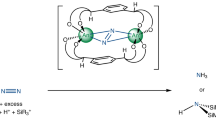
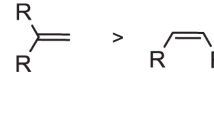Abstract
Carbon oxygenates represent an increasingly important class of feedstock in the development of a sustainable chemical economy. Their catalytic transformation into value-added chemicals is a crucial target, because it would reduce our ties to fossil fuels and non-renewable resources. In this Review, we discuss the unique reactivity offered by actinide metal complexes with respect to s-, p- and d-block metals resulting from the chemical properties particular to these metals. This reactivity is governed by large ionic radii, high coordination numbers, kinetic lability, the involvement of f orbitals in bonding, and single-electron redox processes or σ-bond metathesis, which are distinct from the oxidative addition and reductive elimination pathways commonly seen for catalysts derived from d-block metals. We conclude with a discussion of the current progress in the use of these complexes towards catalytic transformations of oxygenated hydrocarbons.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Bozell, J. J. & Patel, M. K. Feedstocks for the Future Vol. 921 (American Chemical Society, 2006).
Thomas, J. M. & Thomas, W. J. in Principles and Practice of Heterogeneous Catalysis 2nd edn 546–568 (Wiley-VCH, 2014).
Macho, V., Kralik, M. & Komora, L. Progress in commercial and potential industrial processes based on carbon monoxide. Pet. Coal 39, 6–12 (1997).
Zheng, C., Apeloig, Y. & Hoffmann, R. Bonding and coupling of C1 fragments on metal surfaces. J. Am. Chem. Soc. 110, 749–774 (1988).
Olah, G. A., Goeppert, A. & Surya Prakash, G. K. Beyond Oil and Gas: The Methanol Economy 2nd edn (Wiley-VCH, 2009).
Rodemerck, U. et al. Catalyst development for CO2 hydrogenation to fuels. ChemCatChem 5, 1948–1955 (2013).
Finn, C., Schnittger, S., Yellowlees, L. J. & Love, J. B. Molecular approaches to the electrochemical reduction of carbon dioxide. Chem. Commun. 48, 1392–1399 (2012).
Platel, R. H., Hodgson, L. M. & Williams, C. K. Biocompatible initiators for lactide polymerization. Polym. Rev. 48, 11–63 (2008).
Labet, M. & Thielemans, W. Synthesis of polycaprolactone: a review. Chem. Soc. Rev. 38, 3484–3504 (2009).
Olsén, P., Odelius, K. & Albertsson, A.-C. Thermodynamic presynthetic considerations for ring-opening polymerization. Biomacromolecules 17, 699–709 (2016).
Childers, M. I., Longo, J. M., Van Zee, N. J., LaPointe, A. M. & Coates, G. W. Stereoselective epoxide polymerization and copolymerization. Chem. Rev. 114, 8129–8152 (2014).
Lu, E. & Liddle, S. T. Uranium-mediated oxidative addition and reductive elimination. Dalton Trans. 44, 12924–12941 (2015).
Karmel, I., Batrice, R. & Eisen, M. Catalytic organic transformations mediated by actinide complexes. Inorganics 3, 392–428 (2015).
Nugent, L. J., Baybarz, R. D., Burnett, J. L. & Ryan, J. L. Electron-transfer and f → d absorption bands of some lanthanide and actinide complexes and the standard (iii–iv) oxidation potentials for each member of the lanthanide and actinide series. J. Inorg. Nucl. Chem. 33, 2503–2530 (1971).
Nugent, L. J., Baybarz, R. D., Burnett, J. L. & Ryan, J. L. Electron-transfer and f–d absorption bands of some lanthanide and actinide complexes and the standard (ii–iii) oxidation potential for each member of the lanthanide and actinide series. J. Phys. Chem. 77, 1528–1539 (1973).
Sonnenberger, D. C. & Gaudiello, J. G. Cyclic voltammetric study of organoactinide compounds of uranium(iv) and neptunium(iv). Ligand effects on the M(iv)/M(iii) couple. Inorg. Chem. 27, 2747–2748 (1988).
Schelter, E. J. et al. Systematic studies of early actinide complexes: uranium(iv) fluoroketimides. Inorg. Chem. 46, 7477–7488 (2007).
Morris, D. E., Da Re, R. E., Jantunen, K. C., Castro-Rodriguez, I. & Kiplinger, J. L. Trends in electronic structure and redox energetics for early-actinide pentamethylcyclopentadienyl complexes. Organometallics 23, 5142–5153 (2004).
Arnold, P. L. Uranium-mediated activation of small molecules. Chem. Commun. 47, 9005–9010 (2011).
Bruno, J. W., Marks, T. J. & Morss, L. R. Organo-f-element thermochemistry. Metal–ligand bond dissociation enthalpies in (pentamethylcyclopentadienyl)thorium hydrocarbyls, metallacycles, hydrides, and dialkylamides. J. Am. Chem. Soc. 105, 6824–6832 (1983).
Bruno, J. W., Stecher, H. A., Morss, L. R., Sonnenberger, D. C. & Marks, T. J. Organo-f-element thermochemistry. Thorium versus uranium and ancillary ligand effects on metal–ligand bond disruption enthalpies in bis(pentamethylcyclopentadienyl)actinide bis(hydrocarbyls) and bis(pentamethylcyclopentadienyl) alkoxy actinide hydrides and hydrocarbyls. J. Am. Chem. Soc. 108, 7275–7280 (1986).
Bradley, D. C., Saad, M. A. & Wardlaw, W. The preparation of thorium alkoxides. J. Chem. Soc. 1091–1094 (1954).
Bradley, D. C., Chatterjee, A. K. & Wardlaw, W. 439. Structural chemistry of the alkoxides. Part VI. Primary alkoxides of quadrivalent cerium and thorium. J. Chem. Soc. 2260–2264 (1956).
Morss, L. R., Edelstein, N. M. & Fuger, J. (eds) The Chemistry of the Actinide and Transactinide Elements 4th edn 856 (Springer, 2011).
Edelmann, F. T. Lanthanides and actinides: annual survey of their organometallic chemistry covering the year 2015. Coord. Chem. Rev. 318, 29–130 (2016).
Jones, M. B. & Gaunt, A. J. Recent developments in synthesis and structural chemistry of nonaqueous actinide complexes. Chem. Rev. 113, 1137–1198 (2013).
Liddle, S. T. The renaissance of non-aqueous uranium chemistry. Angew. Chem. Int. Ed. 54, 8604–8641 (2015).
Gaunt, A. J. & Neu, M. P. Recent developments in nonaqueous plutonium coordination chemistry. C. R. Chim. 13, 821–831 (2010).
Barnea, E. & Eisen, M. S. Organoactinides in catalysis. Coord. Chem. Rev. 250, 855–899 (2006).
Fox, A. R., Bart, S. C., Meyer, K. & Cummins, C. C. Towards uranium catalysts. Nature 455, 341–349 (2008).
Arnold, P. L. et al. Subtle interactions and electron transfer between UIII, NpIII, or PuIII and uranyl mediated by the oxo group. Angew. Chem. Int. Ed. 55, 12797–12801 (2016).
Gaunt, A. J., Enriquez, A. E., Reilly, S. D., Scott, B. L. & Neu, M. P. Structural characterization of Pu[N(SiMe3)2]3, a synthetically useful nonaqueous plutonium(iii) precursor. Inorg. Chem. 47, 26–28 (2008).
Reilly, S. D., Brown, J. L., Scott, B. L. & Gaunt, A. J. Synthesis and characterization of NpCl4(DME)2 and PuCl4(DME)2 neutral transuranic An(iv) starting materials. Dalton Trans. 43, 1498–1501 (2014).
Brown, J. L. et al. A linear trans-bis(imido) neptunium(v) actinyl analog: NpV(NDipp)2(tBu2bipy)2Cl (Dipp = 2,6-iPr2C6H3). J. Am. Chem. Soc. 137, 9583–9586 (2015).
Brown, J. L. et al. Neptunium and plutonium complexes with a sterically encumbered triamidoamine (TREN) scaffold. Chem. Commun. 52, 5428–5431 (2016).
Macor, J. A. et al. Coordination chemistry of 2,2′-biphenylenedithiophosphinate and diphenyldithiophosphinate with U, Np, and Pu. Dalton Trans. 44, 18923–18936 (2015).
Dutkiewicz, M. S. et al. Organometallic neptunium(iii) complexes. Nat. Chem. 8, 797–802 (2016).
Maron, L., Eisenstein, O. & Andersen, R. A. The bond between CO and Cp′3U in Cp′3U(CO) involves back-bonding from the Cp′3U ligand-based orbitals of π-symmetry, where Cp′represents a substituted cyclopentadienyl ligand. Organometallics 28, 3629–3635 (2009).
Brennan, J. G., Andersen, R. A. & Robbins, J. L. Preparation of the first molecular carbon monoxide complex of uranium, (Me3SiC5H4)3UCO. J. Am. Chem. Soc. 108, 335–336 (1986). The first demonstration that CO can bind to an actinide in a similar manner to its binding to transition-metal cations.
Parry, J., Carmona, E., Coles, S. & Hursthouse, M. Synthesis and single-crystal X-ray diffraction study on the first isolable carbonyl complex of an actinide, (C5Me4H)3U(CO). J. Am. Chem. Soc. 117, 2649–2650 (1995).
Evans, W. J., Kozimor, S. A., Nyce, G. W. & Ziller, J. W. Comparative reactivity of sterically crowded nf3 (C5Me5)3Nd and (C5Me5)3U complexes with CO: formation of a nonclassical carbonium ion versus an f element metal carbonyl complex. J. Am. Chem. Soc. 125, 13831–13835 (2003).
Castro-Rodriguez, I. & Meyer, K. Carbon dioxide reduction and carbon monoxide activation employing a reactive uranium(iii) complex. J. Am. Chem. Soc. 127, 11242–11243 (2005).
Büchner, W. & Weiss, E. Zur Kenntnis der sogenannten «Alkalicarbonyle» iv [1]. Über die Reaktion von geschmolzenem Kalium mit Kohlenmonoxid. Helv. Chim. Acta 47, 1415–1423 (in German) (1964).
Arnold, P. L., Turner, Z. R., Bellabarba, R. M. & Tooze, R. P. Carbon monoxide coupling and functionalisation at a simple uranium coordination complex. Chem. Sci. 2, 77–79 (2011). A reductive coupling of CO to form a C 2 product that demonstrated that further C–H and C–C bond forming (homologation) chemistry is possible.
Mansell, S. M., Kaltsoyannis, N. & Arnold, P. L. Small molecule activation by uranium tris(aryloxides): experimental and computational studies of binding of N2, coupling of CO, and deoxygenation insertion of CO2 under ambient conditions. J. Am. Chem. Soc. 133, 9036–9051 (2011).
Aitken, G. et al. Reductive coupling of carbon monoxide by U(iii) complexes — a computational study. Dalton Trans. 40, 11080–11088 (2011).
McKay, D., Frey, A. S. P., Green, J. C., Cloke, F. G. N. & Maron, L. Computational insight into the reductive oligomerisation of CO at uranium(iii) mixed-sandwich complexes. Chem. Commun. 48, 4118–4120 (2012).
Summerscales, O. T., Cloke, F. G. N., Hitchcock, P. B., Green, J. C. & Hazari, N. Reductive cyclotrimerization of carbon monoxide to the deltate dianion by an organometallic uranium complex. Science 311, 829–831 (2006).
Frey, A. S. et al. Mechanistic studies on the reductive cyclooligomerisation of CO by U(iii) mixed sandwich complexes; the molecular structure of [(U(η-C8H6{(SiiPr3)2-1,4}2)(η-Cp*)]2(μ-η1:η1-C2O2). J. Am. Chem. Soc. 130, 13816–13817 (2008).
Tsoureas, N., Summerscales, O. T., Cloke, F. G. N. & Roe, S. M. Steric effects in the reductive coupling of CO by mixed-sandwich uranium(iii) complexes. Organometallics 32, 1353–1362 (2013).
Gardner, B. M. et al. Homologation and functionalization of carbon monoxide by a recyclable uranium complex. Proc. Natl Acad. Sci. USA 109, 9265–9270 (2012).
Arnold, P. L. & Mansell, S. M. cis-borylated enediolate supported by uranium tris(aryloxide)s. CSD communication 1487672 http://dx.doi.org/10.5517/ccdc.csd.cc1ly1fo (2016).
Buss, J. A. & Agapie, T. Four-electron deoxygenative reductive coupling of carbon monoxide at a single metal site. Nature 529, 72–75 (2016).
Watanabe, T., Ishida, Y., Matsuo, T. & Kawaguchi, H. Reductive coupling of six carbon monoxides by a ditantalum hydride complex. J. Am. Chem. Soc. 131, 3474–3475 (2009).
Frey, A. S. P., Cloke, F. G. N., Coles, M. P. & Hitchcock, P. B. UIII-induced reductive co-coupling of NO and CO to form UIV cyanate and oxo derivates. Chem. Eur. J. 16, 9446–9448 (2010).
Frey, A. S. P., Cloke, F. G. N., Coles, M. P., Maron, L. & Davin, T. Facile conversion of CO/H2 into methoxide at a uranium(iii) center. Angew. Chem. Int. Ed. 50, 6881–6883 (2011).
Maata, E. A. & Marks, T. J. Carbon monoxide activation by organoactinides. Catalytic hydrogenation of inserted carbon monoxide. J. Am. Chem. Soc. 103, 3576–3578 (1981).
Moloy, K. G., Marks, T. J. & Day, V. W. Carbon monoxide activation by organoactinides. η2-acyl-carbon monoxide coupling and the formation of metal-bound ketenes. J. Am. Chem. Soc. 105, 5696–5698 (1983).
Sonnenberger, D. C., Mintz, E. A. & Marks, T. J. Organoactinide carbonylation and carboxylation chemistry. Structural electronic, bond energy, and photochemical effects on migratory insertion in the tris(cyclopentadienyl)thorium hydrocarbyl series. J. Am. Chem. Soc. 106, 3484–3491 (1984).
Fagan, P. J., Moloy, K. G. & Marks, T. J. Carbon monoxide activation by organoactinides. Migratory carbon monoxide insertion into metal–hydrogen bonds to produce mononuclear formyls. J. Am. Chem. Soc. 103, 6959–6962 (1981).
Manriquez, J. M. Fagan, P. J., Marks, T. J., Day, C. S. & Day, V. W. Bis(pentamethylcyclopentadienyl)actinide alkyls: facile activation of carbon monoxide, carbon–carbon double bond formation, and the production of unusual oxygen-bonded migratory insertion products. J. Am. Chem. Soc. 100, 7112–7114 (1978). Not only the first example of CO insertion into an An–C bond but also the demonstration of a wider and complicated range of CO and hydrocarbyl reactions that form various Fischer–Tropsch-like products and have since been extensively built upon.
Marks, T. J. Actinide organometallic chemistry. Science 217, 989–997 (1982).
Kloppenburg, L. & Petersen, J. L. Facile conversion of an appended silylamido to a silyloxy ligand via isocyanate elimination. Synthesis of {[(C5Me4)SiMe2O]Zr(η2-O2CMe)(μ-O2CMe)}2 via the carboxylation of [(C5Me4)SiMe2(NtBu)]ZrMe2 . Organometallics 15, 7–9 (1996).
Weydert, M., Brennan, J. G., Andersen, R. A. & Bergman, R. G. Reactions of a uranium(iv) tertiary alkyl bond: facile ligand-assisted reduction and insertion of ethylene and carbon monoxide. Organometallics 14, 3942–3951 (1995).
Fagan, P. J. et al. Carbon monoxide activation by f-element organometallics. An unusually distorted, carbenelike dihaptoacyl and CO tetramerization. J. Am. Chem. Soc. 102, 5393–5396 (1980).
Katahira, D. A., Moloy, K. G. & Marks, T. J. Carbon monoxide activation by organoactinides. Formyl pathways in CO homologation and hydrogenation. Organometallics 1, 1723–1726 (1982).
Moloy, K. G., Fagan, P. J., Manriquez, J. M. & Marks, T. J. A synthetic and mechanistic study of oxycarbene-like coupling reaction patterns of actinide η2-acyl complexes with carbon monoxide and isocyanides. J. Am. Chem. Soc. 108, 56–67 (1986).
Dormond, A., Aaliti, A., Elbouadili, A. & Moise, C. Réactivité des liaisons σ uranium–carbone: réactions du méthyltris(hexaméthyldisilylamido)uranium. J. Organomet. Chem. 329, 187–199 (in French) (1987).
Arnold, P. L. et al. Carbon monoxide and carbon dioxide insertion chemistry of f-block N-heterocyclic carbene complexes. Dalton Trans. 42, 1333–1337 (2013).
Benaud, O., Berthet, J. C., Thuery, P. & Ephritikhine, M. The bis metallacyclic anion [U(N{SiMe3}2)(CH2SiMe2N{SiMe3})2]−. Inorg. Chem. 49, 8117–8130 (2010).
Evans, W. J., Siladke, N. A. & Ziller, J. W. Synthesis and reactivity of a silylalkyl double tuck-in uranium metallocene [(η5:η1-C5Me4SiMe2CH2)2U] and its conversion to bis(tethered) metallocenes. Chem. Eur. J. 16, 796–800 (2010).
Fagan, P. J. et al. Insertion of carbon monoxide into metal–nitrogen bonds. Synthesis, chemistry, structures, and structural dynamics of bis(pentamethylcyclopentadienyl) organoactinide dialkylamides and η2-carbamoyls. J. Am. Chem. Soc. 103, 2206–2220 (1981).
Castro-Rodriguez, I., Nakai, H., Zakharov, L. N., Rheingold, A. L. & Meyer, K. A linear, O-coordinated η1-CO2 bound to uranium. Science 305, 1757–1759 (2004).
Castro-Rodriguez, I. & Meyer, K. Small molecule activation at uranium coordination complexes: control of reactivity via molecular architecture. Chem. Commun. 1353–1368 (2006).
Pokharel, U. R., Fronczek, F. R. & Maverick, A. W. Reduction of carbon dioxide to oxalate by a binuclear copper complex. Nat. Commun. 5, 5883 (2014).
Horn, B., Limberg, C., Herwig, C. & Braun, B. Nickel(i)-mediated transformations of carbon dioxide in closed synthetic cycles: reductive cleavage and coupling of CO2 generating NiICO, NiIICO3 and NiIIC2O4NiII entities. Chem. Commun. 49, 10923–10925 (2013).
Angamuthu, R., Byers, P., Lutz, M., Spek, A. L. & Bouwman, E. Electrocatalytic CO2 conversion to oxalate by a copper complex. Science 327, 313–315 (2010).
Thomas, A. M., Lin, B.-L., Wasinger, E. C. & Stack, T. D. P. Ligand noninnocence of thiolate/disulfide in dinuclear copper complexes: solvent-dependent redox isomerization and proton-coupled electron transfer. J. Am. Chem. Soc. 135, 18912–18919 (2013).
Evans, W. J., Seibel, C. A. & Ziller, J. W. Organosamarium-mediated transformations of CO2 and COS: monoinsertion and disproportionation reactions and the reductive coupling of CO2 to [O2CCO2]2−. Inorg. Chem. 37, 770–776 (1998).
Schmidt, A.-C. et al. Activation of SO2 and CO2 by trivalent uranium leading to sulfite/dithionite and carbonate/oxalate complexes. Chem. Eur. J. 20, 13501–13506 (2014).
Tsoureas, N., Castro, L., Kilpatrick, A. F. R., Cloke, F. G. N. & Maron, L. Controlling selectivity in the reductive activation of CO2 by mixed sandwich uranium(iii) complexes. Chem. Sci. 5, 3777–3788 (2014). An important observation that ligand tuning for steric rather than electronic effects may be key to generating the first simple homogeneous catalysts for CO 2 coupling to the desirable C 2 product oxalate.
Berthet, J. C. et al. Synthesis and crystal-structure of the oxo-bridged bimetallic organouranium complex [(Me3SiC5H4)3U]2[μ-O]. J. Organomet. Chem. 408, 335–341 (1991).
Summerscales, O. T., Frey, A. S. P., Cloke, F. G. N. & Hitchcock, P. B. Reductive disproportionation of carbon dioxide to carbonate and squarate products using a mixed-sandwich U(III) complex. Chem. Commun. 2, 198–200 (2008).
Mougel, V. et al. Siloxides as supporting ligands in uranium(III)-mediated small-molecule activation. Angew. Chem. Int. Ed. 51, 12280–12284 (2012).
Lam, O. P., Bart, S. C., Kameo, H., Heinemann, F. W. & Meyer, K. Insights into the mechanism of carbonate formation through reductive cleavage of carbon dioxide with low-valent uranium centers. Chem. Commun. 46, 3137–3139 (2010).
Schmidt, A.-C., Nizovtsev, A. V., Scheurer, A., Heinemann, F. W. & Meyer, K. Uranium-mediated reductive conversion of CO2 to CO and carbonate in a single-vessel, closed synthetic cycle. Chem. Commun. 48, 8634–8636 (2012).
Cooper, O. et al. Multimetallic cooperativity in uranium-mediated CO2 activation. J. Am. Chem. Soc. 136, 6716–6723 (2014).
Lam, O. P., Franke, S. M., Heinemann, F. W. & Meyer, K. Reactivity of U–E–U (E=S, Se) toward CO2, CS2, and COS: new mixed-carbonate complexes of the types U–CO2E–U (E=S, Se), U–CS2E–U (E=O, Se), and U–COSSe–U. J. Am. Chem. Soc. 134, 16877–16881 (2012).
Moloy, K. G. & Marks, T. J. The insertion of carbon dioxide into actinide alkyl and hydride bonds. Inorg. Chim. Acta 110, 127–131 (1985).
Evans, W. J., Walensky, J. R. & Ziller, J. W. Insertion reactivity of CO2, PhNCO, Me3CC≡N, and Me3CN≡C with the uranium–alkynyl bonds in (C5Me5)2U(C≡CPh)2 . Organometallics 29, 945–950 (2010).
Webster, C. L., Ziller, J. W. & Evans, W. J. Synthesis and CO2 insertion reactivity of allyluranium metallocene complexes. Organometallics 31, 7191–7197 (2012).
Higgins, J. A., Cloke, F. G. N. & Roe, S. M. Synthesis and CO2 insertion chemistry of uranium(iv) mixed-sandwich alkyl and hydride complexes. Organometallics 32, 5244–5252 (2013).
Button, Z. E., Higgins, J. A., Suvova, M., Cloke, F. G. N. & Roe, S. M. Mixed sandwich thorium complexes incorporating bis(tri-isopropylsilyl)cyclooctatetraenyl and pentamethylcyclopentadienyl ligands: synthesis, structure and reactivity. Dalton Trans. 44, 2588–2596 (2015).
Matson, E. M., Forrest, W. P., Fanwick, P. E. & Bart, S. C. Functionalization of carbon dioxide and carbon disulfide using a stable uranium(III) alkyl complex. J. Am. Chem. Soc. 133, 4948–4954 (2011).
Mora, E. et al. Diamine bis(phenolate) as supporting ligands in organoactinide(iv) chemistry. Synthesis, structural characterization, and reactivity of stable dialkyl derivatives. Organometallics 32, 1409–1422 (2013).
Korobkov, I. & Gambarotta, S. Cis double addition of CO2 to a coordinated arene of a thorium complex. Organometallics 23, 5379–5381 (2004).
Formanuik, A. et al. Concomitant carboxylate and oxalate formation from the activation of CO2 by a thorium(III) complex. Chem. Eur. J.http://dx.doi.org/10.1002/chem.201604622 (2016).
Bagnall, K. W. & Yanir, E. Thorium and uranium carbamates from M(NR2)4 . J. Inorg. Nucl. Chem. 36, 777–779 (1974).
Schmidt, A.-C., Heinemann, F. W., Maron, L. & Meyer, K. A series of uranium (IV, V, VI) tritylimido complexes, their molecular and electronic structures and reactivity with CO2 . Inorg. Chem. 53, 13142–13153 (2014).
Higgins Frey, J. A., Cloke, F. G. N. & Roe, S. M. Synthesis and reactivity of a mixed-sandwich uranium(IV) primary amido complex. Organometallics 34, 2102–2105 (2015).
Kahan, R. J., Cloke, F. G. N., Roe, S. M. & Nief, F. Activation of carbon dioxide by new mixed sandwich uranium(III) complexes incorporating cyclooctatetraenyl and pyrrolide, phospholide, or arsolide ligands. New J. Chem. 39, 7602–7607 (2015).
Bart, S. C. et al. Carbon dioxide activation with sterically pressured mid- and high-valent uranium complexes. J. Am. Chem. Soc. 130, 12536–12546 (2008).
Schmidt, A.-C., Heinemann, F. W., Lukens, W. W. & Meyer, K. Molecular and electronic structure of dinuclear uranium bis-μ-oxo complexes with diamond core structural motifs. J. Am. Chem. Soc. 136, 11980–11993 (2014).
Falcone, M., Chatelain, L. & Mazzanti, M. Nucleophilic reactivity of a nitride-bridged diuranium(IV) complex: CO2 and CS2 functionalization. Angew. Chem. Int. Ed. 55, 4074–4078 (2016).
Camp, C. et al. CO2 conversion to isocyanate via multiple N–Si bond cleavage at a bulky uranium(III) complex. Chem. Commun. 51, 15454–15457 (2015).
Ding, W. & Wang, D. Does NHC directly participate in the CO2 insertion into the UIII-N bond? A density functional theory study. Organometallics 33, 7007–7010 (2014).
Matson, E. M., Fanwick, P. E. & Bart, S. C. Formation of trivalent U–C, U–N, and U–S bonds and their reactivity toward carbon dioxide and acetone. Organometallics 30, 5753–5762 (2011).
Zuend, S. J., Lam, O. P., Heinemann, F. W. & Meyer, K. Carbon dioxide insertion into uranium-activated dicarbonyl complexes. Angew. Chem. Int. Ed. 50, 10626–10630 (2011).
Coates, G. W. & Moore, D. R. Discrete metal-based catalysts for the copolymerization of CO2 and epoxides: discovery, reactivity, optimization, and mechanism. Angew. Chem. Int. Ed. 43, 6618–6639 (2004).
Kember, M. R., Buchard, A. & Williams, C. K. Catalysts for CO2/epoxide copolymerisation. Chem. Commun. 47, 141–163 (2011).
Trott, G., Saini, P. K. & Williams, C. K. Catalysts for CO2/epoxide ring-opening copolymerization. Philos. Trans. R. Soc. A 374, 20150085 (2016).
Baker, R. J. & Walshe, A. New reactivity of the uranyl ion: ring opening polymerisation of epoxides. Chem. Commun. 48, 985–987 (2012).
Fang, J., Walshe, A., Maron, L. & Baker, R. J. Ring-opening polymerization of epoxides catalyzed by uranyl complexes: an experimental and theoretical study of the reaction mechanism. Inorg. Chem. 51, 9132–9140 (2012).
Arnold, P. L. et al. C 3-symmetric lanthanide tris(alkoxide) complexes formed by preferential complexation and their stereoselective polymerization of rac-lactide. Angew. Chem. Int. Ed. 47, 6033–6036 (2008).
Buffet, J.-C., Kapelski, A. & Okuda, J. Stereoselective polymerization of meso-lactide: syndiotactic polylactide by heteroselective initiators based on trivalent metals. Macromolecules 43, 10201–10203 (2010).
Otero, A. et al. Ring-opening polymerization of cyclic esters by an enantiopure heteroscorpionate rare earth initiator. Angew. Chem. Int. Ed. 48, 2176–2179 (2009).
Mou, Z. et al. Efficient and heteroselective heteroscorpionate rare-earth-metal zwitterionic initiators for ROP of rac-lactide: role of σ-ligand. Macromolecules 47, 2233–2241 (2014).
Ren, W., Zhao, N., Chen, L. & Zi, G. Synthesis, structure, and catalytic activity of benzyl thorium metallocenes. Inorg. Chem. Commun. 30, 26–28 (2013).
Ren, W., Zhao, N., Chen, L., Song, H. & Zi, G. Synthesis, structure, and catalytic activity of an organothorium hydride complex. Inorg. Chem. Commun. 14, 1838–1841 (2011).
Barnea, E., Moradove, D., Berthet, J.-C., Ephritikhine, M. & Eisen, M. S. Surprising activity of organoactinide complexes in the polymerization of cyclic mono- and diesters. Organometallics 25, 320–322 (2006).
Hayes, C. E., Sarazin, Y., Katz, M. J., Carpentier, J.-F. & Leznoff, D. B. Diamido-ether actinide complexes as initiators for lactide ring-opening polymerization. Organometallics 32, 1183–1192 (2013).
Villiers, C., Thué ry, P. & Ephritikhine, M. A comparison of analogous 4f- and 5f-element compounds: syntheses, X-ray crystal structures and catalytic activity of the homoleptic amidinate complexes [M{MeC(NCy)2}3] (M=La, Nd or U). Eur. J. Inorg. Chem. 2004, 4624–4632 (2004).
Das, R. K. et al. Group 4 lanthanide and actinide organometallic inclusion complexes. Organometallics 34, 742–752 (2015).
Rabinovich, E., Aharonovich, S., Botoshansky, M. & Eisen, M. S. Thorium 2-pyridylamidinates: synthesis, structure and catalytic activity towards the cyclo-oligomerization of ε-caprolactone. Dalton Trans. 39, 6667–6676 (2010).
Karmel, I. S. R., Elkin, T., Fridman, N. & Eisen, M. S. Dimethylsilyl bis(amidinate)actinide complexes: synthesis and reactivity towards oxygen containing substrates. Dalton Trans. 43, 11376–11387 (2014).
Karmel, I. S. R., Fridman, N. & Eisen, M. S. Actinide amidinate complexes with a dimethylamine side arm: synthesis, structural characterization, and reactivity. Organometallics 34, 636–643 (2015).
Karmel, I. S. R., Botoshansky, M., Tamm, M. & Eisen, M. S. Uranium(IV) imidazolin-2-iminato complexes: a new class of actinide complexes. Inorg. Chem. 53, 694–696 (2014).
Karmel, I. S. R., Fridman, N., Tamm, M. & Eisen, M. S. Mixed imidazolin-2-iminato–Cp* thorium(IV) complexes: synthesis and reactivity toward oxygen-containing substrates. Organometallics 34, 2933–2942 (2015).
Karmel, I. S. R., Khononov, M., Tamm, M. & Eisen, M. S. Uranium-mediated ring-opening polymerization of ε-caprolactone: a comparative study. Catal. Sci. Technol. 5, 5110–5119 (2015).
Walshe, A., Fang, J., Maron, L. & Baker, R. J. New mechanism for the ring-opening polymerization of lactones? Uranyl aryloxide-induced intermolecular catalysis. Inorg. Chem. 52, 9077–9086 (2013). A new reaction mechanism for the polymerization of lactones identified through the study of a uranyl aryloxide catalyst.
Barnea, E., Andrea, T., Kapon, M. & Eisen, M. S. Formation of inclusion organoactinide complexes with boron-containing macrocycles. J. Am. Chem. Soc. 126, 5066–5067 (2004).
Andrea, T., Barnea, E. & Eisen, M. S. Organoactinides promote the Tishchenko reaction: the myth of inactive actinide–alkoxo complexes. J. Am. Chem. Soc. 130, 2454–2455 (2008). The first example of catalytic coupling of aldehydes by actinide complexes that also proceeds through an unexpected An–O bond activation, indicating that actinide alkoxide complexes can be efficient catalysts.
Karmel, I. S. R., Fridman, N., Tamm, M. & Eisen, M. S. Mono(imidazolin-2-iminato) actinide complexes: synthesis and application in the catalytic dimerization of aldehydes. J. Am. Chem. Soc. 136, 17180–17192 (2014).
Sharma, M. et al. Organoactinides promote the dimerization of aldehydes: scope, kinetics, thermodynamics, and calculation studies. J. Am. Chem. Soc. 133, 1341–1356 (2011).
Wobser, S. D. & Marks, T. J. Organothorium-catalyzed hydroalkoxylation/cyclization of alkynyl alcohols. Scope, mechanism, and ancillary ligand effects. Organometallics 32, 2517–2528 (2013). The rate-limiting step of this actinide-mediated hydroalkoxylation and cyclization involves the first example of C≡C bond insertion into the strong An–O bond.
Weiss, C. J., Wobser, S. D. & Marks, T. J. Organoactinide-mediated hydrothiolation of terminal alkynes with aliphatic, aromatic, and benzylic thiols. J. Am. Chem. Soc. 131, 2062–2063 (2009).
Weiss, C. J., Wobser, S. D. & Marks, T. J. Lanthanide- and actinide-mediated terminal alkyne hydrothiolation for the catalytic synthesis of Markovnikov vinyl sulfides. Organometallics 29, 6308–6320 (2010).
Cramer, R. E., Jeong, J. H. & Gilje, J. W. Uranium–carbon multiple-bond chemistry. 9. The insertion of phenyl isocyanate into the uranium–carbon bond of Cp3U:CHP(Ph)(R)(Me) to form Cp3U[(NPh)(O)CCHP(Ph)(R)(Me)]. Organometallics 6, 2010–2012 (1987).
Cooper, O. J., Mills, D. P., Lewis, W., Blake, A. J. & Liddle, S. T. Reactivity of the uranium(iv) carbene complex [U(BIPMTMS)(Cl)(μ-Cl)2Li(THF)2] (BIPMTMS={C(PPh2NSiMe3)2}) towards carbonyl and heteroallene substrates: metallo-Wittig, adduct formation, C–F bond activation, and [2 + 2]-cycloaddition reactions. Dalton Trans. 43, 14275–14283 (2014).
Karmel, I. S. R., Tamm, M. & Eisen, M. S. Actinide-mediated catalytic addition of E–H Bonds (E=N, P, S) to carbodiimides, Iisocyanates, and isothiocyanates. Angew. Chem. Int. Ed. 54, 12422–12425 (2015). Demonstration that the actinide mediated addition of E–H bonds to heterocumulenes is both catalytic and also tolerant of functional groups and heteroatoms.
Batrice, R. J. & Eisen, M. S. Catalytic insertion of E–H bonds (E=C, N, P, S) into heterocumulenes by amido–actinide complexes. Chem. Sci. 7, 939–944 (2016).
Batrice, R. J., Kefalidis, C. E., Maron, L. & Eisen, M. S. Actinide-catalyzed intermolecular addition of alcohols to carbodiimides. J. Am. Chem. Soc. 138, 2114–2117 (2016).
Cooper, O. J. et al. The nature of the U=C double bond: pushing the stability of high-oxidation-state uranium carbenes to the limit. Chem. Eur. J. 19, 7071–7083 (2013).
Lam, O. P., Anthon, C., Heinemann, F. W., O'Connor, J. M. & Meyer, K. Structural and spectroscopic characterization of a charge-separated uranium benzophenone ketyl radical complex. J. Am. Chem. Soc. 130, 6567–6576 (2008). By using a complex ligand architecture that is sterically demanding, a rare benzophenone ketyl radical metal complex can be isolated.
Sternal, R. S., Sabat, M. & Marks, T. J. Metal–metal bonds involving actinides. Functionalization of activated carbon–hydrogen bonds and unusual oligomerization chemistry mediated by a thorium–ruthenium complex. J. Am. Chem. Soc. 109, 7920–7921 (1987).
Campello, M. P. C. et al. Hydrocarbyl derivatives of [UCl2{HB(pz)3}2]: synthesis, characterization and reactivity studies towards protic substrates and ketones. J. Organomet. Chem. 538, 223–239 (1997).
Fagan, P. J., Manriquez, J. M., Maatta, E. A., Seyam, A. M. & Marks, T. J. Synthesis and properties of bis(pentamethylcyclopentadienyl) actinide hydrocarbyls and hydrides. A new class of highly reactive f-element organometallic compounds. J. Am. Chem. Soc. 103, 6650–6667 (1981).
Pedrick, E. A. et al. Synthesis and reactivity of a U(IV) dibenzyne complex. Organometallics 35, 494–502 (2016).
Kraft, S. J., Fanwick, P. E. & Bart, S. C. Exploring the insertion chemistry of tetrabenzyluranium using carbonyls and organoazides. Organometallics 32, 3279–3285 (2013).
Silva, M., Domingos, A., Pires de Matos, A., Marques, N. & Trofimenko, S. Hydrotris(mesitylpyrazol-1-yl)borate uranium(IV) compounds: synthesis, structure, and ligand isomerization. Dalton Trans. 4628–4634 (2000).
Matson, E. M., Forrest, W. P., Fanwick, P. E. & Bart, S. C. Synthesis and reactivity of trivalent Tp*U(CH2Ph)2(THF): insertion versus oxidation at low-valent uranium–carbon bonds. Organometallics 32, 1484–1492 (2013).
Kiernicki, J. J. et al. Multielectron C–O bond activation mediated by a family of reduced uranium complexes. Inorg. Chem. 53, 3730–3741 (2014). An interesting demonstration of metal–ligand cooperativity as uranium complexes bearing redox-active ligands promote the pinacol coupling; here, the ligand provides the reducing equivalents necessary rather than the uranium centre.
Mohammad, A., Cladis, D. P., Forrest, W. P., Fanwick, P. E. & Bart, S. C. Reductive heterocoupling mediated by Cp*2U(2,2′-bpy). Chem. Commun. 48, 1671–1673 (2012).
Crozier, A. R., Tö rnroos, K. W., Maichle-Mössmer, C. & Anwander, R. Trivalent cerium and praseodymium aromatic ketone adducts. Eur. J. Inorg. Chem. 2013, 409–414 (2013).
Heeres, H. J., Maters, M., Teuben, J. H., Helgesson, G. & Jagner, S. Organolanthanide-induced carbon–carbon bond formation. Preparation and properties of monomeric lanthanide aldolates and enolates. Organometallics 11, 350–356 (1992).
Kim, J. E., Zabula, A. V., Carroll, P. J. & Schelter, E. J. 1,2-Addition or enolization? Variable reactivity of a cerium acetylide complex toward carbonyl compounds. Organometallics 35, 2086–2091 (2016).
Sen, A., Stecher, H. A. & Rheingold, A. L. Synthesis, structure, and reactivity of homoleptic cerium(IV) and cerium(III) alkoxides. Inorg. Chem. 31, 473–479 (1992).
Hou, Z., Miyano, T., Yamazaki, H. & Wakatsuki, Y. Well-defined metal ketyl complex: Sm(ketyl)(OAr)2(THF)2 and its reversible coupling to a disamarium(III) pinacolate. J. Am. Chem. Soc. 117, 4421–4422 (1995).
Hou, Z. et al. One-electron reduction of aromatic ketones by low-valent lanthanides. Isolation, structural characterization, and reactivity of lanthanide ketyl complexes. J. Am. Chem. Soc. 120, 754–766 (1998).
Hou, Z., Koizumi, T.-a., Nishiura, M. & Wakatsuki, Y. Lanthanide(II) complexes bearing linked cyclopentadienyl–anilido ligands: synthesis, structures, and one-electron-transfer and ethylene polymerization reactions. Organometallics 20, 3323–3328 (2001).
Qin, J., Xu, B., Zhang, Y., Yuan, D. & Yao, Y. Cooperative rare earth metal–zinc based heterometallic catalysts for copolymerization of CO2 and cyclohexene oxide. Green Chem. 18, 4270–4275(2016).
Dong, Y., Wang, X., Zhao, X. & Wang, F. Facile synthesis of poly(ether carbonate)s via copolymerization of CO2 and propylene oxide under combinatorial catalyst of rare earth ternary complex and double metal cyanide complex. J. Polym. Sci. A 50, 362–370 (2012).
Cui, D., Nishiura, M., Tardif, O. & Hou, Z. Rare-earth-metal mixed hydride/aryloxide complexes bearing mono(cyclopentadienyl) ligands. Synthesis, CO2 fixation, and catalysis on copolymerization of CO2 with cyclohexene oxide. Organometallics 27, 2428–2435 (2008).
Vitanova, D. V., Hampel, F. & Hultzsch, K. C. Rare earth metal complexes based on β-diketiminato and novel linked bis(β-diketiminato) ligands: synthesis, structural characterization and catalytic application in epoxide/CO2-copolymerization. J. Organomet. Chem. 690, 5182–5197 (2005).
Quan, Z., Wang, X., Zhao, X. & Wang, F. Copolymerization of CO2 and propylene oxide under rare earth ternary catalyst: design of ligand in yttrium complex. Polymer 44, 5605–5610 (2003).
Zhao, B., Lu, C. R. & Shen, Q. Ring-opening polymerization of trimethylene carbonate and its copolymerization with ε-caprolactone by lanthanide(II) aryloxide complexes. J. Appl. Polym. Sci. 106, 1383–1389 (2007).
Mas, C., Ramis, X., Salla, J. M., Mantecon, A. & Serra, A. Cationic copolymerization of diglycidyl ether of bisphenol A with phthalide or 3,3′-diphthalide catalyzed by lanthanide triflates. J. Polym. Sci. A 44, 1711–1721 (2006).
Li, C., Wang, Y., Zhou, L., Sun, H. & Shen, Q. Homoleptic lanthanide amidinate complexes: a single-component initiator for ring-opening polymerization of trimethylene carbonate and copolymerization with ε-caprolactone. J. Appl. Polym. Sci. 102, 22–28 (2006).
Zhou, L., Sun, H., Chen, J., Yao, Y. & Shen, Q. Homoleptic lanthanide guanidinate complexes: the effective initiators for the polymerization of trimethylene carbonate and its copolymerization with ε-caprolactone. J. Polym. Sci. A 43, 1778–1786 (2005).
Fan, L., Xiong, Y.-B., Xu, H. & Shen, Z.-Q. l-Lactide homopolymerization and l-lactide-ε-caprolactone block copolymerization by lanthanide tris(2,4,6-trimethylphenolate)s. Eur. Polym. J. 41, 1647–1653 (2005).
Cervellera, R., Ramis, X., Salla, J. M., Serra, A. & Mantecon, A. New thermosets obtained by copolymerization of DGEBA with 1,5,7,11-tetraoxaspiro[5,5]undecane catalyzed by lanthanide triflates. Polymer 46, 6878–6887 (2005).
Yasuda, H. & Desurmont, G. Block copolymerizations of higher 1-olefins with traditional polar monomers using metallocene-type single component lanthanide initiators. Polym. Int. 53, 1017–1024 (2004).
Gromada, J., Chenal, T., Mortreux, A., Leising, F. & Carpentier, J.-F. Homogeneous and heterogeneous alkyl-alkoxo-lanthanide type catalysts for polymerization and block-copolymerization of ethylene and methyl methacrylate. J. Mol. Catal. A 182–183, 525–531 (2002).
Desurmont, G., Tokimitsu, T. & Yasuda, H. First controlled block copolymerizations of higher 1-olefins with polar monomers using metallocene type single component lanthanide initiators. Macromolecules 33, 7679–7681 (2000).
Kaltsoyannis, N. Does covalency increase or decrease across the actinide series? Implications for minor actinide partitioning. Inorg. Chem. 52, 3407–3413 (2013).
Horiuchi, S. et al. Above-room-temperature ferroelectricity in a single-component molecular crystal. Nature 463, 789–792 (2010).
Henrici-Olivé, G. & Olivé, S. The Fischer–Tropsch synthesis: molecular weight distribution of primary products and reaction mechanism. Angew. Chem. Int. Ed. Engl. 15, 136–141 (1976).
Yang, P., Warnke, I., Martin, R. L. & Hay, P. J. Theoretical studies of the sp2 versus sp3 C–H bond activation chemistry of 2-picoline by (C5Me5)2An(CH3)2 complexes (An=Th, U). Organometallics 27, 1384–1392 (2008).
Lohr, T. L., Li, Z. & Marks, T. J. Thermodynamic strategies for C–O bond formation and cleavage via tandem catalysis. Acc. Chem. Res. 49, 824–834 (2016).
Andrez, J. et al. Synthesis and reactivity of a terminal uranium(iv) sulfide supported by siloxide ligands. Chem. Sci. 7, 5846–5856 (2016).
Smiles, D. E., Wu, G. & Hayton, T. W. Reversible chalcogen-atom transfer to a terminal uranium sulfide. Inorg. Chem. 53, 12683–12685 (2014).
Zhu, Q. et al. Sulfur as a selective ‘soft’ oxidant for catalytic methane conversion probed by experiment and theory. Nat. Chem. 5, 104–109 (2013).
Hutchings, G. J., Heneghan, C. S., Hudson, I. D. & Taylor, S. H. Uranium-oxide-based catalysts for the destruction of volatile chloro-organic compounds. Nature 384, 341–343 (1996).
Collette, H. et al. The potential use of uranium oxides and uranium–bismuth mixed oxides in catalysis. Faraday Trans. 83, 1263–1271 (1987).
Ismagilov, Z. R. et al. Characterization of new catalysts based on uranium oxides. Kinet. Catal. 48, 511–520 (2007).
Davachi, S. M. & Kaffashi, B. Polylactic acid in medicine. Polym. Plast. Technol. Eng. 54, 944–967 (2015).
Baumann, M. & Baxendale, I. R. An overview of the synthetic routes to the best selling drugs containing 6-membered heterocycles. Beil. Org. Chem. 9, 2265–2319 (2013).
Weiss, C. J. & Marks, T. J. Organo-f-element catalysts for efficient and highly selective hydroalkoxylation and hydrothiolation. Dalton Trans. 39, 6576–6588 (2010).
Nolan, S. P., Stern, D. & Marks, T. J. Organo-f-element thermochemistry. Absolute metal–ligand bond disruption enthalpies in bis(pentamethylcyclopentadienyl)samarium hydrocarbyl, hydride, dialkylamide, alkoxide, halide, thiolate, and phosphide complexes. Implications for organolanthanide bonding and reactivity. J. Am. Chem. Soc. 111, 7844–7853 (1989).
Dias, A. R. & Martinho Simõ es, J. A. Thermochemistry of M(η5-C5H5)2Ln complexes (M=Ti, Mo, W). Polyhedron 7, 1531–1544 (1988).
Schock, L. E. & Marks, T. J. Organometallic thermochemistry. Metal hydrocarbyl, hydride, halide, carbonyl, amide, and alkoxide bond enthalpy relationships and their implications in pentamethylcyclopentadienyl and cyclopentadienyl complexes of zirconium and hafnium. J. Am. Chem. Soc. 110, 7701–7715 (1988).
Simoes, J. A. M. & Beauchamp, J. L. Transition metal–hydrogen and metal–carbon bond strengths: the keys to catalysis. Chem. Rev. 90, 629–688 (1990).
Acknowledgements
The authors thank the Engineering and Physical Sciences Research Council (EPSRC), UK (grant numbers EP/M010554/1, EP/K014714/1 and EP/J018139/1), the European Cooperation in Science and Technology (COST) Network CM1205, and Z.R.T. thanks SCG Chemicals for funding. Z.R.T also thanks Trinity College, Oxford for a Junior Research Fellowship.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Glossary
- σ-Bond metathesis
-
A reaction that replaces one σ-bonded ligand in M–X with another in a substrate E–X′ to form M–X′ via a four-centre transition state and without any change in formal oxidation state of the metal.
- Oxidative addition
-
A common and widely exploited reaction in homogeneous d-block catalytic chemistry that involves the addition of the E–X bond of a substrate to a metal centre (L)nM using the two electrons from the E–X bond and two metal valence electrons, resulting in the formation of (L)nM(E)(X) and an increase in formal oxidation state of the metal by two.
- Reductive elimination
-
Formally the reverse of oxidative addition, in which a molecule or fragment is released from a metal complex (L)nM(E)(X), returning two electrons to the metal and forming a new E–X bond. Often the key to substrate release and, therefore, turnover in redox-based homogeneous catalysis.
- Cp*
-
(C5Me5). A cyclic aromatic monoanionic ligand that binds strongly with a moderate degree of covalency through the five-ring carbon atoms to an actinide centre. The hapto (η) prefix indicates that all ring C atoms bind to the metal. A pair provide an excellent supporting, ancillary ligand set for various organo-actinide chemistry studies at X in [(η-Cp*)2AnX2]-type complexes. The related cyclic, aromatic η-C8R8 dianions, binding through eight ring carbon atoms, are also commonly used.
- Migratory insertion
-
The coupling of two M-bound ligands, one anionic (X; generally an alkyl, hydride, amide or alkoxide in this Review) and one neutral (L; generally CO or an ether, ketone or aldehyde in this Review, for which the initial binding may not necessarily have been an observable event) that generates a new M-bound X ligand that includes the group L.
- Turnover frequency
-
Related to the turnover number, this is the number of substrate molecules converted per active catalyst site in a given unit time.
- Turnover number
-
The number of substrate molecules converted per active catalyst site. Overall turnover number can be related to catalyst stability or longevity.
- Markovnikov addition
-
Addition of HX across a C–C multiple bond with H addition at the least substituted carbon.
Rights and permissions
About this article
Cite this article
Arnold, P., Turner, Z. Carbon oxygenate transformations by actinide compounds and catalysts. Nat Rev Chem 1, 0002 (2017). https://doi.org/10.1038/s41570-016-0002
Published:
DOI: https://doi.org/10.1038/s41570-016-0002
This article is cited by
-
Oxalate production via oxidation of ascorbate rather than reduction of carbon dioxide
Nature Communications (2021)
-
Relativistic quantum chemical calculations show that the uranium molecule U2 has a quadruple bond
Nature Chemistry (2019)
-
Transition-metal-bridged bimetallic clusters with multiple uranium–metal bonds
Nature Chemistry (2019)
-
Direct observation of pure pentavalent uranium in U2O5 thin films by high resolution photoemission spectroscopy
Scientific Reports (2018)
-
Double dative bond between divalent carbon(0) and uranium
Nature Communications (2018)