Abstract
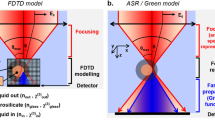
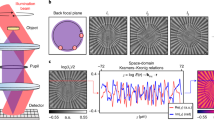
Second-harmonic generation microscopy is a valuable label-free modality for imaging non-centrosymmetric structures and has important biomedical applications from live-cell imaging to cancer diagnosis. Conventional second-harmonic generation microscopy measures intensity signals that originate from tightly focused laser beams, preventing researchers from solving the scattering inverse problem for second-order nonlinear materials. Here, we present harmonic optical tomography (HOT) as a novel modality for imaging microscopic, nonlinear and inhomogeneous objects. The HOT principle of operation relies on interferometrically measuring the complex harmonic field and using a scattering inverse model to reconstruct the three-dimensional distribution of harmonophores. HOT enables strong axial sectioning via the momentum conservation of spatially and temporally broadband fields. We illustrate the HOT operation with experiments and reconstructions on a beta-barium borate crystal and various biological specimens. Although our results involve second-order nonlinear materials, we show that this approach applies to any coherent nonlinear process.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
Code availability
The MATLAB code used for HOT reconstruction is available from the corresponding authors upon reasonable request.
References
Campagnola, P. J., Wei, M. D., Lewis, A. & Loew, L. M. High-resolution nonlinear optical imaging of live cells by second harmonic generation. Biophys. J. 77, 3341–3349 (1999).
Campagnola, P. J. & Loew, L. M. Second-harmonic imaging microscopy for visualizing biomolecular arrays in cells, tissues and organisms. Nat. Biotechnol. 21, 1356–1360 (2003).
Masters, B. R. & So, P. T. C. Handbook of Biomedical Nonlinear Optical Microscopy (Oxford University Press, 2008).
Ambekar, R., Lau, T. Y., Walsh, M., Bhargava, R. & Toussaint, K. C. Quantifying collagen structure in breast biopsies using second-harmonic generation imaging. Biomed. Opt. Express 3, 2021–2035 (2012).
Boyd, R. W. Nonlinear Optics 3rd edn (Academic Press, 2008).
Zipfel, W. R. et al. Live tissue intrinsic emission microscopy using multiphoton-excited native fluorescence and second harmonic generation. Proc. Natl Acad. Sci. USA 100, 7075–7080 (2003).
Zoumi, A., Yeh, A. & Tromberg, B. J. Imaging cells and extracellular matrix in vivo by using second-harmonic generation and two-photon excited fluorescence. Proc. Natl Acad. Sci. USA 99, 11014–11019 (2002).
Zoumi, A., Yeh, A. & Tromberg, B. J. Imaging cells and extracellular matrix—in vivo—by using second-harmonic generation and two-photon excited fluorescence. Proc. Natl Acad. Sci. USA 99, 11014–11019 (2002).
Vuillemin, N. et al. Efficient second-harmonic imaging of collagen in histological slides using Bessel beam excitation. Sci Rep. 6, 29863 (2016).
Ducourthial, G. et al. Monitoring dynamic collagen reorganization during skin stretching with fast polarization-resolved second harmonic generation imaging. J. Biophoton. 12, e201800336 (2019).
Campagnola, P. J. et al. Three-dimensional high-resolution second-harmonic generation imaging of endogenous structural proteins in biological tissues. Biophys. J. 82, 493–508 (2002).
Mohler, W., Millard, A. C. & Campagnola, P. J. Second harmonic generation imaging of endogenous structural proteins. Methods 29, 97–109 (2003).
Chu, S. W. et al. Studies of χ (2)/χ (3) tensors in submicron-scaled bio-tissues by polarization harmonics optical microscopy. Biophys. J. 86, 3914–3922 (2004).
Dombeck, D. A. et al. Uniform polarity microtubule assemblies imaged in native brain tissue by second-harmonic generation microscopy. Proc. Natl Acad. Sci. USA 100, 7081–7086 (2003).
Brown, E. et al. Dynamic imaging of collagen and its modulation in tumors in vivo using second-harmonic generation. Nat. Med. 9, 796–800 (2003).
Ajeti, V. et al. Structural changes in mixed Col I/Col V collagen gels probed by SHG microscopy: implications for probing stromal alterations in human breast cancer. Biomed. Opt. Express 2, 2307–2316 (2011).
Provenzano, P. P. et al. Collagen reorganization at the tumor-stromal interface facilitates local invasion. BMC Med. 4, 38 (2006).
Provenzano, P. P. et al. Collagen density promotes mammary tumor initiation and progression. BMC Med. 6, 11 (2008).
Hunter, M. et al. Tissue self-affinity and polarized light scattering in the Born approximation: a new model for precancer detection. Phys. Rev. Lett. 97, 138102 (2006).
Birk, J. W. et al. Second harmonic generation imaging distinguishes both high-grade dysplasia and cancer from normal colonic mucosa. Digest Dis. Sci. 59, 1529–1534 (2014).
Campagnola, P. Second harmonic generation imaging microscopy: applications to diseases diagnostics. Anal. Chem. 83, 3224–3231 (2011).
Tilbury, K. & Campagnola, P. J. Applications of second-harmonic generation imaging microscopy in ovarian and breast cancer. Perspect. Med. Chem. 7, 21–32 (2015).
Zipfel, W. R., Williams, R. M. & Webb, W. W. Nonlinear magic: multiphoton microscopy in the biosciences. Nat. Biotechnol. 21, 1368–1376 (2003).
Mertz, J. & Moreaux, L. Second-harmonic generation by focused excitation of inhomogeneously distributed scatterers. Opt. Commun. 196, 325–330 (2001).
Ozcan, A., Digonnet, M. & Kino, G. Iterative processing of second-order optical nonlinearity depth profiles. Opt. Express 12, 3367–3376 (2004).
Ozcan, A., Digonnet, M. J. F. & Kino, G. S. Detailed analysis of inverse fourier transform techniques to uniquely infer second-order nonlinearity profile of thin films. J. Appl. Phys. 97, 013502 (2005).
Balu, M., Mikami, H., Hou, J., Potma, E. O. & Tromberg, B. J. Rapid mesoscale multiphoton microscopy of human skin. Biomed. Opt. Express 7, 4375–4387 (2016).
Campbell, K. R. et al. 3D second harmonic generation imaging tomography by multi-view excitation. Optica 4, 1171–1179 (2017).
Pu, Y., Centurion, M. & Psaltis, D. Harmonic holography: a new holographic principle. Appl. Optics 47, A103–A110 (2008).
Smith, D. R., Winters, D. G. & Bartels, R. A. Submillisecond second harmonic holographic imaging of biological specimens in three dimensions. Proc. Natl Acad. Sci. USA 110, 18391–18396 (2013).
Goodman, A. J. & Tisdale, W. A. Enhancement of second-order nonlinear-optical signals by optical stimulation. Phys. Rev. Lett. 114, 183902 (2015).
Shaffer, E., Marquet, P. & Depeursinge, C. Real time, nanometric 3D-tracking of nanoparticles made possible by second harmonic generation digital holographic microscopy. Opt. Express 18, 17392–17403 (2010).
Couture, C. A. et al. The impact of collagen fibril polarity on second harmonic generation microscopy. Biophys. J. 109, 2501–2510 (2015).
Winters, D. G., Smith, D. R., Schlup, P. & Bartels, R. A. Measurement of orientation and susceptibility ratios using a polarization-resolved second-harmonic generation holographic microscope. Biomed. Opt. Express 3, 2004–2011 (2012).
Reider, G. A., Cernusca, M. & Hofer, M. Coherence artifacts in second harmonic microscopy. Appl. Phys. B 68, 343–347 (1999).
Pinsard, M. et al. Elimination of imaging artifacts in second harmonic generation microscopy using interferometry. Biomed. Opt. Express 10, 3938–3952 (2019).
Popescu, G. Quantitative Phase Imaging of Cells and Tissues (McGraw-Hill, 2011).
Hu, C. & Popescu, G. in Label-Free Super-Resolution Microscopy 1–24 (Springer, 2019).
Popescu, G. Principles of Biophotonics: Linear Systems and the Fourier Transform in Optics Vol. 1 (IOP, 2018).
Sung, Y. et al. Optical diffraction tomography for high resolution live cell imaging. Opt. Express 17, 266–277 (2009).
Choi, W. et al. Tomographic phase microscopy. Nat. Methods 4, 717–719 (2007).
Jin, D., Zhou, R. J., Yaqoob, Z. & So, P. T. C. Tomographic phase microscopy: principles and applications in bioimaging [invited]. J. Opt. Soc. Am. B 34, B64–B77 (2017).
Gokhin, D. S., Dubuc, E. A., Lian, K. Q., Peters, L. L. & Fowler, V. M. Alterations in thin filament length during postnatal skeletal muscle development and aging in mice. Front. Physiol. 5, 375 (2014).
Moo, E. K., Fortuna, R., Sibole, S. C., Abusara, Z. & Herzog, W. In vivo sarcomere lengths and sarcomere elongations are not uniform across an intact muscle. Front. Physiol. 7, 187 (2016).
Park, Y., Depeursinge, C. & Popescu, G. Quantitative phase imaging in biomedicine. Nat. Photon. 12, 578–589 (2018).
Kim, K. et al. Diffraction optical tomography using a quantitative phase imaging unit. Opt. Lett. 39, 6935–6938 (2014).
Kim, T. et al. White-light diffraction tomography of unlabeled live cells. Nat. Photon. 8, 256–263 (2014).
Kandel, M. E. et al. Epi-illumination gradient light interference microscopy for imaging opaque structures. Nat. Commun. 10, 4691 (2019).
Nguyen, T. H., Kandel, M. E., Rubessa, M., Wheeler, M. B. & Popescu, G. Gradient light interference microscopy for 3D imaging of unlabeled specimens. Nat. Commun. 8, 210 (2017).
Conklin, M. W. et al. Aligned collagen is a prognostic signature for survival in human breast carcinoma. Am. J. Pathol 178, 1221–1232 (2011).
Hu, C. et al. Imaging collagen properties in the uterosacral ligaments of women with pelvic organ prolapse using spatial light interference microscopy (SLIM). Front. Phys. 7, 72 (2019).
Majeed, H., Okoro, C., Kajdacsy-Balla, A., Toussaint, K. C. & Popescu, G. Quantifying collagen fiber orientation in breast cancer using quantitative phase imaging. BIOMEDO 22, 046004 (2017).
Kieu, K., Renninger, W. H., Chong, A. & Wise, F. W. Sub-100 fs pulses at watt-level powers from a dissipative-soliton fiber laser. Opt. Lett. 34, 593–595 (2009).
Domingue, S. R. & Bartels, R. A. Nonlinear fiber amplifier with tunable transform limited pulse duration from a few 100 to sub-100-fs at watt-level powers. Opt. Lett. 39, 359–362 (2014).
Sidorenko, P., Fu, W. & Wise, F. Nonlinear ultrafast fiber amplifiers beyond the gain-narrowing limit. Optica 6, 1328–1333 (2019).
Acknowledgements
This work is supported in part by the National Science Foundation (0939511, 1450962, 1353368) and the National Institutes of Health (R01CA238191, R01GM129709, R21EB025389 and R21MH117786).
Author information
Authors and Affiliations
Contributions
C.H., V.K. and G.P. developed the theoretical model, with input from R.A.B. and K.C.T. K.W. built the fibre laser system, J.J.F. built the microscope system and collected experimental data. C.H. developed the numerical reconstruction. C.H. and B.C. rendered figures and Supplememtary videos. R.A.B. supervised the experimental work. G.P. supervised the theoretical work. C.H. and G.P. wrote the manuscript with input from all authors.
Corresponding authors
Ethics declarations
Competing interests
G.P. has financial interest in Phi Optics, a company developing quantitative phase imaging technology for materials and life science applications, which, however, did not sponsor the research. The authors disclosed this invention to the UIUC Office of Technology management.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Fig. 1 and discussion.
Supplementary Video 1
A 3D rendering of an inhomogeneous nonlinear crystal.
Supplementary Video 2
A 3D rendering of murine skeletal muscle.
Rights and permissions
About this article
Cite this article
Hu, C., Field, J.J., Kelkar, V. et al. Harmonic optical tomography of nonlinear structures. Nat. Photonics 14, 564–569 (2020). https://doi.org/10.1038/s41566-020-0638-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41566-020-0638-5
This article is cited by
-
Wide-field coherent anti-Stokes Raman scattering microscopy using random illuminations
Nature Photonics (2023)
-
Bond-selective intensity diffraction tomography
Nature Communications (2022)
-
Live-dead assay on unlabeled cells using phase imaging with computational specificity
Nature Communications (2022)
-
Dehydration of plant cells shoves nuclei rotation allowing for 3D phase-contrast tomography
Light: Science & Applications (2021)
-
High-speed 3D mapping of nonlinear structures
Nature Photonics (2020)