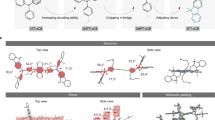
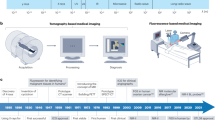
Abstract
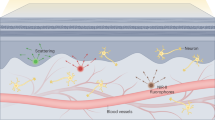
Owing to reduced light scattering and tissue autofluorescence, in vivo fluorescence imaging in the 1,000–3,000-nm near-infrared II (NIR-II) spectral range can afford non-invasive imaging at depths of millimetres within biological tissue. Infrared fluorescent probes labelled with antibodies or other targeting ligands also enable NIR-II molecular imaging at the single-cell level. Here we present recent developments in the design of fluorophores and probes emitting in the NIR-II window based on organic synthesis and nanoscience approaches. We also review advances in NIR-II wide-field and microscopy imaging modalities, with a focus on preclinical imaging and promising clinical translation case studies. Finally, we outline current issues and challenges for the wider adoption of NIR-II imaging in biomedical research and clinical imaging.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Genack, A. Z., Chabanov, A. A., Sebbah, P. & van Tiggelen, B. A. in Encyclopedia of Condensed Matter Physics (eds Bassani, F., Liedl, G. L. & Wyder, P.) 307–317 (Elsevier, 2005).
Shi, L. & Alfano, R. R. Deep Imaging in Tissue and Biomedical Materials: using Linear and Nonlinear Optical Methods (CRC Press, 2017).
Jacques, S. L. Optical properties of biological tissues: a review. Phys. Med. Biol. 58, R37–R61 (2013).
Hong, G., Antaris, A. L. & Dai, H. Near-infrared fluorophores for biomedical imaging. Nat. Biomed. Eng. 1, 0010 (2017).
Hong, G. et al. Through-skull fluorescence imaging of the brain in a new near-infrared window. Nat. Photon. 8, 723–730 (2014).
Weissleder, R. A clearer vision for in vivo imaging. Nat. Biotechnol. 19, 316–317 (2001).
Friebel, M., Helfmann, J., Netz, U. & Meinke, M. Influence of oxygen saturation on the optical scattering properties of human red blood cells in the spectral range 250 to 2,000 nm. J. Biomed. Opt. 14, 034001 (2009).
Reinhart, M. B., Huntington, C. R., Blair, L. J., Heniford, B. T. & Augenstein, V. A. Indocyanine green: historical context, current applications and future considerations. Surg. Innov. 23, 166–175 (2016).
Smith, A. M., Mancini, M. C. & Nie, S. Second window for in vivo imaging. Nat. Nanotechnol. 4, 710–711 (2009).
Diao, S. et al. Biological imaging without autofluorescence in the second near-infrared region. Nano Res. 8, 3027–3034 (2015).
Welsher, K. et al. A route to brightly fluorescent carbon nanotubes for near-infrared imaging in mice. Nat. Nanotechnol. 4, 773–780 (2009).
Wang, F. et al. In vivo non-invasive confocal fluorescence imaging beyond 1,700 nm using superconducting nanowire single-photon detectors. Nat. Nanotechnol. 17, 653–660 (2022).
Liu, Z., Tabakman, S., Welsher, K. & Dai, H. Carbon nanotubes in biology and medicine: in vitro and in vivo detection, imaging and drug delivery. Nano Res. 2, 85–120 (2009).
Welsher, K., Sherlock, S. P. & Dai, H. Deep-tissue anatomical imaging of mice using carbon nanotube fluorophores in the second near-infrared window. Proc. Natl Acad. Sci. USA 108, 8943–8948 (2011).
Hong, G. et al. Multifunctional in vivo vascular imaging using near-infrared II fluorescence. Nat. Med. 18, 1841–1846 (2012).
Hong, G. et al. In vivo fluorescence imaging with Ag2S quantum dots in the second near-infrared region. Angew. Chem. Int. Edit. 51, 9818–9821 (2012).
Diao, S. et al. Fluorescence imaging in vivo at wavelengths beyond 1,500 nm. Angew. Chem. Int. Ed. 54, 14758–14762 (2015).
Antaris, A. L. et al. A small-molecule dye for NIR-II imaging. Nat. Mater. 15, 235–242 (2016).
Zhong, Y. et al. Boosting the down-shifting luminescence of rare-earth nanocrystals for biological imaging beyond 1,500 nm. Nat. Commun. 8, 737 (2017).
Wan, H. et al. Developing a bright NIR-II fluorophore with fast renal excretion and its application in molecular imaging of immune checkpoint PD-L1. Adv. Funct. Mater. 28, 1804956 (2018).
Zhang, M. et al. Bright quantum dots emitting at ∼1,600 nm in the NIR-IIb window for deep tissue fluorescence imaging. Proc. Natl Acad. Sci. USA 115, 6590–6595 (2018).
Ma, Z. et al. A theranostic agent for cancer therapy and imaging in the second near-infrared window. Nano Res. 12, 273–279 (2019).
Zhong, Y. et al. In vivo molecular imaging for immunotherapy using ultra-bright near-infrared-IIb rare-earth nanoparticles. Nat. Biotechnol. 37, 1322–1331 (2019).
Wang, F. et al. Light-sheet microscopy in the near-infrared II window. Nat. Methods 16, 545–552 (2019).
Ma, Z. et al. Cross-link-functionalized nanoparticles for rapid excretion in nanotheranostic applications. Angew. Chem. Int. Ed. 59, 20552–20560 (2020).
Wang, F. et al. In vivo NIR-II structured-illumination light-sheet microscopy. Proc. Natl Acad. Sci. USA 118, e2023888118 (2021).
Baghdasaryan, A. et al. Phosphorylcholine-conjugated gold-molecular clusters improve signal for lymph node NIR-II fluorescence imaging in preclinical cancer models. Nat. Commun. 13, 5613 (2022).
Wang, F. et al. High-precision tumor resection down to few-cell level guided by NIR-IIb molecular fluorescence imaging. Proc. Natl Acad. Sci. USA 119, e2123111119 (2022).
Ren, F. et al. Shortwave-infrared-light-emitting probes for the in vivo tracking of cancer vaccines and the elicited immune responses. Nat. Biomed. Eng. (2023); https://doi.org/10.1038/s41551-023-01083-5
Carr, J. A. et al. Absorption by water increases fluorescence image contrast of biological tissue in the shortwave infrared. Proc. Natl Acad. Sci. USA 115, 9080–9085 (2018).
Yoo, K. M., Liu, F. & Alfano, R. R. Imaging through a scattering wall using absorption. Opt. Lett. 16, 1068–1070 (1991).
Shao, W. et al. Tunable narrow band emissions from dye-sensitized core/shell/shell nanocrystals in the second near-infrared biological window. J. Am. Chem. Soc. 138, 16192–16195 (2016).
Wei, Y.-C. et al. Overcoming the energy gap law in near-infrared OLEDs by exciton–vibration decoupling. Nat. Photon. 14, 570–577 (2020).
Zhu, S., Tian, R., Antaris, A. L., Chen, X. & Dai, H. Near-infrared-II molecular dyes for cancer imaging and surgery. Adv. Mater. 31, 1900321 (2019).
Chen, R. J., Zhang, Y., Wang, D. & Dai, H. Noncovalent sidewall functionalization of single-walled carbon nanotubes for protein immobilization. J. Am. Chem. Soc. 123, 3838–3839 (2001).
Zhang, Y. et al. Ag2S quantum dot: a bright and biocompatible fluorescent nanoprobe in the second near-infrared window. ACS Nano 6, 3695–3702 (2012).
Bruns, O. T. et al. Next-generation in vivo optical imaging with short-wave infrared quantum dots. Nat. Biomed. Eng. 1, 0056 (2017).
Pei, P. et al. X-ray-activated persistent luminescence nanomaterials for NIR-II imaging. Nat. Nanotechnol. 16, 1011–1018 (2021).
Zhong, Y. & Dai, H. A mini-review on rare-earth down-conversion nanoparticles for NIR-II imaging of biological systems. Nano Res. 13, 1281–1294 (2020).
Gu, Y. et al. High-sensitivity imaging of time-domain near-infrared light transducer. Nat. Photon. 13, 525–531 (2019).
Yang, Y. et al. Fluorescence-amplified nanocrystals in the second near-infrared window for in vivo real-time dynamic multiplexed imaging. Nat. Nanotechnol. 18, 1195–1204 (2023).
Chen, Y. et al. Shortwave infrared in vivo imaging with gold nanoclusters. Nano Lett. 17, 6330–6334 (2017).
Ma, H. et al. Bioactive NIR-II gold clusters for three-dimensional imaging and acute inflammation inhibition. Sci. Adv. 9, eadh7828 (2023).
Cosco, E. D. et al. Shortwave infrared polymethine fluorophores matched to excitation lasers enable non-invasive, multicolour in vivo imaging in real time. Nat. Chem. 12, 1123–1130 (2020).
Sun, C. et al. J-aggregates of cyanine dye for NIR-II in vivo dynamic vascular imaging beyond 1,500 nm. J. Am. Chem. Soc. 141, 19221–19225 (2019).
Yang, Q. et al. Rational design of molecular fluorophores for biological imaging in the NIR-II window. Adv. Mater. 29, 1605497 (2017).
Zhu, X. et al. High brightness NIR-II nanofluorophores based on fused-ring acceptor molecules. Nano Res. 13, 2570–2575 (2020).
Yao, C. et al. A bright, renal-clearable NIR-II brush macromolecular probe with long blood circulation time for kidney disease bioimaging. Angew. Chem. Int. Ed. 61, e202114273 (2022).
Qin, Z. et al. NIRII-HDs: a versatile platform for developing activatable NIR-II fluorogenic probes for reliable in vivo analyte sensing. Angew. Chem. Int. Ed. 61, e202201541 (2022).
Wang, T. et al. A hybrid erbium(III)–bacteriochlorin near-infrared probe for multiplexed biomedical imaging. Nat. Mater. 20, 1571–1578 (2021).
Lei, Z. et al. Stable, wavelength-tunable fluorescent dyes in the NIR-II region for in vivo high-contrast bioimaging and multiplexed biosensing. Angew. Chem. Int. Ed. 58, 8166–8171 (2019).
Liu, S. et al. Incorporation of planar blocks into twisted skeletons: boosting brightness of fluorophores for bioimaging beyond 1,500 nanometer. ACS Nano 14, 14228–14239 (2020).
Liu, C. et al. 3,4-Ethylenedithio thiophene donor for NIR-II fluorophores with improved quantum yields. Mater. Chem. Front. 7, 2419–2425 (2023).
Ma, H. et al. Propylenedioxy thiophene donor to achieve NIR-II molecular fluorophores with enhanced brightness. Chem. Mater. 32, 2061–2069 (2020).
Yang, Q. et al. Donor engineering for NIR-II molecular fluorophores with enhanced fluorescent performance. J. Am. Chem. Soc. 140, 1715–1724 (2018).
Huang, J. et al. Renal-clearable molecular semiconductor for second near-infrared fluorescence imaging of kidney dysfunction. Angew. Chem. Int. Ed. 58, 15120–15127 (2019).
Shcherbakova, D. M. & Verkhusha, V. V. Near-infrared fluorescent proteins for multicolor in vivo imaging. Nat. Methods 10, 751–754 (2013).
Oliinyk, O. S., Chernov, K. G. & Verkhusha, V. V. Bacterial phytochromes, cyanobacteriochromes and allophycocyanins as a source of near-infrared fluorescent probes. Int. J. Mol. Sci. 18, 1691 (2017).
Chen, M. et al. Long-term monitoring of intravital biological processes using fluorescent protein-assisted NIR-II imaging. Nat. Commun. 13, 6643 (2022).
Oliinyk, O. S. et al. Deep-tissue SWIR imaging using rationally designed small red-shifted near-infrared fluorescent protein. Nat. Methods 20, 70–74 (2023).
Naczynski, D. J. et al. X-ray-induced shortwave infrared biomedical imaging using rare-earth nanoprobes. Nano Lett. 15, 96–102 (2015).
Cao, X. et al. Cherenkov excited short-wavelength infrared fluorescence imaging in vivo with external beam radiation. J. Biomed. Opt. 24, 051405 (2018).
Shen, H. et al. Rational design of NIR-II AIEgens with ultrahigh quantum yields for photo- and chemiluminescence imaging. J. Am. Chem. Soc. 144, 15391–15402 (2022).
Lu, L. et al. NIR-II bioluminescence for in vivo high contrast imaging and in situ ATP-mediated metastases tracing. Nat. Commun. 11, 4192 (2020).
Wang, S. et al. Anti-quenching NIR-II molecular fluorophores for in vivo high-contrast imaging and pH sensing. Nat. Commun. 10, 1058 (2019).
Starosolski, Z. et al. Indocyanine green fluorescence in second near-infrared (NIR-II) window. PLoS ONE 12, e0187563 (2017).
Ren, T.-B. et al. A general strategy for development of activatable NIR-II fluorescent probes for in vivo high-contrast bioimaging. Angew. Chem. Int. Ed. 60, 800–805 (2021).
Wang, Z. et al. Dynamically monitoring lymphatic and vascular systems in physiological and pathological conditions of a swine model via a portable NIR-II imaging system with ICG. Int. J. Med. Sci. 19, 1864–1874 (2022).
Xue, D. et al. Structural and functional NIR-II fluorescence bioimaging in urinary system via clinically approved dye methylene blue. Engineering 22, 149–158 (2023).
Chang, Y. et al. Bright Tm3+-based downshifting luminescence nanoprobe operating around 1,800 nm for NIR-IIb and c bioimaging. Nat. Commun. 14, 1079 (2023).
Arús, B. A. et al. Shortwave infrared fluorescence imaging of peripheral organs in awake and freely moving mice. Front. Neurosci 17, 1135494 (2023).
Kim, T., O’Brien, C., Choi, H. S. & Jeong, M. Y. Fluorescence molecular imaging systems for intraoperative image-guided surgery. Appl. Spectrosc. Rev. 53, 349–359 (2018).
Wan, H. et al. A bright organic NIR-II nanofluorophore for three-dimensional imaging into biological tissues. Nat. Commun. 9, 1171 (2018).
Yu, W. et al. NIR-II fluorescence in vivo confocal microscopy with aggregation-induced emission dots. Sci. Bull. 64, 410–416 (2019).
Yu, J. et al. Intravital confocal fluorescence lifetime imaging microscopy in the second near-infrared window. Opt. Lett. 45, 3305–3308 (2020).
Xia, F. et al. Short-wave infrared confocal fluorescence imaging of deep mouse brain with a superconducting nanowire single-photon detector. ACS Photon. 8, 2800–2810 (2021).
Kobat, D., Horton, N. & Xu, C. In vivo two-photon microscopy to 1.6-mm depth in mouse cortex. J. Biomed. Opt. 16, 106014 (2011).
Horton, N. G. et al. In vivo three-photon microscopy of subcortical structures within an intact mouse brain. Nat. Photon. 7, 205–209 (2013).
Gu, M., Gan, X., Kisteman, A. & Xu, M. G. Comparison of penetration depth between two-photon excitation and single-photon excitation in imaging through turbid tissue media. Appl. Phys. Lett. 77, 1551–1553 (2000).
Huisken, J., Swoger, J., Del Bene, F., Wittbrodt, J. & Stelzer, E. H. K. Optical sectioning deep inside live embryos by selective plane illumination microscopy. Science 305, 1007–1009 (2004).
Liu, T.-L. et al. Observing the cell in its native state: imaging subcellular dynamics in multicellular organisms. Science 360, eaaq1392 (2018).
Yu, H. et al. Combining near-infrared excitation with Swept Confocally-Aligned Planar Excitation (SCAPE) microscopy for fast, volumetric imaging in mouse brain. In Biophotonics Congress: Biomedical Optics Congress 2018 (Microscopy/Translational/Brain/OTS). BF3C.3 (Optica Publishing Group, 2018).
Olarte, O. E. et al. Image formation by linear and nonlinear digital scanned light-sheet fluorescence microscopy with Gaussian and Bessel beam profiles. Biomed. Opt. Express 3, 1492–1505 (2012).
Nylk, J. et al. Light-sheet microscopy with attenuation-compensated propagation-invariant beams. Sci. Adv. 4, eaar4817 (2018).
Herisson, F. et al. Direct vascular channels connect skull bone marrow and the brain surface enabling myeloid cell migration. Nat. Neurosci. 21, 1209–1217 (2018).
Wang, F. et al. Scanning superlens microscopy for non-invasive large field-of-view visible light nanoscale imaging. Nat. Commun. 7, 13748 (2016).
Liu, P. et al. Airy beam assisted NIR-II light-sheet microscopy. Nano Today 47, 101628 (2022).
Gustafsson, M. G. L. Nonlinear structured-illumination microscopy: wide-field fluorescence imaging with theoretically unlimited resolution. Proc. Natl Acad. Sci. USA 102, 13081–13086 (2005).
Hong, G. et al. Near-Infrared II fluorescence for imaging hindlimb vessel regeneration with dynamic tissue perfusion measurement. Circ. Cardiovasc. Imag. 7, 517–525 (2014).
Qi, J. et al. Real-time and high-resolution bioimaging with bright aggregation-induced emission dots in short-wave infrared region. Adv. Mater. 30, 1706856 (2018).
Liu, H. et al. Atomic-precision gold clusters for NIR-II imaging. Adv. Mater. 31, 1901015 (2019).
Zhang, X.-D. et al. Traumatic brain injury imaging in the second near-infrared window with a molecular fluorophore. Adv. Mater. 28, 6872–6879 (2016).
Chatterjee, A., Serniak, N. & Czerniecki, B. J. Sentinel lymph node biopsy in breast cancer: a work in progress. Cancer J. 21, 7–10 (2015).
Tian, R. et al. Multiplexed NIR-II probes for lymph node-invaded cancer detection and imaging-guided surgery. Adv. Mater. 32, 1907365 (2020).
Li, M. et al. Near-infrared-II ratiometric fluorescence probes for non-invasive detection and precise navigation surgery of metastatic sentinel lymph nodes. Theranostics 12, 7191–7202 (2022).
Hu, Z. et al. First-in-human liver-tumour surgery guided by multispectral fluorescence imaging in the visible and near-infrared-I/II windows. Nat. Biomed. Eng. 4, 259–271 (2020).
Morlandt, A. B. et al. Fluorescently labeled Cetuximab-IRDye800 for guided surgical excision of ameloblastoma: a proof of principle study. J. Oral. Maxil. Surg. 78, 1736–1747 (2020).
Suo, Y. et al. NIR-II fluorescence endoscopy for targeted imaging of colorectal cancer. Adv. Healthc. Mater. 8, 1900974 (2019).
Girard, J.-P., Moussion, C. & Förster, R. HEVs, lymphatics and homeostatic immune cell trafficking in lymph nodes. Nat. Rev. Immunol. 12, 762–773 (2012).
He, L. et al. Engineering of reversible NIR-II redox-responsive fluorescent probes for imaging of inflammation in vivo. Angew. Chem. Int. Ed. 61, e202211409 (2022).
Lan, Q. et al. Polymethine molecular platform for ratiometric fluorescent probes in the second near-infrared window. J. Am. Chem. Soc. 144, 21010–21015 (2022).
Miao, J. et al. An activatable NIR-II fluorescent reporter for in vivo imaging of amyloid-β plaques. Angew. Chem. Int. Ed. 62, e202216351 (2023).
He, Y. et al. NIR-II cell endocytosis-activated fluorescent probes for in vivo high-contrast bioimaging diagnostics. Chem. Sci. 12, 10474–10482 (2021).
Chen, Z. et al. Design and synthesis of a small molecular NIR-II chemiluminescence probe for in vivo-activated H2S imaging. Proc. Natl Acad. Sci. USA 120, e2205186120 (2023).
Fang, Z. et al. Oxyhaemoglobin saturation NIR-IIb imaging for assessing cancer metabolism and predicting the response to immunotherapy. Nat. Nanotechnol 19, 124–130 (2024).
Tian, R. et al. Rational design of a super-contrast NIR-II fluorophore affords high-performance NIR-II molecular imaging guided microsurgery. Chem. Sci. 10, 326–332 (2019).
Wang, P. et al. NIR-II nanoprobes in-vivo assembly to improve image-guided surgery for metastatic ovarian cancer. Nat. Commun. 9, 2898 (2018).
Fan, X. et al. Aggregation-induced emission (AIE) nanoparticles-assisted NIR-II fluorescence imaging-guided diagnosis and surgery for inflammatory bowel disease (IBD). Adv. Healthc. Mater. 10, 2101043 (2021).
Antaris, A. L. et al. A high quantum yield molecule-protein complex fluorophore for near-infrared II imaging. Nat. Commun. 8, 15269 (2017).
Wu, Y. et al. First clinical applications for the NIR-II imaging with ICG in microsurgery. Front. Bioeng. Biotechnol. 10, 1042546 (2022).
Carr, J. A., Valdez, T. A., Bruns, O. T. & Bawendi, M. G. Using the shortwave infrared to image middle ear pathologies. Proc. Natl Acad. Sci. USA 113, 9989–9994 (2016).
Shi, X. et al. Zn-doping enhances the photoluminescence and stability of PbS quantum dots for in vivo high-resolution imaging in the NIR-II window. Nano Res. 13, 2239–2245 (2020).
Magdaong, N. C. M., Niedzwiedzki, D. M., Goodson, C. & Blankenship, R. E. Carotenoid-to-bacteriochlorophyll energy transfer in the LH1–RC core complex of a bacteriochlorophyll b containing purple photosynthetic bacterium blastochloris viridis. J. Phys. Chem. B 120, 5159–5171 (2016).
Chang, Z. et al. Near infrared-II fluorescent protein for in-vivo imaging. Preprint at https://www.biorxiv.org/content/10.1101/2022.03.04.482971v1.full (2022).
Ma, Z., Wang, F., Wang, W., Zhong, Y. & Dai, H. Deep learning for in vivo near-infrared imaging. Proc. Natl Acad. Sci. USA 118, e2021446118 (2021).
Tummers, W. S. et al. Regulatory aspects of optical methods and exogenous targets for cancer detection. Cancer Res. 77, 2197–2206 (2017).
Scheuer, W., van Dam, G. M., Dobosz, M., Schwaiger, M. & Ntziachristos, V. Drug-based optical agents: infiltrating clinics at lower risk. Sci. Transl. Med. 4, 134ps111 2012).
DSouza, A., Lin, H., Henderson, E., Samkoe, K. & Pogue, B. Review of fluorescence guided surgery systems: identification of key performance capabilities beyond indocyanine green imaging. J. Biomed. Opt. 21, 080901 (2016).
Wang, M. et al. Comparing the effective attenuation lengths for long wavelength in vivo imaging of the mouse brain. Biomed. Opt. Express 9, 3534–3543 (2018).
Acknowledgements
H. Dai acknowledges the National Institutes of Health (NIH DP1-NS-105737) as the sole funding source for the Dai group’s work reviewed in this paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Photonics thanks Jun Qian and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, F., Zhong, Y., Bruns, O. et al. In vivo NIR-II fluorescence imaging for biology and medicine. Nat. Photon. (2024). https://doi.org/10.1038/s41566-024-01391-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41566-024-01391-5