Abstract
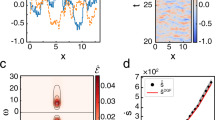
The circadian clock and the cell cycle are two biological oscillatory processes that coexist within individual cells. These two oscillators were found to interact, which can lead to their synchronization. Here, we develop a method to identify a low-dimensional stochastic model of the coupled system directly from time-lapse imaging in single cells. In particular, we infer the coupling and nonlinear dynamics of the two oscillators from thousands of mouse and human single-cell fluorescence microscopy traces. This coupling predicts multiple phase-locked states showing different degrees of robustness against molecular fluctuations inherent to cellular-scale biological oscillators. For the 1:1 state, the predicted phase-shifts following period perturbations were validated experimentally. Moreover, the phase-locked states are temperature-independent and evolutionarily conserved from mouse to human, hinting at a common underlying dynamical mechanism. Finally, we detect a signature of the coupled dynamics in a physiological context, explaining why tissues with different proliferation states exhibited shifted circadian clock phases.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data supporting the figures and other findings of this study are available from the corresponding author on request.
Code availability
The code is available at https://c4science.ch/diffusion/9123/.
References
Nagoshi, E. et al. Circadian gene expression in individual fibroblasts: cell-autonomous and self-sustained oscillators pass time to daughter cells. Cell 119, 693–705 (2004).
Mermet, J., Yeung, J. & Naef, F. Systems chronobiology: global analysis of gene regulation in a 24-hour periodic world. Cold Spring Harb. Perspect. Biol. 9, https://doi.org/10.1101/cshperspect.a028720 (2017).
Hahn, A. T., Jones, J. T. & Meyer, T. Quantitative analysis of cell cycle phase durations and PC12 differentiation using fluorescent biosensors. Cell Cycle 8, 1044–1052 (2009).
Spencer, S. L. et al. The proliferation–quiescence decision is controlled by a bifurcation in CDK2 activity at mitotic exit. Cell 155, 369–383 (2013).
Bieler, J. et al. Robust synchronization of coupled circadian and cell cycle oscillators in single mammalian cells. Mol. Syst. Biol. 10, 739 (2014).
Feillet, C. et al. Phase locking and multiple oscillating attractors for the coupled mammalian clock and cell cycle. Proc. Natl Acad. Sci. USA 111, 9828–9833 (2014).
Matsuo, T. et al. Control mechanism of the circadian clock for timing of cell division in vivo. Science 302, 255–259 (2003).
Kowalska, E. et al. NONO couples the circadian clock to the cell cycle. Proc. Natl Acad. Sci. USA 110, 1592–1599 (2013).
Mori, T., Binder, B. & Johnson, C. H. Circadian gating of cell division in cyanobacteria growing with average doubling times of less than 24 hours. Proc. Natl Acad. Sci. USA 93, 10183–10188 (1996).
Yang, Q., Pando, B. F., Dong, G., Golden, S. S. & van Oudenaarden, A. Circadian gating of the cell cycle revealed in single cyanobacterial cells. Science 327, 1522–1526 (2010).
Matsu-Ura, T. et al. Intercellular coupling of the cell cycle and circadian clock in adult stem cell culture. Mol. Cell 64, 900–912 (2016).
Plikus, M. V. et al. Local circadian clock gates cell cycle progression of transient amplifying cells during regenerative hair cycling. Proc. Natl Acad. Sci. USA 110, E2106–E2115 (2013).
Gerard, C. & Goldbeter, A. Entrainment of the mammalian cell cycle by the circadian clock: modeling two coupled cellular rhythms. PLoS Comput. Biol. 8, e1002516 (2012).
Paijmans, J., Bosman, M., Ten Wolde, P. R. & Lubensky, D. K. Discrete gene replication events drive coupling between the cell cycle and circadian clocks. Proc. Natl Acad. Sci. USA 113, 4063–4068 (2016).
Shostak, A. et al. MYC/MIZ1-dependent gene repression inversely coordinates the circadian clock with cell cycle and proliferation. Nat. Commun. 7, 11807 (2016).
Rougemont, J. & Naef, F. Collective synchronization in populations of globally coupled phase oscillators with drifting frequencies. Phys. Rev. E 73, 011104 (2006).
Rabiner, L. R. & Juang, B.-H. An introduction to hidden Markov models. IEEE ASSP Mag. 3, 4–16 (1986).
Yeom, M., Pendergast, J. S., Ohmiya, Y. & Yamazaki, S. Circadian-independent cell mitosis in immortalized fibroblasts. Proc. Natl Acad. Sci. USA 107, 9665–9670 (2010).
Vollmers, C., Panda, S. & DiTacchio, L. A high-throughput assay for siRNA-based circadian screens in human U2OS cells. PloS One 3, e3457 (2008).
Maier, B. et al. A large-scale functional RNAi screen reveals a role for CK2 in the mammalian circadian clock. Genes Dev. 23, 708–718 (2009).
Nicolas, D., Zoller, B., Suter, D. M. & Naef, F. Modulation of transcriptional burst frequency by histone acetylation. Proc. Natl Acad. Sci. USA 115, 7153–7158 (2018).
Balsalobre, A. et al. Resetting of circadian time in peripheral tissues by glucocorticoid signaling. Science 289, 2344–2347 (2000).
Dibner, C., Schibler, U. & Albrecht, U. The mammalian circadian timing system: organization and coordination of central and peripheral clocks. Annu. Rev. Physiol. 72, 517–549 (2010).
Saini, C., Morf, J., Stratmann, M., Gos, P. & Schibler, U. Simulated body temperature rhythms reveal the phase-shifting behavior and plasticity of mammalian circadian oscillators. Genes Dev. 26, 567–580 (2012).
Hayes, O. et al. Cell confluency is as efficient as serum starvation for inducing arrest in the G0/G1 phase of the cell cycle in granulosa and fibroblast cells of cattle. Anim. Reprod. Sci. 87, 181–192 (2005).
Zhang, R., Lahens, N. F., Ballance, H. I., Hughes, M. E. & Hogenesch, J. B. A circadian gene expression atlas in mammals: implications for biology and medicine. Proc. Natl Acad. Sci. USA 111, 16219–16224 (2014).
Yeung, J. et al. Transcription factor activity rhythms and tissue-specific chromatin interactions explain circadian gene expression across organs. Genome Res. 28, 182–191 (2018).
Cermakian, N., Monaco, L., Pando, M. P., Dierich, A. & Sassone-Corsi, P. Altered behavioral rhythms and clock gene expression in mice with a targeted mutation in the Period1 gene. EMBO J. 20, 3967–3974 (2001).
Debruyne, J. P. et al. A clock shock: mouse CLOCK is not required for circadian oscillator function. Neuron 50, 465–477 (2006).
Granada, A. E. & Herzel, H. How to achieve fast entrainment? The timescale to synchronization. PloS One 4, e7057 (2009).
Heltberg, M., Kellogg, R. A., Krishna, S., Tay, S. & Jensen, M. H. Noise induces hopping between NF-κB entrainment modes. Cell Syst. 3, 532–539 e533 (2016).
Aryal, R. P. et al. Macromolecular assemblies of the mammalian circadian clock. Mol. Cell 67, 770–782.e6 (2017).
Ukai-Tadenuma, M. et al. Delay in feedback repression by cryptochrome 1 is required for circadian clock function. Cell 144, 268–281 (2011).
Mermet, J. et al. Clock-dependent chromatin topology modulates circadian transcription and behavior. Genes Dev. 32, 347–358 (2018).
Siepka, S. M. et al. Circadian mutant overtime reveals F-box protein FBXL3 regulation of cryptochrome and period gene expression. Cell 129, 1011–1023 (2007).
King, R. W., Deshaies, R. J., Peters, J. M. & Kirschner, M. W. How proteolysis drives the cell cycle. Science 274, 1652–1659 (1996).
Hastings, M. H., Maywood, E. S. & Brancaccio, M. Generation of circadian rhythms in the suprachiasmatic nucleus. Nat. Rev. Neurosci. 19, 453–469 (2018).
Mohawk, J. A., Green, C. B. & Takahashi, J. S. Central and peripheral circadian clocks in mammals. Annu. Rev. Neurosci. 35, 445–462 (2012).
Pikovsky, A. R. M. K. J. Synchronization (Cambridge University Press, 2001).
Shilts, J., Chen, G. & Hughey, J. J. Evidence for widespread dysregulation of circadian clock progression in human cancer. PeerJ 6, e4327 (2018).
Kelleher, F. C., Rao, A. & Maguire, A. Circadian molecular clocks and cancer. Cancer Lett. 342, 9–18 (2014).
Brunton, S. L., Proctor, J. L. & Kutz, J. N. Discovering governing equations from data by sparse identification of nonlinear dynamical systems. Proc. Natl Acad. Sci. USA 113, 3932–3937 (2016).
Soroldoni, D. et al. Genetic oscillations. A Doppler effect in embryonic pattern formation. Science 345, 222–225 (2014).
Dempster, A. P., Laird, N. M. & Rubin, D. B. Maximum likelihood from incomplete data via the EM algorithm. J. R. Stat. Soc. Ser. B 39, 1–38 (1977).
Green, P. J. On use of the EM for penalized likelihood estimation. J. R. Stat. Soc. Ser. B 52, 443–452 (1990).
Koksharov, M. I. & Ugarova, N. N. Approaches to engineer stability of beetle luciferases. Comput. Struct. Biotechnol. J. 2, e201209004 (2012).
Huang da, W., Sherman, B. T. & Lempicki, R. A. Bioinformatics enrichment tools: paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res. 37, 1–13 (2009).
Acknowledgements
We thank R. Cannavo for engineering the U2OS-Dual cell line and J. Bieler for initial analyses. We also thank F. Kuttler from the EPFL Biomolecular Screening Facility and L. Bozzo and J. Artacho from the EPFL Bioimaging and Optics Core Facility for assistance with the imaging. Fluorescence-activated cell sorting was performed at the EPFL Flow Cytometry Core Facility. This work was supported by the Swiss National Science Foundation grant 310030_173079 and the EPFL. E.R.P. was supported by a Canadian Institute of Health Research (CIHR 358808) and a SystemsX.ch Transition Postdoc Fellowship (51FSP0163584).
Author information
Authors and Affiliations
Contributions
C.D., E.R.P. and F.N. designed and participated in the study concept. C.D. and E.R.P. developed computational analysis tools. E.R.P. performed the experiments. C.D. and E.R.P. processed and analysed the experimental data. C.D., E.R.P. and F.N. interpreted the results. E.R.P. and F.N. acquired the funding. F.N. supervised the study. C.D., E.R.P. and F.N. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information: Nature Physics thanks Mogens Jensen, Joris Paijmans and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–5, methods and refs. 1–9.
Supplementary Table 1
Correlation analysis of tissue-specific phases and amplitudes with the mean transcript levels in mouse for every gene. Gene Ontology analyses for the top 200 positively and top 200 negatively correlated genes are provided for both phase and amplitude correlations.
Supplementary Video 1
Single-cell trajectories of circadian and cell-cycle phases cluster around a 1:1 phase-locked state.
Supplementary Video 2
Simulations of the deterministic model show phase-locking and quasiperiodicity.
Supplementary Video 3
Spectral analysis of the deterministic and stochastic simulations shows qualitative differences as a function of the cell-cycle period.
Rights and permissions
About this article
Cite this article
Droin, C., Paquet, E.R. & Naef, F. Low-dimensional dynamics of two coupled biological oscillators. Nat. Phys. 15, 1086–1094 (2019). https://doi.org/10.1038/s41567-019-0598-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41567-019-0598-1
This article is cited by
-
Circa-SCOPE: high-throughput live single-cell imaging method for analysis of circadian clock resetting
Nature Communications (2021)
-
Locked body clocks
Nature Physics (2019)