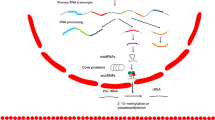
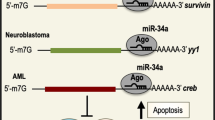
Abstract
Transfer RNAs (tRNAs) have been historically viewed as non-dynamic adaptors that decode the genetic code into proteins. Recent work has uncovered dynamic regulatory roles for these fascinating molecules. Advances in tRNA detection methods have revealed that specific tRNAs can become modulated upon DNA copy number and chromatin alterations and can also be perturbed by oncogenic signalling and transcriptional regulators in cancer cells or the tumour microenvironment. Such alterations in the levels of specific tRNAs have been shown to causally impact cancer progression, including metastasis. Moreover, sequencing methods have identified tRNA-derived small RNAs that influence various aspects of cancer progression, such as cell proliferation and invasion, and could serve as diagnostic and prognostic biomarkers or putative therapeutic targets in various cancers. Finally, there is accumulating evidence, including from genetic models, that specific tRNA synthetases — the enzymes responsible for charging tRNAs with amino acids — can either promote or suppress tumour formation. In this Review, we provide an overview of how deregulation of tRNAs influences cancer formation and progression.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hoagland, M. B., Zamecnik, P. C. & Stephenson, M. L. Intermediate reactions in protein biosynthesis. Biochim. Biophys. Acta 24, 215–216 (1957).
Ogata, K. & Nohara, H. The possible role of the ribonucleic acid (RNA) of the pH 5 enzyme in amino acid activation. Biochim. Biophys. Acta 25, 659–660 (1957).
Holley, R. W. An alanine-dependent, ribonuclease-inhibited conversion of AMP to ATP, and its possible relationship to protein synthesis. J. Am. Chem. Soc. 79, 658–662 (1957). This study, together with Hoagland et al. (1957) and Ogata et al. (1957), is one of the original studies that discovered tRNAs.
Söll, D. & RajBhandary, U. L. tRNA: Structure, Biosynthesis, and Function (Wiley, 1995).
Suzuki, T. The expanding world of tRNA modifications and their disease relevance. Nat. Rev. Mol. Cell Biol. 22, 375–392 (2021).
Suzuki, T., Nagao, A. & Suzuki, T. Human mitochondrial tRNAs: biogenesis, function, structural aspects, and diseases. Annu. Rev. Genet. 45, 299–329 (2011). This is a comprehensive review on human mt-tRNAs.
Orellana, E. A., Siegal, E. & Gregory, R. I. tRNA dysregulation and disease. Nat. Rev. Genet. 23, 651–664 (2022).
Holley, R. W. et al. Structure of a ribonucleic acid. Science 147, 1462–1465 (1965). This paper describes the first sequence of a tRNA.
Kim, S. H. et al. Three-dimensional tertiary structure of yeast phenylalanine transfer RNA. Science 185, 435–440 (1974).
Cramer, F., Erdmann, V. A., von der Haar, F. & Schlimme, E. Structure and reactivity of tRNA. J. Cell Physiol. 74, 163–178 (1969).
Ramsay, E. P. & Vannini, A. Structural rearrangements of the RNA polymerase III machinery during tRNA transcription initiation. Biochim. Biophys. Acta Gene Regul. Mech. 1861, 285–294 (2018).
Phizicky, E. M. & Hopper, A. K. tRNA biology charges to the front. Genes Dev. 24, 1832–1860 (2010).
Rak, R. et al. Dynamic changes in tRNA modifications and abundance during T cell activation. Proc. Natl Acad. Sci. USA https://doi.org/10.1073/pnas.2106556118 (2021).
Torrent, M., Chalancon, G., de Groot, N. S., Wuster, A. & Madan Babu, M. Cells alter their tRNA abundance to selectively regulate protein synthesis during stress conditions. Sci. Signal. 11, eaat6409 (2018).
Dittmar, K. A., Goodenbour, J. M. & Pan, T. Tissue-specific differences in human transfer RNA expression. PLoS Genet. 2, e221 (2006).
Pinkard, O., McFarland, S., Sweet, T. & Coller, J. Quantitative tRNA-sequencing uncovers metazoan tissue-specific tRNA regulation. Nat. Commun. 11, 4104 (2020).
Kimura, S., Dedon, P. C. & Waldor, M. K. Comparative tRNA sequencing and RNA mass spectrometry for surveying tRNA modifications. Nat. Chem. Biol. 16, 964–972 (2020).
Gingold, H. et al. A dual program for translation regulation in cellular proliferation and differentiation. Cell 158, 1281–1292 (2014). This paper classifies ‘proliferation-specific’ and ‘differentiation-specific’ tRNAs.
Oler, A. J. et al. Human RNA polymerase III transcriptomes and relationships to Pol II promoter chromatin and enhancer-binding factors. Nat. Struct. Mol. Biol. 17, 620–628 (2010).
Barski, A. et al. Pol II and its associated epigenetic marks are present at Pol III-transcribed noncoding RNA genes. Nat. Struct. Mol. Biol. 17, 629–634 (2010).
Goodarzi, H. et al. Modulated expression of specific tRNAs drives gene expression and cancer progression. Cell 165, 1416–1427 (2016).
Boccaletto, P. et al. MODOMICS: a database of RNA modification pathways. 2021 update. Nucleic Acids Res. 50, D231–D235 (2022).
Zhang, W., Foo, M., Eren, A. M. & Pan, T. tRNA modification dynamics from individual organisms to metaepitranscriptomics of microbiomes. Mol. Cell 82, 891–906 (2022). This article includes a systematic analysis of current tRNA profiling methods.
Chujo, T. & Tomizawa, K. Human transfer RNA modopathies: diseases caused by aberrations in transfer RNA modifications. FEBS J. 288, 7096–7122 (2021).
Barbieri, I. & Kouzarides, T. Role of RNA modifications in cancer. Nat. Rev. Cancer 20, 303–322 (2020).
Clark, W. C., Evans, M. E., Dominissini, D., Zheng, G. & Pan, T. tRNA base methylation identification and quantification via high-throughput sequencing. RNA 22, 1771–1784 (2016).
Crick, F. H. Codon–anticodon pairing: the wobble hypothesis. J. Mol. Biol. 19, 548–555 (1966).
Chan, P. P. & Lowe, T. M. GtRNAdb 2.0: an expanded database of transfer RNA genes identified in complete and draft genomes. Nucleic Acids Res. 44, D184–D189 (2016).
Chan, P. P., Lin, B. Y., Mak, A. J. & Lowe, T. M. tRNAscan-SE 2.0: improved detection and functional classification of transfer RNA genes. Nucleic Acids Res. 49, 9077–9096 (2021). This resource paper, together with Chan et al. (2016), describes the GtRNAdb database of predicted tRNA sequences.
Pavon-Eternod, M. et al. tRNA over-expression in breast cancer and functional consequences. Nucleic Acids Res. 37, 7268–7280 (2009). This paper is one of the first to attempt global profiling of tRNA levels in cancer using microarrays.
Pavon-Eternod, M., Gomes, S., Rosner, M. R. & Pan, T. Overexpression of initiator methionine tRNA leads to global reprogramming of tRNA expression and increased proliferation in human epithelial cells. RNA 19, 461–466 (2013).
Earnest-Noble, L. B. et al. Two isoleucyl tRNAs that decode synonymous codons divergently regulate breast cancer metastatic growth by controlling translation of proliferation-regulating genes. Nat. Cancer 3, 1484–1497 (2022).
Taylor, M. W., Granger, G. A., Buck, C. A. & Holland, J. J. Similarities and differences among specific tRNA’s in mammalian tissues. Proc. Natl Acad. Sci. USA 57, 1712–1719 (1967).
Gallo, R. C. Transfer RNA’s in human leukemia. J. Cell Physiol. 74, 149–153 (1969).
Baliga, B. S., Borek, E., Weinstein, I. B. & Srinivasan, P. R. Differences in the transfer RNA’s of normal liver and Novikoff hepatoma. Proc. Natl Acad. Sci. USA 62, 899–905 (1969).
Sonenberg, N. & Hinnebusch, A. G. Regulation of translation initiation in eukaryotes: mechanisms and biological targets. Cell 136, 731–745 (2009).
Birch, J. et al. The initiator methionine tRNA drives cell migration and invasion leading to increased metastatic potential in melanoma. Biol. Open 5, 1371–1379 (2016).
Ruggero, D. Translational control in cancer etiology. Cold Spring Harb. Perspect. Biol. 5, a012336 (2013).
Silvera, D., Formenti, S. C. & Schneider, R. J. Translational control in cancer. Nat. Rev. Cancer 10, 254–266 (2010).
Clarke, C. J. et al. The initiator methionine tRNA drives secretion of type II collagen from stromal fibroblasts to promote tumor growth and angiogenesis. Curr. Biol. 26, 755–765 (2016).
Yang, J. et al. SOX4-mediated repression of specific tRNAs inhibits proliferation of human glioblastoma cells. Proc. Natl Acad. Sci. USA 117, 5782–5790 (2020).
Wang, B. et al. miR-34a directly targets tRNAiMet precursors and affects cellular proliferation, cell cycle, and apoptosis. Proc. Natl Acad. Sci. USA 115, 7392–7397 (2018).
Shigematsu, M. et al. YAMAT-seq: an efficient method for high-throughput sequencing of mature transfer RNAs. Nucleic Acids Res. 45, e70 (2017).
Behrens, A., Rodschinka, G. & Nedialkova, D. D. High-resolution quantitative profiling of tRNA abundance and modification status in eukaryotes by mim-tRNAseq. Mol. Cell 81, 1802–1815.e7 (2021).
Gogakos, T. et al. Characterizing expression and processing of precursor and mature human tRNAs by hydro-tRNAseq and PAR-CLIP. Cell Rep. 20, 1463–1475 (2017).
Lucas, M. C. et al. Quantitative analysis of tRNA abundance and modifications by nanopore RNA sequencing. Nat. Biotechnol. https://doi.org/10.1038/s41587-023-01743-6 (2023).
Iben, J. R. & Maraia, R. J. tRNA gene copy number variation in humans. Gene 536, 376–384 (2014).
Darrow, E. M. & Chadwick, B. P. A novel tRNA variable number tandem repeat at human chromosome 1q23.3 is implicated as a boundary element based on conservation of a CTCF motif in mouse. Nucleic Acids Res. 42, 6421–6435 (2014).
Parisien, M., Wang, X. & Pan, T. Diversity of human tRNA genes from the 1000-Genomes Project. RNA Biol. 10, 1853–1867 (2014).
Berg, M. D. et al. Targeted sequencing reveals expanded genetic diversity of human transfer RNAs. RNA Biol. 16, 1574–1585 (2019).
Thornlow, B. P. et al. Transfer RNA genes experience exceptionally elevated mutation rates. Proc. Natl Acad. Sci. USA 115, 8996–9001 (2018).
Ishimura, R. et al. Ribosome stalling induced by mutation of a CNS-specific tRNA causes neurodegeneration. Science 345, 455–459 (2014). This is one of the first reports of a tRNA mutation causing a tissue-specific phenotype in a mouse model.
Schoenmakers, E. et al. Mutation in human selenocysteine transfer RNA selectively disrupts selenoprotein synthesis. J. Clin. Invest. 126, 992–996 (2016).
Santos, M. et al. Codon misreading tRNAs promote tumor growth in mice. RNA Biol. 15, 773–786 (2018).
Doran, J. L., Bingle, W. H. & Roy, K. L. Two human genes encoding tRNA. Gene 65, 329–336 (1988).
Sprague, K. U., Larson, D. & Morton, D. 5′ Flanking sequence signals are required for activity of silkworm alanine tRNA genes in homologous in vitro transcription systems. Cell 22, 171–178 (1980).
Park, J. L. et al. Epigenetic regulation of noncoding RNA transcription by mammalian RNA polymerase III. Epigenomics 9, 171–187 (2017).
White, R. J. Transcription by RNA polymerase III: more complex than we thought. Nat. Rev. Genet. 12, 459–463 (2011).
Larminie, C. G. et al. Mechanistic analysis of RNA polymerase III regulation by the retinoblastoma protein. EMBO J. 16, 2061–2071 (1997).
Cairns, C. A. & White, R. J. p53 is a general repressor of RNA polymerase III transcription. EMBO J. 17, 3112–3123 (1998).
Crighton, D. et al. p53 represses RNA polymerase III transcription by targeting TBP and inhibiting promoter occupancy by TFIIIB. EMBO J. 22, 2810–2820 (2003).
Gomez-Roman, N., Grandori, C., Eisenman, R. N. & White, R. J. Direct activation of RNA polymerase III transcription by c-Myc. Nature 421, 290–294 (2003).
Kenneth, N. S. et al. TRRAP and GCN5 are used by c-Myc to activate RNA polymerase III transcription. Proc. Natl Acad. Sci. USA 104, 14917–14922 (2007).
Willis, I. M. & Moir, R. D. Signaling to and from the RNA polymerase III transcription and processing machinery. Annu. Rev. Biochem. 87, 75–100 (2018).
Raha, D. et al. Close association of RNA polymerase II and many transcription factors with Pol III genes. Proc. Natl Acad. Sci. USA 107, 3639–3644 (2010).
Zhang, J. et al. SOX4 inhibits GBM cell growth and induces G0/G1 cell cycle arrest through Akt–p53 axis. BMC Neurol. 14, 207 (2014).
Smith, D. K., Yang, J., Liu, M. L. & Zhang, C. L. Small molecules modulate chromatin accessibility to promote NEUROG2-mediated fibroblast-to-neuron reprogramming. Stem Cell Rep. 7, 955–969 (2016).
Lee, H., Goodarzi, H., Tavazoie, S. F. & Alarcon, C. R. TMEM2 is a SOX4-regulated gene that mediates metastatic migration and invasion in breast cancer. Cancer Res. 76, 4994–5005 (2016).
Vervoort, S. J., van Boxtel, R. & Coffer, P. J. The role of SRY-related HMG box transcription factor 4 (SOX4) in tumorigenesis and metastasis: friend or foe? Oncogene 32, 3397–3409 (2013).
Besser, D. et al. DNA methylation inhibits transcription by RNA polymerase III of a tRNA gene, but not of a 5S rRNA gene. FEBS Lett. 269, 358–362 (1990).
Bartke, T. et al. Nucleosome-interacting proteins regulated by DNA and histone methylation. Cell 143, 470–484 (2010).
Rossello-Tortella, M., Bueno-Costa, A., Martinez-Verbo, L., Villanueva, L. & Esteller, M. DNA methylation-associated dysregulation of transfer RNA expression in human cancer. Mol. Cancer 21, 48 (2022).
Tavtigian, S. V. et al. A candidate prostate cancer susceptibility gene at chromosome 17p. Nat. Genet. 27, 172–180 (2001).
Xu, B., Tong, N., Li, J. M., Zhang, Z. D. & Wu, H. F. ELAC2 polymorphisms and prostate cancer risk: a meta-analysis based on 18 case-control studies. Prostate Cancer Prostatic Dis. 13, 270–277 (2010).
Lubas, M., Chlebowski, A., Dziembowski, A. & Jensen, T. H. Biochemistry and function of RNA exosomes. Enzymes 31, 1–30 (2012).
Kadaba, S. et al. Nuclear surveillance and degradation of hypomodified initiator tRNAMet in S. cerevisiae. Genes Dev. 18, 1227–1240 (2004).
Whipple, J. M., Lane, E. A., Chernyakov, I., D’Silva, S. & Phizicky, E. M. The yeast rapid tRNA decay pathway primarily monitors the structural integrity of the acceptor and T-stems of mature tRNA. Genes Dev. 25, 1173–1184 (2011).
Alexandrov, A. et al. Rapid tRNA decay can result from lack of nonessential modifications. Mol. Cell 21, 87–96 (2006).
De Zoysa, T. & Phizicky, E. M. Hypomodified tRNA in evolutionarily distant yeasts can trigger rapid tRNA decay to activate the general amino acid control response, but with different consequences. PLoS Genet. 16, e1008893 (2020).
Wilusz, J. E., Whipple, J. M., Phizicky, E. M. & Sharp, P. A. tRNAs marked with CCACCA are targeted for degradation. Science 334, 817–821 (2011).
Orellana, E. A. et al. METTL1-mediated m7G modification of Arg-TCT tRNA drives oncogenic transformation. Mol. Cell 81, 3323–3338.e14 (2021).
Han, H. et al. N7-methylguanosine tRNA modification promotes esophageal squamous cell carcinoma tumorigenesis via the RPTOR/ULK1/autophagy axis. Nat. Commun. 13, 1478 (2022).
Chen, J. et al. Aberrant translation regulated by METTL1/WDR4-mediated tRNA N7-methylguanosine modification drives head and neck squamous cell carcinoma progression. Cancer Commun. 42, 223–244 (2022).
Dai, Z. et al. N7-methylguanosine tRNA modification enhances oncogenic mRNA translation and promotes intrahepatic cholangiocarcinoma progression. Mol. Cell 81, 3339–3355.e8 (2021).
Chen, Z. et al. METTL1 promotes hepatocarcinogenesis via m7G tRNA modification-dependent translation control. Clin. Transl. Med. 11, e661 (2021).
Pandolfini, L. et al. METTL1 promotes let-7 microRNA processing via m7G methylation. Mol. Cell 74, 1278–1290.e9 (2019).
Cheng, W., Gao, A., Lin, H. & Zhang, W. Novel roles of METTL1/WDR4 in tumor via m7G methylation. Mol. Ther. Oncolytics 26, 27–34 (2022).
Girstmair, H. et al. Depletion of cognate charged transfer RNA causes translational frameshifting within the expanded CAG stretch in huntingtin. Cell Rep. 3, 148–159 (2013).
Guzy, R. D. et al. Mitochondrial complex III is required for hypoxia-induced ROS production and cellular oxygen sensing. Cell Metab. 1, 401–408 (2005).
Bell, E. L. et al. The Qo site of the mitochondrial complex III is required for the transduction of hypoxic signaling via reactive oxygen species production. J. Cell Biol. 177, 1029–1036 (2007).
Nawrot, B., Sochacka, E. & Duchler, M. tRNA structural and functional changes induced by oxidative stress. Cell Mol. Life Sci. 68, 4023–4032 (2011).
Endres, L. et al. Alkbh8 regulates selenocysteine-protein expression to protect against reactive oxygen species damage. PLoS ONE 10, e0131335 (2015).
Chan, C. T. et al. Reprogramming of tRNA modifications controls the oxidative stress response by codon-biased translation of proteins. Nat. Commun. 3, 937 (2012).
Netzer, N. et al. Innate immune and chemically triggered oxidative stress modifies translational fidelity. Nature 462, 522–526 (2009).
Schwenzer, H. et al. Oxidative stress triggers selective tRNA retrograde transport in human cells during the integrated stress response. Cell Rep. 26, 3416–3428.e5 (2019).
Thompson, D. M., Lu, C., Green, P. J. & Parker, R. tRNA cleavage is a conserved response to oxidative stress in eukaryotes. RNA 14, 2095–2103 (2008).
Goodarzi, H. et al. Endogenous tRNA-derived fragments suppress breast cancer progression via YBX1 displacement. Cell 161, 790–802 (2015).
Huh, D. et al. A stress-induced tyrosine-tRNA depletion response mediates codon-based translational repression and growth suppression. EMBO J. 40, e106696 (2021).
Yamasaki, S., Ivanov, P., Hu, G. F. & Anderson, P. Angiogenin cleaves tRNA and promotes stress-induced translational repression. J. Cell Biol. 185, 35–42 (2009).
Masson, G. R. Towards a model of GCN2 activation. Biochem. Soc. Trans. 47, 1481–1488 (2019).
Pataskar, A. et al. Tryptophan depletion results in tryptophan-to-phenylalanine substitutants. Nature 603, 721–727 (2022).
Hsu, D. J. et al. Arginine limitation drives a directed codon-dependent DNA sequence evolution response in colorectal cancer cells. Sci. Adv. 9, eade9120 (2023).
Passarelli, M. C. et al. Leucyl-tRNA synthetase is a tumour suppressor in breast cancer and regulates codon-dependent translation dynamics. Nat. Cell Biol. 24, 307–315 (2022).
Turvey, A. K., Horvath, G. A. & Cavalcanti, A. R. O. Aminoacyl-tRNA synthetases in human health and disease. Front. Physiol. 13, 1029218 (2022).
Hyeon, D. Y. et al. Evolution of the multi-tRNA synthetase complex and its role in cancer. J. Biol. Chem. 294, 5340–5351 (2019).
Wang, J. & Yang, X. L. Novel functions of cytoplasmic aminoacyl-tRNA synthetases shaping the hallmarks of cancer. Enzymes 48, 397–423 (2020).
Sung, Y., Yoon, I., Han, J. M. & Kim, S. Functional and pathologic association of aminoacyl-tRNA synthetases with cancer. Exp. Mol. Med. 54, 553–566 (2022).
Guo, M. & Schimmel, P. Essential nontranslational functions of tRNA synthetases. Nat. Chem. Biol. 9, 145–153 (2013).
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Wang, J. et al. Multi-omics database analysis of aminoacyl-tRNA synthetases in cancer. Genes 11, 1384 (2020).
Thandapani, P. et al. Valine tRNA levels and availability regulate complex I assembly in leukaemia. Nature 601, 428–433 (2022).
Su, Z., Wilson, B., Kumar, P. & Dutta, A. Noncanonical roles of tRNAs: tRNA fragments and beyond. Annu. Rev. Genet. 54, 47–69 (2020).
Borek, E. et al. High turnover rate of transfer RNA in tumor tissue. Cancer Res. 37, 3362–3366 (1977).
Speer, J., Gehrke, C. W., Kuo, K. C., Waalkes, T. P. & Borek, E. tRNA breakdown products as markers for cancer. Cancer 44, 2120–2123 (1979).
Kumar, P., Anaya, J., Mudunuri, S. B. & Dutta, A. Meta-analysis of tRNA derived RNA fragments reveals that they are evolutionarily conserved and associate with AGO proteins to recognize specific RNA targets. BMC Biol. 12, 78 (2014).
Pliatsika, V. et al. MINTbase v2.0: a comprehensive database for tRNA-derived fragments that includes nuclear and mitochondrial fragments from all the cancer genome atlas projects. Nucleic Acids Res. 46, D152–D159 (2018).
Yao, D. et al. OncotRF: an online resource for exploration of tRNA-derived fragments in human cancers. RNA Biol. 17, 1081–1091 (2020).
Lee, S. R. & Collins, K. Starvation-induced cleavage of the tRNA anticodon loop in Tetrahymena thermophila. J. Biol. Chem. 280, 42744–42749 (2005).
Tao, E. W., Cheng, W. Y., Li, W. L., Yu, J. & Gao, Q. Y. tiRNAs: a novel class of small noncoding RNAs that helps cells respond to stressors and plays roles in cancer progression. J. Cell Physiol. 235, 683–690 (2020).
Fu, H. et al. Stress induces tRNA cleavage by angiogenin in mammalian cells. FEBS Lett. 583, 437–442 (2009).
Donovan, J., Rath, S., Kolet-Mandrikov, D. & Korennykh, A. Rapid RNase L-driven arrest of protein synthesis in the dsRNA response without degradation of translation machinery. RNA 23, 1660–1671 (2017).
Nechooshtan, G., Yunusov, D., Chang, K. & Gingeras, T. R. Processing by RNase 1 forms tRNA halves and distinct Y RNA fragments in the extracellular environment. Nucleic Acids Res. 48, 8035–8049 (2020).
Lee, Y. S., Shibata, Y., Malhotra, A. & Dutta, A. A novel class of small RNAs: tRNA-derived RNA fragments (tRFs). Genes Dev. 23, 2639–2649 (2009).
Oberbauer, V. & Schaefer, M. R. tRNA-derived small RNAs: biogenesis, modification, function and potential impact on human disease development. Genes 9, 607 (2018).
Guzzi, N. & Bellodi, C. Novel insights into the emerging roles of tRNA-derived fragments in mammalian development. RNA Biol. 17, 1214–1222 (2020).
Fagan, S. G., Helm, M. & Prehn, J. H. M. tRNA-derived fragments: a new class of non-coding RNA with key roles in nervous system function and dysfunction. Prog. Neurobiol. 205, 102118 (2021).
Thompson, D. M. & Parker, R. Stressing out over tRNA cleavage. Cell 138, 215–219 (2009).
Maute, R. L. et al. tRNA-derived microRNA modulates proliferation and the DNA damage response and is down-regulated in B cell lymphoma. Proc. Natl Acad. Sci. USA 110, 1404–1409 (2013).
Balatti, V. et al. TCL1 targeting miR-3676 is codeleted with tumor protein p53 in chronic lymphocytic leukemia. Proc. Natl Acad. Sci. USA 112, 2169–2174 (2015).
Pekarsky, Y. et al. Dysregulation of a family of short noncoding RNAs, tsRNAs, in human cancer. Proc. Natl Acad. Sci. USA 113, 5071–5076 (2016).
Wang, H. et al. The tRNA-derived fragment tRF-24-V29K9UV3IU functions as a miRNA-like RNA to prevent gastric cancer progression by inhibiting GPR78 expression. J. Oncol. 2022, 8777697 (2022).
Dong, D. D., Zhou, H. & Li, G. GPR78 promotes lung cancer cell migration and metastasis by activation of Gαq-Rho GTPase pathway. BMB Rep. 49, 623–628 (2016).
Shi, Z. & Barna, M. Translating the genome in time and space: specialized ribosomes, RNA regulons, and RNA-binding proteins. Annu. Rev. Cell Dev. Biol. 31, 31–54 (2015).
Falconi, M. et al. A novel 3′-tRNAGlu-derived fragment acts as a tumor suppressor in breast cancer by targeting nucleolin. FASEB J. 33, 13228–13240 (2019).
Guzzi, N. et al. Pseudouridine-modified tRNA fragments repress aberrant protein synthesis and predict leukaemic progression in myelodysplastic syndrome. Nat. Cell Biol. 24, 299–306 (2022).
Guzzi, N. et al. Pseudouridylation of tRNA-derived fragments steers translational control in stem cells. Cell 173, 1204–1216.e26 (2018).
Pan, L. et al. Inflammatory cytokine-regulated tRNA-derived fragment tRF-21 suppresses pancreatic ductal adenocarcinoma progression. J. Clin. Invest. 131, e148130 (2021).
Tong, L. et al. The tRNA-derived fragment-3017A promotes metastasis by inhibiting NELL2 in human gastric cancer. Front. Oncol. 10, 570916 (2020).
Zhang, F. et al. A 3′-tRNA-derived fragment enhances cell proliferation, migration and invasion in gastric cancer by targeting FBXO47. Arch. Biochem. Biophys. 690, 108467 (2020).
Han, L. et al. A 5′-tRNA halve, tiRNA-Gly promotes cell proliferation and migration via binding to RBM17 and inducing alternative splicing in papillary thyroid cancer. J. Exp. Clin. Cancer Res. 40, 222 (2021).
Liu, X. et al. A pro-metastatic tRNA fragment drives nucleolin oligomerization and stabilization of its bound metabolic mRNAs. Mol. Cell 82, 2604–2617.e8 (2022).
Lampson, B. L. et al. Rare codons regulate KRas oncogenesis. Curr. Biol. 23, 70–75 (2013).
Pershing, N. L. et al. Rare codons capacitate Kras-driven de novo tumorigenesis. J. Clin. Invest. 125, 222–233 (2015).
Li, S. & Counter, C. M. Signaling levels mold the RAS mutation tropism of urethane. eLife 10, e67172 (2021).
Ali, M. et al. Codon bias imposes a targetable limitation on KRAS-driven therapeutic resistance. Nat. Commun. 8, 15617 (2017).
Keller, T. L. et al. Halofuginone and other febrifugine derivatives inhibit prolyl-tRNA synthetase. Nat. Chem. Biol. 8, 311–317 (2012).
Kim, Y. et al. Aminoacyl-tRNA synthetase inhibition activates a pathway that branches from the canonical amino acid response in mammalian cells. Proc. Natl Acad. Sci. USA 117, 8900–8911 (2020).
Kim, S. H., Bae, S. & Song, M. Recent development of aminoacyl-tRNA synthetase inhibitors for human diseases: a future perspective. Biomolecules 10, 1625 (2020).
Zuko, A. et al. tRNA overexpression rescues peripheral neuropathy caused by mutations in tRNA synthetase. Science 373, 1161–1166 (2021).
Wang, J. et al. AAV-delivered suppressor tRNA overcomes a nonsense mutation in mice. Nature 604, 343–348 (2022).
Albers, S. et al. Engineered tRNAs suppress nonsense mutations in cells and in vivo. Nature 618, 842–848 (2023).
Chang, J. C., Temple, G. F., Trecartin, R. F. & Kan, Y. W. Suppression of the nonsense mutation in homozygous β0 thalassaemia. Nature 281, 602–603 (1979).
Winkle, M., El-Daly, S. M., Fabbri, M. & Calin, G. A. Noncoding RNA therapeutics — challenges and potential solutions. Nat. Rev. Drug Discov. 20, 629–651 (2021).
Papadimitriou, M. A. et al. tRNA-derived fragments (tRFs) in bladder cancer: increased 5′-tRF-LysCTT results in disease early progression and patients’ poor treatment outcome. Cancers 12, 3661 (2020).
Wang, J. et al. Circulating tRNA-derived small RNAs (tsRNAs) signature for the diagnosis and prognosis of breast cancer. NPJ Breast Cancer 7, 4 (2021).
Wang, X. et al. Identification of tRNA-derived fragments expression profile in breast cancer tissues. Curr. Genomics 20, 199–213 (2019).
Sun, C. et al. tRNA-derived fragments as novel predictive biomarkers for trastuzumab-resistant breast cancer. Cell. Physiol. Biochem. 49, 419–431 (2018).
Veneziano, D. et al. Dysregulation of different classes of tRNA fragments in chronic lymphocytic leukemia. Proc. Natl Acad. Sci. USA 116, 24252–24258 (2019).
Nientiedt, M. et al. Identification of aberrant tRNA-halves expression patterns in clear cell renal cell carcinoma. Sci. Rep. 6, 37158 (2016).
Wu, Y. et al. 5′-tRF-GlyGCC: a tRNA-derived small RNA as a novel biomarker for colorectal cancer diagnosis. Genome Med. 13, 20 (2021).
Zhu, Y. et al. Comprehensive analysis of a tRNA-derived small RNA in colorectal cancer. Front. Oncol. 11, 701440 (2021).
Shao, Y. et al. tRF-Leu-CAG promotes cell proliferation and cell cycle in non-small cell lung cancer. Chem. Biol. Drug Des. 90, 730–738 (2017).
Li, J., Cao, C., Fang, L. & Yu, W. Serum transfer RNA-derived fragment tRF-31-79MP9P9NH57SD acts as a novel diagnostic biomarker for non-small cell lung cancer. J. Clin. Lab. Anal. https://doi.org/10.1002/jcla.24492 (2022).
Huang, Y. et al. Elucidating the role of serum tRF-31-U5YKFN8DYDZDD as a novel diagnostic biomarker in gastric cancer (GC). Front. Oncol. 11, 723753 (2021).
Panoutsopoulou, K. et al. tRNAGlyGCC-derived internal fragment (i-tRF-GlyGCC) in ovarian cancer treatment outcome and progression. Cancers 14, 24 (2021).
Olvedy, M. et al. A comprehensive repertoire of tRNA-derived fragments in prostate cancer. Oncotarget 7, 24766–24777 (2016).
Rich, A. & RajBhandary, U. L. Transfer RNA: molecular structure, sequence, and properties. Annu. Rev. Biochem. 45, 805–860 (1976).
Crothers, D. M., Seno, T. & Söll, D. G. Is there a discriminator site in transfer RNA? Proc. Natl Acad. Sci. USA 69, 3063–3067 (1972).
Sprinzl, M., Horn, C., Brown, M., Ioudovitch, A. & Steinberg, S. Compilation of tRNA sequences and sequences of tRNA genes. Nucleic Acids Res. 26, 148–153 (1998).
Shi, H. & Moore, P. B. The crystal structure of yeast phenylalanine tRNA at 1.93 Å resolution: a classic structure revisited. RNA 6, 1091–1105 (2000).
Galli, G., Hofstetter, H. & Birnstiel, M. L. Two conserved sequence blocks within eukaryotic tRNA genes are major promoter elements. Nature 294, 626–631 (1981).
Hofstetter, H., Kressmann, A. & Birnstiel, M. L. A split promoter for a eucaryotic tRNA gene. Cell 24, 573–585 (1981).
Takaku, H., Minagawa, A., Takagi, M. & Nashimoto, M. A candidate prostate cancer susceptibility gene encodes tRNA 3′ processing endoribonuclease. Nucleic Acids Res. 31, 2272–2278 (2003).
Rideout, E. J., Marshall, L. & Grewal, S. S. Drosophila RNA polymerase III repressor Maf1 controls body size and developmental timing by modulating tRNAiMet synthesis and systemic insulin signaling. Proc. Natl Acad. Sci. USA 109, 1139–1144 (2012).
Chu, A. et al. Large-scale profiling of microRNAs for the cancer genome atlas. Nucleic Acids Res. 44, e3 (2016).
Cozen, A. E. et al. ARM-seq: AlkB-facilitated RNA methylation sequencing reveals a complex landscape of modified tRNA fragments. Nat. Methods 12, 879–884 (2015).
Zheng, G. et al. Efficient and quantitative high-throughput tRNA sequencing. Nat. Methods 12, 835–837 (2015).
Smith, A. M., Abu-Shumays, R., Akeson, M. & Bernick, D. L. Capture, unfolding, and detection of individual tRNA molecules using a nanopore device. Front. Bioeng. Biotechnol. 3, 91 (2015).
Thomas, N. K. et al. Direct nanopore sequencing of individual full length tRNA strands. ACS Nano 15, 16642–16653 (2021).
Hughes, L. A. et al. Copy number variation in tRNA isodecoder genes impairs mammalian development and balanced translation. Nat. Commun. 14, 2210 (2023). This is one of the first studies to systematically generate single and combinatorial deletions of related tRNA isodecoder genes and assess their consequences at an organismal level in mice.
Koukuntla, R., Ramsey, W. J., Young, W. B. & Link, C. J. U6 promoter-enhanced GlnUAG suppressor tRNA has higher suppression efficacy and can be stably expressed in 293 cells. J. Gene Med. 15, 93–101 (2013).
Acknowledgements
The authors thank the members of the Tavazoie laboratory for critically reading the manuscript and for their helpful suggestions and comments. The authors apologize to all the scientists whose work could not be cited owing to space limitations. S.F.T. and A.M.P. were supported by grants from the National Cancer Institute of the National Institutes of Health under award numbers R01CA257153, R35CA274446 and U54CA261701, as well as the Black Family Metastasis Center and the Breast Cancer Research Foundation. Molecular graphics were performed with UCSF ChimeraX, developed by the Resource for Biocomputing, Visualization and Informatics at the University of California, San Francisco, with support from National Institutes of Health R01-GM129325 and the Office of Cyber Infrastructure and Computational Biology, National Institute of Allergy and Infectious Diseases.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Cancer thanks Pierre Close and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
RCSB Protein Data Bank: https://www.rcsb.org/
UCSF ChimeraX software: https://www.rbvi.ucsf.edu/chimerax
Glossary
- Argonaute 2 (AGO2) cleavage assay
-
An in vitro assay that uses recombinant AGO2 to show specific cleavage of target oligonucleotides.
- Epistatic interactions
-
Interactions between two or more genes that influence a particular phenotype.
- Lineage-negative haematopoietic stem and progenitor cells
-
Immature blood cells that lack surface protein markers found on mature blood cells.
- Rare codons
-
Codons found with the lowest frequency in genes compared with synonymous codons in the same group.
- Ribosome
-
An essential ribonucleoprotein complex that performs protein synthesis in cells.
- Ribosome profiling
-
A technique that provides a snapshot of all ribosome-bound messenger RNAs in cells with codon-level resolution.
- RNA polymerase III
-
(Pol III). An RNA polymerase complex that translates short, non-coding RNAs, including transfer RNAs, 5S ribosomal RNA, U6 small nuclear RNA, vault RNA and other small RNAs.
- tRNA misacylation
-
The charging of a transfer RNA with the wrong amino acid, not the amino acid cognate to the anticodon of the transfer RNA.
- Wobble pairing
-
Non-Watson–Crick base pairing of the first anticodon nucleoside: inosine can pair with adenosine, uridine or cytidine; guanosine can pair with cytidine or uridine; uridine can pair with adenosine or guanosine.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pinzaru, A.M., Tavazoie, S.F. Transfer RNAs as dynamic and critical regulators of cancer progression. Nat Rev Cancer 23, 746–761 (2023). https://doi.org/10.1038/s41568-023-00611-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41568-023-00611-4