Oncotarget 10, 5194–5206 (2019).
Payne, K. K. et al. BTN3A1 governs antitumor responses by coordinating αβ and γδ T cells. Science 369, 942–949 (2020).
Sun, Y. et al. Single-cell landscape of the ecosystem in early-relapse hepatocellular carcinoma. Cell 184, 404–421.e16 (2021).
Song, G. et al. Global immune characterization of HBV/HCV-related hepatocellular carcinoma identifies macrophage and T-cell subsets associated with disease progression. Cell Discov. 6, 90 (2020).
Zecca, A. et al. Energy metabolism and cell motility defect in NK-cells from patients with hepatocellular carcinoma. Cancer Immunol. Immunother. 69, 1589–1603 (2020).
Flecken, T. et al. Immunodominance and functional alterations of tumor-associated antigen-specific CD8+ T-cell responses in hepatocellular carcinoma. Hepatology 59, 1415–1426 (2014).
Keenan, T. E., Burke, K. P. & Van Allen, E. M. Genomic correlates of response to immune checkpoint blockade. Nat. Med. 25, 389–402 (2019).
Craig, A. J. et al. Transcriptomic characterization of cancer-testis antigens identifies MAGEA3 as a driver of tumor progression in hepatocellular carcinoma. PLoS Genet. 17, e1009589 (2021).
Michler, T. et al. Knockdown of virus antigen expression increases therapeutic vaccine efficacy in high-titer hepatitis B virus carrier mice. Gastroenterology 158, 1762–1775.e9 (2020).
Liu, D. et al. Integrative molecular and clinical modeling of clinical outcomes to PD1 blockade in patients with metastatic melanoma. Nat. Med. 25, 1916–1927 (2019).
Samstein, R. M. et al. Tumor mutational load predicts survival after immunotherapy across multiple cancer types. Nat. Genet. 51, 202–206 (2019).
Rosenthal, R. et al. Neoantigen-directed immune escape in lung cancer evolution. Nature 567, 479–485 (2019).
Haber, P. K. et al. Molecular markers of response to anti-PD1 therapy in advanced hepatocellular carcinoma. J. Clin. Oncol. 39, 4110 (2021).
Davoli, T., Uno, H., Wooten, E. C. & Elledge, S. J. Tumor aneuploidy correlates with markers of immune evasion and with reduced response to immunotherapy. Sci 355, eaaf8399 (2017).
Montironi, C. et al. Immune Classification of Hepatocellular Carcinoma Based on New Molecular Features: The Inflamed Class (EASL, 2020).
Bassaganyas, L. et al. Copy-number alteration burden differentially impacts immune profiles and molecular features of hepatocellular carcinoma. Clin. Cancer Res. 26, 6350–6361 (2020).
Zhou, S.-L. et al. Overexpression of CXCL5 mediates neutrophil infiltration and indicates poor prognosis for hepatocellular carcinoma. Hepatology 56, 2242–2254 (2012).
Liu, L.-Z. et al. CCL15 recruits suppressive monocytes to facilitate immune escape and disease progression in hepatocellular carcinoma. Hepatology 69, 143–159 (2019).
Shalapour, S. et al. Inflammation-induced IgA+ cells dismantle anti-liver cancer immunity. Nature 551, 340–345 (2017).
Sangro, B. et al. Association of inflammatory biomarkers with clinical outcomes in nivolumab-treated patients with advanced hepatocellular carcinoma. J. Hepatol. 73, 1460–1469 (2020).
Litchfield, K. et al. Meta-analysis of tumor- and T cell-intrinsic mechanisms of sensitization to checkpoint inhibition. Cell 184, 596–614.e14 (2021).
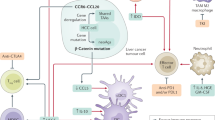
Ruiz de Galarreta, M. et al. β-Catenin activation promotes immune escape and resistance to anti-PD-1 therapy in hepatocellular carcinoma. Cancer Discov. 9, 1124–1141 (2019). This study demonstrates that β-catenin activation leads to immune evasion by interfering with dendritic cell chemotaxis.
Cadoux, M. et al. Expression of NKG2D ligands is downregulated by β-catenin signalling and associates with HCC aggressiveness. J. Hepatol. 74, 1386–1397 (2021).
Moeini, A. et al. An immune gene expression signature associated with development of human hepatocellular carcinoma identifies mice that respond to chemopreventive agents. Gastroenterology 157, 1383–1397.e11 (2019).
Xu, Y. et al. Translation control of the immune checkpoint in cancer and its therapeutic targeting. Nat. Med. 25, 301–311 (2019).
Shen, J. et al. ARID1A deficiency promotes mutability and potentiates therapeutic antitumor immunity unleashed by immune checkpoint blockade. Nat. Med. 24, 556–562 (2018).
Li, J. et al. Epigenetic driver mutations in ARID1A shape cancer immune phenotype and immunotherapy. J. Clin. Invest. 130, 2712–2726 (2020).
Zhou, J. et al. Hepatoma-intrinsic CCRK inhibition diminishes myeloid-derived suppressor cell immunosuppression and enhances immune-checkpoint blockade efficacy. Gut 67, 931–944 (2018).
Chiang, D. Y. et al. Focal Gains of VEGFA and molecular classification of hepatocellular carcinoma. Cancer Res. 68, 6779–6788 (2008).
Maini, M. K. & Pallett, L. J. Defective T-cell immunity in hepatitis B virus infection: why therapeutic vaccination needs a helping hand. Lancet Gastroenterol. Hepatol. 3, 192–202 (2018).
Kassel, R. et al. Chronically inflamed livers up-regulate expression of inhibitory B7 family members. Hepatology 50, 1625–1637 (2009).
Wieland, D. et al. TCF1+ hepatitis C virus-specific CD8+ T cells are maintained after cessation of chronic antigen stimulation. Nat. Commun. 8, 15050 (2017).
Dudek, M. et al. Auto-aggressive CXCR6+CD8 T cells cause liver immune pathology in NASH. Nature 592, 444–449 (2021).
Wolf, M. J. et al. Metabolic activation of intrahepatic CD8+ T cells and NKT cells causes nonalcoholic steatohepatitis and liver cancer via cross-talk with hepatocytes. Cancer Cell 26, 549–564 (2014).
Mason, W. S. et al. HBV DNA integration and clonal hepatocyte expansion in chronic hepatitis B patients considered immune tolerant. Gastroenterology 151, 986–998.e4 (2016).
Goto, K., Roca Suarez, A. A., Wrensch, F., Baumert, T. F. & Lupberger, J. Hepatitis C virus and hepatocellular carcinoma: when the host loses its grip. Int. J. Mol. Sci. 21, 3057 (2020).
Simon, T. G. et al. Association of aspirin with hepatocellular carcinoma and liver-related mortality. N. Engl. J. Med. 382, 1018–1028 (2020).
Sitia, G. et al. Antiplatelet therapy prevents hepatocellular carcinoma and improves survival in a mouse model of chronic hepatitis B. Proc. Natl Acad. Sci. USA 109, E2165–E2172 (2012).
Tan, A. T. et al. Use of expression profiles of HBV-DNA integrated into genomes of hepatocellular carcinoma cells to select T cells for immunotherapy. Gastroenterology 156, 1862–1876.e9 (2019).
Dunn, C. et al. Temporal analysis of early immune responses in patients with acute hepatitis B virus infection. Gastroenterology 137, 1289–1300 (2009).
Das, A. et al. IL-10–producing regulatory B cells in the pathogenesis of chronic hepatitis B virus infection. J. Immunol. 189, 3925–3935 (2012).
Xue, H. et al. Overrepresentation of IL-10-expressing B cells suppresses cytotoxic CD4+ T cell activity in HBV-induced hepatocellular carcinoma. PLoS ONE 11, e0154815 (2016).
Lim, C. J. et al. Multidimensional analyses reveal distinct immune microenvironment in hepatitis B virus-related hepatocellular carcinoma. Gut 68, 916–927 (2019).
Heim, M. H. & Thimme, R. Innate and adaptive immune responses in HCV infections. J. Hepatol. 61, S14–S25 (2014).
Semmo, N. et al. Preferential loss of IL-2-secreting CD4+ T helper cells in chronic HCV infection. Hepatology 41, 1019–1028 (2005).
Cabrera, R. et al. An immunomodulatory role for CD4+CD25+ regulatory T lymphocytes in hepatitis C virus infection. Hepatology 40, 1062–1071 (2004).
Abel, M. et al. Intrahepatic virus-specific IL-10-producing CD8 T cells prevent liver damage during chronic hepatitis C virus infection. Hepatology 44, 1607–1616 (2006).
Hirano, J. et al. Hepatitis C virus modulates signal peptide peptidase to alter host protein processing. Proc. Natl Acad. Sci. USA 118, e2026184118 (2021).
Pallett, L. J. et al. Metabolic regulation of hepatitis B immunopathology by myeloid-derived suppressor cells. Nat. Med. 21, 591–600 (2015).
Jiang, Y. et al. Proteomics identifies new therapeutic targets of early-stage hepatocellular carcinoma. Nature 567, 257–261 (2019).
Schmidt, N. M. et al. Targeting human Acyl-CoA:cholesterol acyltransferase as a dual viral and T cell metabolic checkpoint. Nat. Commun. 12, 2814 (2021).
Pikarsky, E. & Heikenwalder, M. Focal and local: ectopic lymphoid structures and aggregates of myeloid and other immune cells in liver. Gastroenterology 151, 780–783 (2016).
Anstee, Q. M., Reeves, H. L., Kotsiliti, E., Govaere, O. & Heikenwalder, M. From NASH to HCC: current concepts and future challenges. Nat. Rev. Gastroenterol. Hepatol. 16, 411–428 (2019).
Malehmir, M. et al. Platelet GPIbα is a mediator and potential interventional target for NASH and subsequent liver cancer. Nat. Med. 25, 641–655 (2019).
Simon, T. G. et al. Association between aspirin use and risk of hepatocellular carcinoma. JAMA Oncol. 4, 1683 (2018).
Gomes, A. L. et al. Metabolic inflammation-associated IL-17A causes non-alcoholic steatohepatitis and hepatocellular carcinoma. Cancer Cell 30, 161–175 (2016).
Ma, C. et al. NAFLD causes selective CD4+ T lymphocyte loss and promotes hepatocarcinogenesis. Nature 531, 253–257 (2016). This study shows that non-alcoholic fatty liver disease causes dysregulation of lipid metabolism, which leads to CD4+ T cell depletion and promotes hepatocarcinogenesis.
Ganne-Carrié, N. & Nahon, P. Hepatocellular carcinoma in the setting of alcohol-related liver disease. J. Hepatol. 70, 284–293 (2019).
Parlesak, A., Schäfer, C., Schütz, T., Bode, J. C. & Bode, C. Increased intestinal permeability to macromolecules and endotoxemia in patients with chronic alcohol abuse in different stages of alcohol-induced liver disease. J. Hepatol. 32, 742–747 (2000).
Gao, M. et al. Granulocytic myeloid-derived suppressor cell population increases with the severity of alcoholic liver disease. J. Cell. Mol. Med. 23, 2032–2041 (2019).
Yan, G. et al. Chronic alcohol consumption promotes diethylnitrosamine-induced hepatocarcinogenesis via immune disturbances. Sci. Rep. 7, 2567 (2017).
Gao, B. & Bataller, R. Alcoholic liver disease: pathogenesis and new therapeutic targets. Gastroenterology 141, 1572–1585 (2011).
Tilg, H., Moschen, A. R. & Szabo, G. Interleukin-1 and inflammasomes in alcoholic liver disease/acute alcoholic hepatitis and nonalcoholic fatty liver disease/nonalcoholic steatohepatitis. Hepatology 64, 955–965 (2016).
Ma, H.-Y. et al. IL-17 signaling in steatotic hepatocytes and macrophages promotes hepatocellular carcinoma in alcohol-related liver disease. J. Hepatol. 72, 946–959 (2020).
Ponziani, F. R. et al. Hepatocellular carcinoma is associated with gut microbiota profile and inflammation in nonalcoholic fatty liver disease. Hepatology 69, 107–120 (2019).
Schwabe, R. F. & Greten, T. F. Gut microbiome in HCC – mechanisms, diagnosis and therapy. J. Hepatol. 72, 230–238 (2020).
Dapito, D. H. et al. Promotion of hepatocellular carcinoma by the intestinal microbiota and TLR4. Cancer Cell 21, 504–516 (2012).
Ma, C. et al. Gut microbiome-mediated bile acid metabolism regulates liver cancer via NKT cells. Sci 360, eaan5931 (2018).
Yoshimoto, S. et al. Obesity-induced gut microbial metabolite promotes liver cancer through senescence secretome. Nature 499, 97–101 (2013).
Thorsson, V. V. et al. The immune landscape of cancer. Immunity 48, 812–830.e14 (2018).
Montal, R. et al. Molecular classification and therapeutic targets in extrahepatic cholangiocarcinoma. J. Hepatol. 73, 315–327 (2020).
Job, S. et al. Identification of four immune subtypes characterized by distinct composition and functions of tumor microenvironment in intrahepatic cholangiocarcinoma. Hepatology 72, 965–981 (2020).
Gay, C. M. et al. Patterns of transcription factor programs and immune pathway activation define four major subtypes of SCLC with distinct therapeutic vulnerabilities. Cancer Cell 39, 346–360.e7 (2021).
Nsengimana, J. et al. β-Catenin–mediated immune evasion pathway frequently operates in primary cutaneous melanomas. J. Clin. Invest. 128, 2048–2063 (2018).
Chen, Y.-P. et al. Identification and validation of novel microenvironment-based immune molecular subgroups of head and neck squamous cell carcinoma: implications for immunotherapy. Ann. Oncol. 30, 68–75 (2019).
Wei, S. C., Duffy, C. R. & Allison, J. P. Fundamental mechanisms of immune checkpoint blockade therapy. Cancer Discov. 8, 1069–1086 (2018).
Yokosuka, T. et al. Programmed cell death 1 forms negative costimulatory microclusters that directly inhibit T cell receptor signaling by recruiting phosphatase SHP2. J. Exp. Med. 209, 1201–1217 (2012).
Ahn, E. et al. Role of PD-1 during effector CD8 T cell differentiation. Proc. Natl Acad. Sci. USA 115, 4749–4754 (2018).
Maker, A. V., Attia, P. & Rosenberg, S. A. Analysis of the cellular mechanism of antitumor responses and autoimmunity in patients treated with CTLA-4 blockade. J. Immunol. 175, 7746–7754 (2005).
Peggs, K. S., Quezada, S. A., Chambers, C. A., Korman, A. J. & Allison, J. P. Blockade of CTLA-4 on both effector and regulatory T cell compartments contributes to the antitumor activity of anti-CTLA-4 antibodies. J. Exp. Med. 206, 1717–1725 (2009).
Agdashian, D. et al. The effect of anti-CTLA4 treatment on peripheral and intra-tumoral T cells in patients with hepatocellular carcinoma. Cancer Immunol. Immunother. 68, 599–608 (2019).
Calderaro, J. et al. Programmed death ligand 1 expression in hepatocellular carcinoma: relationship with clinical and pathological features. Hepatology 64, 2038–2046 (2016).
Ribas, A. & Wolchok, J. D. Cancer immunotherapy using checkpoint blockade. Science 359, 1350–1355 (2018).
Duan, J. et al. Use of immunotherapy with programmed cell death 1 vs programmed cell death ligand 1 inhibitors in patients with cancer. JAMA Oncol. 6, 375 (2020).
Kurino, T. et al. Poor outcome with anti-programmed death-ligand 1 (PD-L1) antibody due to poor pharmacokinetic properties in PD-1/PD-L1 blockade-sensitive mouse models. J. Immunother. Cancer 8, e000400 (2020).
Yearley, J. H. et al. PD-L2 expression in human tumors: relevance to Anti-PD-1 therapy in cancer. Clin. Cancer Res. 23, 3158–3167 (2017).
Butterfield, L. H. Immunotherapeutic strategies for hepatocellular carcinoma. Gastroenterology 127, S232–S241 (2004).
Palmer, D. H. et al. A phase II study of adoptive immunotherapy using dendritic cells pulsed with tumor lysate in patients with hepatocellular carcinoma. Hepatology 49, 124–132 (2009).
Meng, W. S. et al.α-Fetoprotein-specific tumor immunity induced by plasmid prime-adenovirus boost genetic vaccination. Cancer Res. 61, 8782–8786 (2001).
Butterfield, L. H., Ribas, A., Potter, D. M. & Economou, J. S. Spontaneous and vaccine induced AFP-specific T cell phenotypes in subjects with AFP-positive hepatocellular cancer. Cancer Immunol. Immunother. 56, 1931–1943 (2007).
Greten, T. F. et al. A phase II open label trial evaluating safety and efficacy of a telomerase peptide vaccination in patients with advanced hepatocellular carcinoma. BMC Cancer 10, 209 (2010).
Goyal, L. et al. Abstract 3183: Initial safety of AFP SPEAR T-cells in patients with advanced hepatocellular carcinoma. Cancer Res. https://doi.org/10.1158/1538-7445.AM2019-3183 (2019).
Dai, H. et al. Efficacy and biomarker analysis of CD133-directed CAR T cells in advanced hepatocellular carcinoma: a single-arm, open-label, phase II trial. Oncoimmunology 9, 1846926 (2020).
Shi, D. et al. Chimeric antigen receptor-glypican-3 T-cell therapy for advanced hepatocellular carcinoma: results of phase I trials. Clin. Cancer Res. 26, 3979–3989 (2020).
Finn, R. S. et al. Phase Ib study of lenvatinib plus pembrolizumab in patients with unresectable hepatocellular carcinoma. J. Clin. Oncol. 38, 2960–2970 (2020).
Xu, J. et al. Camrelizumab in combination with apatinib in patients with advanced hepatocellular carcinoma (RESCUE): a nonrandomized, open-label, phase II trial. Clin. Cancer Res. 27, 1003–1011 (2021).
Fukumura, D., Kloepper, J., Amoozgar, Z., Duda, D. G. & Jain, R. K. Enhancing cancer immunotherapy using antiangiogenics: opportunities and challenges. Nat. Rev. Clin. Oncol. 15, 325–340 (2018).
Torrens, L. et al. Immunomodulatory effects of lenvatinib plus anti‐PD1 in mice and rationale for patient enrichment in hepatocellular carcinoma. Hepatology https://doi.org/10.1002/hep.32023 (2021).
Esteban-Fabró, R. et al. Cabozantinib enhances the efficacy and immune modulatory activity of anti-PD1 therapy in a syngeneic mouse model of hepatocellular carcinoma. J. Hepatol. 73, S40 (2020).
Sprinzl, M. F. et al. Sorafenib inhibits macrophage-induced growth of hepatoma cells by interference with insulin-like growth factor-1 secretion. J. Hepatol. 62, 863–870 (2015).
Shigeta, K. et al. Regorafenib combined with PD1 blockade increases CD8 T-cell infiltration by inducing CXCL10 expression in hepatocellular carcinoma. J. Immunother. Cancer 8, e001435 (2020).
Zhuang, P.-Y. et al. Prognostic roles of cross-talk between peritumoral hepatocytes and stromal cells in hepatocellular carcinoma involving peritumoral VEGF-C, VEGFR-1 and VEGFR-3. PLoS ONE 8, e64598 (2013).
Ma, L. et al. Tumor cell biodiversity drives microenvironmental reprogramming in liver cancer. Cancer Cell 36, 418–430.e6 (2019).
Horwitz, E. et al. Human and Mouse VEGFA -amplified hepatocellular carcinomas are highly sensitive to sorafenib treatment. Cancer Discov. 4, 730–743 (2014).
Zerbini, A. et al. Radiofrequency thermal ablation for hepatocellular carcinoma stimulates autologous NK-cell response. Gastroenterology 138, 1931–1942.e2 (2010).
Lee, M. S. et al. Atezolizumab with or without bevacizumab in unresectable hepatocellular carcinoma (GO30140): an open-label, multicentre, phase 1b study. Lancet Oncol. 21, 808–820 (2020).
Qin, S. et al. Camrelizumab in patients with previously treated advanced hepatocellular carcinoma: a multicentre, open-label, parallel-group, randomised, phase 2 trial. Lancet Oncol. 21, 571–580 (2020).
Kelley, R. K. et al. Safety, efficacy, and pharmacodynamics of tremelimumab plus durvalumab for patients with unresectable hepatocellular carcinoma: randomized expansion of a phase I/II study. J. Clin. Oncol. 39, 2991–3001 (2021).
Yen, C.-J. et al. Preliminary results of a phase 1A/1B study of BGB-A317, an anti-PD-1 monoclonal antibody (mAb), in patients with advanced hepatocellular carcinoma (HCC). Ann. Oncol. 28, iii54 (2017).
Ducreux, M. et al. O-1 Results from a global phase 2 study of tislelizumab, an investigational PD-1 antibody, in patients with unresectable hepatocellular carcinoma. Ann. Oncol. 32, S217 (2021).
Huang, A. C. et al. T-cell invigoration to tumour burden ratio associated with anti-PD-1 response. Nature 545, 60–65 (2017).
Topalian, S. L. et al. Five-year survival and correlates among patients with advanced melanoma, renal cell carcinoma, or non-small cell lung cancer treated with nivolumab. JAMA Oncol. 5, 1411–1420 (2019).
Takayama, T. et al. Adoptive immunotherapy to lower postsurgical recurrence rates of hepatocellular carcinoma: a randomised trial. Lancet 356, 802–807 (2000).
Lee, J. H. et al. Adjuvant immunotherapy with autologous cytokine-induced killer cells for hepatocellular carcinoma. Gastroenterology 148, 1383–1391.e6 (2015).
European Association for the Study of the Liver. EASL clinical practice guidelines: management of hepatocellular carcinoma. J. Hepatol. 69, 182–236 (2018).
Heimbach, J. K. et al. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology 67, 358–380 (2018).
Haber, P. K. et al. Evidence-based management of HCC: systematic review and meta-analysis of randomized controlled trials (2002-2020). Gastroenterology 161, 879–898 (2021).
Topalian, S. L., Taube, J. M. & Pardoll, D. M. Neoadjuvant checkpoint blockade for cancer immunotherapy. Science 367, eaax0182 (2020).
Liu, J. et al. Improved efficacy of neoadjuvant compared to adjuvant immunotherapy to eradicate metastatic disease. Cancer Discov. 6, 1382–1399 (2016).
Pinato, D. J., Fessas, P., Sapisochin, G. & Marron, T. U. Perspectives on the neoadjuvant use of immunotherapy in hepatocellular carcinoma. Hepatology 74, 483–490 (2021).
Kaseb, A. O. et al. Immunologic correlates of pathologic complete response to preoperative immunotherapy in hepatocellular carcinoma. Cancer Immunol. Res. 7, 1390–1395 (2019).
Marron, T. U. et al. Abstract CT182: Neoadjuvant cemiplimab demonstrates complete pathological responses in hepatocellular carcinoma. Cancer Res. https://doi.org/10.1158/1538-7445.AM2021-CT182 (2021).
Ho, W. J. et al. Neoadjuvant cabozantinib and nivolumab convert locally advanced hepatocellular carcinoma into resectable disease with enhanced antitumor immunity. Nat. Cancer 2, 891–903 (2021).
Greten, T. F., Mauda-Havakuk, M., Heinrich, B., Korangy, F. & Wood, B. J. Combined locoregional-immunotherapy for liver cancer. J. Hepatol. 70, 999–1007 (2019).
Duffy, A. G. et al. Tremelimumab in combination with ablation in patients with advanced hepatocellular carcinoma. J. Hepatol. 66, 545–551 (2017).
Sangro, B. et al. A clinical trial of CTLA-4 blockade with tremelimumab in patients with hepatocellular carcinoma and chronic hepatitis C. J. Hepatol. 59, 81–88 (2013).
El-Khoueiry, A. B. et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet 389, 2492–2502 (2017).
El-Khoueiry, A. B. et al. Impact of antitumor activity on survival outcomes, and nonconventional benefit, with nivolumab (NIVO) in patients with advanced hepatocellular carcinoma (aHCC): subanalyses of CheckMate-040. J. Clin. Oncol. 36, 475 (2018).
Yau, T. et al. CheckMate 459: a randomized, multi-center phase III study of nivolumab (NIVO) vs sorafenib (SOR) as first-line (1L) treatment in patients (pts) with advanced hepatocellular carcinoma (aHCC). Ann. Oncol. 30, v874–v875 (2019).
Finn, R. S. et al. Pembrolizumab as second-line therapy in patients with advanced hepatocellular carcinoma in KEYNOTE-240: a randomized, double-blind, phase III trial. J. Clin. Oncol. 38, 193–202 (2020). This study was the first phase III trial assessing the efficacy of pembrolizumab, an anti-PD-1 antibody, in the second-line treatment of advanced-stage HCC.
Bristol Myers Squibb Statement. Bristol Myers Squibb Statement on Opdivo® (nivolumab) Monotherapy Post-Sorafenib Hepatocellular Carcinoma U.S. Indication. https://news.bms.com/news/corporate-financial/2021/Bristol-Myers-Squibb-Statement-on-Opdivo-nivolumab-Monotherapy-Post-Sorafenib-Hepatocellular-Carcinoma-U.S.-Indication/default.aspx (2021).
Merck. Merck Announces KEYTRUDA® (pembrolizumab) Met Primary Endpoint of Overall Survival (OS) in Patients with Advanced Hepatocellular Carcinoma Previously Treated with Sorafenib. Merck.com https://www.merck.com/news/merck-announces-keytruda-pembrolizumab-met-primary-endpoint-of-overall-survival-os-in-patients-with-advanced-hepatocellular-carcinoma-previously-treated-with-sorafenib/ (27 September 2021).
Sangro, B. et al. LBA-3 CheckMate 459: long-term (minimum follow-up 33.6 months) survival outcomes with nivolumab versus sorafenib as first-line treatment in patients with advanced hepatocellular carcinoma. Ann. Oncol. 31, S241–S242 (2020).
Finn, R. S. et al. IMbrave150: updated overall survival (OS) data from a global, randomized, open-label phase III study of atezolizumab (atezo) + bevacizumab (bev) versus sorafenib (sor) in patients (pts) with unresectable hepatocellular carcinoma (HCC). J. Clin. Oncol. 39, 267 (2021).
Exelixis. Exelixis and Ipsen Announce Cabozantinib in Combination with an Immune Checkpoint Inhibitor Significantly Improved Progression-Free Survival in Phase 3 COSMIC-312 Pivotal Trial in Patients with Previously Untreated Advanced Liver Cancer. https://ir.exelixis.com/news-releases/news-release-details/exelixis-and-ipsen-announce-cabozantinib-combination-immune (2021).
Llovet, J. M., Montal, R. & Villanueva, A. Randomized trials and endpoints in advanced HCC: role of PFS as a surrogate of survival. J. Hepatol. 70, 1262–1277 (2019).
Hu, J. et al. Image-guided percutaneous microwave ablation versus cryoablation for hepatocellular carcinoma in high-risk locations: intermediate-term results. Cancer Manag. Res. 11, 9801–9811 (2019).
Yu, J. et al. Liver metastasis restrains immunotherapy efficacy via macrophage-mediated T cell elimination. Nat. Med. 27, 152–164 (2021).
Ducreux, M. et al. IMbrave150: exploratory analysis to examine the association between treatment response and overall survival (OS) in patients (pts) with unresectable hepatocellular carcinoma (HCC) treated with atezolizumab (atezo) + bevacizumab (bev) versus sorafenib (sor). J. Clin. Oncol. 39 (Suppl. 15), 4071 (2021).
Enrico, D., Paci, A., Chaput, N., Karamouza, E. & Besse, B. Antidrug antibodies against immune checkpoint blockers: impairment of drug efficacy or indication of immune activation? Clin. Cancer Res. 26, 787–792 (2020).
FDA. Highlights of Prescribing Information: TECENTRIQ. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/761034s025lbl.pdf (2020).
Pinato, D. J. et al. Clinical implications of heterogeneity in PD-L1 immunohistochemical detection in hepatocellular carcinoma: the Blueprint-HCC study. Br. J. Cancer 120, 1033–1036 (2019).
Hause, R. J., Pritchard, C. C., Shendure, J. & Salipante, S. J. Classification and characterization of microsatellite instability across 18 cancer types. Nat. Med. 22, 1342–1350 (2016).
Ang, C. et al. Prevalence of established and emerging biomarkers of immune checkpoint inhibitor response in advanced hepatocellular carcinoma. Oncotarget 10, 4018–4025 (2019).
Wong, C. N. et al. Qualification of tumour mutational burden by targeted next‐generation sequencing as a biomarker in hepatocellular carcinoma. Liver Int. 41, 192–203 (2021).
Harding, J. J. et al. Prospective genotyping of hepatocellular carcinoma: clinical implications of next-generation sequencing for matching patients to targeted and immune therapies. Clin. Cancer Res. 25, 2116–2126 (2019).
von Felden, J. et al. Mutations in circulating tumor DNA predict primary resistance to systemic therapies in advanced hepatocellular carcinoma. Oncogene 40, 140–151 (2021).
Zheng, Y. et al. Gut microbiome affects the response to anti-PD-1 immunotherapy in patients with hepatocellular carcinoma. J. Immunother. Cancer 7, 193 (2019).
Fessas, P. et al. Immunotoxicity from checkpoint inhibitor therapy: clinical features and underlying mechanisms. Immunology 159, 167–177 (2020).
Puzanov, I. et al. Managing toxicities associated with immune checkpoint inhibitors: consensus recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J. Immunother. Cancer 5, 95 (2017).
Nogueira, E., Newsom-Davis, T. & Morganstein, D. L. Immunotherapy-induced endocrinopathies: assessment, management and monitoring. Ther. Adv. Endocrinol. Metab. 10, 204201881989618 (2019).
Brahmer, J. R. et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: american society of clinical oncology clinical practice guideline. J. Clin. Oncol. 36, 1714–1768 (2018). These guidelines provide detailed recommendations on the management of the most common immune-related adverse events.
Pinato, D. J. et al. Treatment-related toxicity and improved outcome from immunotherapy in hepatocellular cancer: evidence from an FDA pooled analysis of landmark clinical trials with validation from routine practice. Eur. J. Cancer 157, 140–152 (2021).
Dolladille, C. et al. Immune checkpoint inhibitor rechallenge after immune-related adverse events in patients with cancer. JAMA Oncol. 6, 865 (2020).
Pinato, D. J. et al. Impact of corticosteroid therapy on the outcomes of hepatocellular carcinoma treated with immune checkpoint inhibitor therapy. J. Immunother. Cancer 8, e000726 (2020).
Gudd, C. L. C. et al. Activation and transcriptional profile of monocytes and CD8+ T cells are altered in checkpoint inhibitor-related hepatitis. J. Hepatol. 75, 177–189 (2021).
Fessas, P. et al. Post-registration experience of nivolumab in advanced hepatocellular carcinoma: an international study. J. Immunother. Cancer 8, e001033 (2020).
Pinato, D. J. et al. Immunotherapy in hepatocellular cancer patients with mild to severe liver dysfunction: adjunctive role of the ALBI Grade. Cancers 12, 1862 (2020).
Sangro, B. et al. Diagnosis and management of toxicities of immune checkpoint inhibitors in hepatocellular carcinoma. J. Hepatol. 72, 320–341 (2020).
d’Izarny‐Gargas, T., Durrbach, A. & Zaidan, M. Efficacy and tolerance of immune checkpoint inhibitors in transplant patients with cancer: a systematic review. Am. J. Transplant. 20, 2457–2465 (2020).
Gordan, J. D. et al. Systemic therapy for advanced hepatocellular carcinoma: ASCO guideline. J. Clin. Oncol. 38, 4317–4345 (2020).
Bruix, J., Chan, S. L., Galle, P. R., Rimassa, L. & Sangro, B. Systemic treatment of hepatocellular carcinoma: An EASL position paper. J. Hepatol. 75, 960–974 (2021).
Vogel, A. et al. Updated treatment recommendations for hepatocellular carcinoma (HCC) from the ESMO clinical practice guidelines. Ann. Oncol. 32, 801–805 (2021).
Kudo, M. et al. CheckMate 040 cohort 5: a phase I/II study of nivolumab in patients with advanced hepatocellular carcinoma and Child-Pugh B cirrhosis. J. Hepatol. 75, 600–609 (2021).
Siu, L. L. et al. Challenges and opportunities in adapting clinical trial design for immunotherapies. Clin. Cancer Res. 23, 4950–4958 (2017).
Ritchie, G. et al. Defining the most appropriate primary end point in phase 2 trials of immune checkpoint inhibitors for advanced solid cancers. JAMA Oncol. 4, 522 (2018).
Rimola, J. et al. Radiological response to nivolumab in patients with hepatocellular carcinoma: A multicenter analysis of real-life practice. Eur. J. Radiol. 135, 109484 (2021).
Kim, C. G. et al. Hyperprogressive disease during PD-1 blockade in patients with advanced hepatocellular carcinoma. J. Hepatol. 74, 350–359 (2021).
Hodi, F. S. et al. Immune-modified response evaluation criteria in solid tumors (imRECIST): refining guidelines to assess the clinical benefit of cancer immunotherapy. J. Clin. Oncol. 36, 850–858 (2018).
Anagnostou, V. et al. Immuno-oncology trial endpoints: capturing clinically meaningful activity. Clin. Cancer Res. 23, 4959–4969 (2017).
André, T. et al. Pembrolizumab in microsatellite-instability–high advanced colorectal cancer. N. Engl. J. Med. 383, 2207–2218 (2020).
Martins, F. et al. Adverse effects of immune-checkpoint inhibitors: epidemiology, management and surveillance. Nat. Rev. Clin. Oncol. 16, 563–580 (2019).
Pinato, D. J. et al. PRIME-HCC: phase Ib study of neoadjuvant ipilimumab and nivolumab prior to liver resection for hepatocellular carcinoma. BMC Cancer 21, 301 (2021).
Nordness, M. F. et al. Fatal hepatic necrosis after nivolumab as a bridge to liver transplant for HCC: Are checkpoint inhibitors safe for the pretransplant patient? Am. J. Transplant. 20, 879–883 (2020).
Tabrizian, P. et al. A US multicenter analysis of 2529 HCC patients undergoing liver transplantation: 10‐year outcome assessing the role of down‐staging to within Milan criteria. Hepatology 70, 10A–11A (2019).
Mazzaferro, V. et al. Liver transplantation in hepatocellular carcinoma after tumour downstaging (XXL): a randomised, controlled, phase 2b/3 trial. Lancet Oncol. 21, 947–956 (2020).
Chu, K. F. & Dupuy, D. E. Thermal ablation of tumours: biological mechanisms and advances in therapy. Nat. Rev. Cancer 14, 199–208 (2014).
Chew, V. et al. Immune activation underlies a sustained clinical response to yttrium-90 radioembolisation in hepatocellular carcinoma. Gut 68, 335–346 (2019).
Pinato, D. J. et al. A phase Ib study of pembrolizumab following trans-arterial chemoembolization (TACE) in hepatocellular carcinoma (HCC): PETAL. Ann. Oncol. 30, v288 (2019).
AstraZeneca. Imfinzi plus tremelimumab significantly improved overall survival in HIMALAYA Phase III trial in 1st-line unresectable liver cancer. astrazeneca.com, https://www.astrazeneca.com/media-centre/press-releases/2021/imfinzi-and-tremelimumab-improved-os-in-liver-cancer.html (15 October 2021).
Yau, T. et al. Development of Hong Kong liver cancer staging system with treatment stratification for patients with hepatocellular carcinoma. Gastroenterology 146, 1691–1700.e3 (2014).
Zhou, J. et al. Guidelines for the diagnosis and treatment of hepatocellular carcinoma (2019 Edition). Liver Cancer 9, 682–720 (2020).
Acknowledgements
The work of J.M.L. is supported by grants from the European Commission (EC) Horizon 2020 Programme (HEPCAR, proposal number 667273-2), the US NIH (RO1DK56621 and RO1DK128289), the Samuel Waxman Cancer Research Foundation, EIT Health (CRISH2, Ref. 18053) and the Spanish National Health Institute (MICINN, PID2019-105378RB-I00) through a partnership between Cancer Research UK (CRUK), Fondazione AIRC and Fundación Científica de la Asociación Española Contra el Cáncer (HUNTER, ref. C9380/A26813), and by the Generalitat de Catalunya (AGAUR, SGR-1358). The work of M.H. has been supported by a European Research Council (ERC) Consolidator grant (HepatoMetaboPath), the German Research Foundation (Deutsche Forschungsgemeinschaft; grants SFB/TR 209 Project-ID 314905040 and SFBTR1335 Project-ID 360372040), the Rainer Hoenig Foundation, a Horizon 2020 Programme grant (HEPCAR) and German Cancer Aid (Deutsche Krebshilfe; projects 70113166 and 70113167). M.K.M. acknowledges research support from the CRUK Immunology Project (grant 26603) and the CRUK Accelerator award HUNTER. D.P. acknowledges infrastructural support from the National Institute for Health Research (NIHR) Imperial Biomedical Research Centre and the Imperial Experimental Cancer Medicine Centre (ECMC), and grant funding from the Wellcome Trust Strategic Fund (PS3416), the ASCO Conquer Cancer Foundation Global Oncology Young Investigator Award 2019 (14704), CRUK (C57701/A26137), and by CW+ and the Westminster Medical School Research Trust (JRC SG 009 2018-19).
Author information
Authors and Affiliations
Contributions
All authors contributed to all aspects of the preparation of this manuscript.
Corresponding author
Ethics declarations
Competing interests
J.M.L. has received research support from Bayer HealthCare, Boehringer Ingelheim, Bristol Myers Squibb (BMS), Eisai and Ipsen, and consulting fees from AstraZeneca, Bayer HealthCare, BMS, Eisai, Eli Lilly, Exelixis, Genentech, Glycotest, Ipsen, Merck, Mina Alpha, Nucleix and Roche. M.K.M. is named co-inventor on international patent application No.1917498.6, entitled Treatment of Hepatitis B Virus (HBV) Infection, filed by UCL Business Ltd. M.K.M. has received unrestricted funding from Gilead, Immunocore and Roche, and advisory board/consulting fees from Abbvie, Freeline, Galapagos NV, Gilead, GlaxoSmithKline, Hoffmann La Roche, Immunocore and VIR. D.P. has received consulting fees from AstraZeneca, Bayer HealthCare, BMS, DaVolterra, Eisai, H3 Biomedicine, Ipsen, Mina Alpha, Roche and ViiV Healthcare, and research funding (to institution) from BMS and Merck Sharp & Dohme. The other authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Clinical Oncology thanks A. Bertoletti, Q. Gao and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Llovet, J.M., Castet, F., Heikenwalder, M. et al. Immunotherapies for hepatocellular carcinoma. Nat Rev Clin Oncol 19, 151–172 (2022). https://doi.org/10.1038/s41571-021-00573-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41571-021-00573-2
This article is cited by
-
AMPKα2 promotes tumor immune escape by inducing CD8+ T-cell exhaustion and CD4+ Treg cell formation in liver hepatocellular carcinoma
BMC Cancer (2024)
-
G6PD and machine learning algorithms as prognostic and diagnostic indicators of liver hepatocellular carcinoma
BMC Cancer (2024)
-
Efficacy and safety of the combination of camrelizumab and apatinib in the treatment of liver cancer: a systematic review and single-arm meta-analysis
BMC Gastroenterology (2024)
-
S100A9+CD14+ monocytes contribute to anti-PD-1 immunotherapy resistance in advanced hepatocellular carcinoma by attenuating T cell-mediated antitumor function
Journal of Experimental & Clinical Cancer Research (2024)
-
Prognostic analysis of hepatocellular carcinoma based on cuproptosis -associated lncRNAs
BMC Gastroenterology (2024)