Abstract
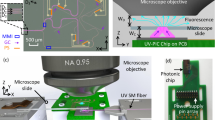
Structured illumination microscopy (SIM) enables live-cell super-resolution imaging of subcellular structures at high speeds. At present, linear SIM uses free-space optics to illuminate the sample with the desired light patterns; however, such arrangements are prone to misalignment and add cost and complexity to the microscope. Here, we present an alternative photonic chip-based two-dimensional SIM approach (cSIM) in which the conventional glass sample slide in a microscope is replaced by a planar photonic chip that importantly both holds and illuminates the specimen. The photonic chip reduces the footprint of the light illumination path of SIM to around 4 × 4 cm2. An array of optical waveguides on the chip creates standing wave interference patterns at different angles, which illuminate the sample via evanescent fields. High-refractive-index silicon nitride waveguides allow a 2.3 times enhancement in imaging spatial resolution, exceeding the usual 2 times limit of SIM. In summary, cSIM offers a simple, stable and affordable approach for performing two-dimensional super-resolution imaging over a large field of view.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data associated with the figures and Supplementary information can be obtained at https://github.com/UiTcSIM/cSIM.git.
Code availability
The SIM reconstructions in Figs. 3 and 4 and in Supplementary Fig. 11 were performed using FAIR SIM, an open source code available at https://github.com/fairSIM. The SIM reconstructions in Fig. 5 were performed using code provided by R. Heintzmann at the Friedrich-Schiller-University Jena, Germany.
References
Goodman, J. W. Introduction to Fourier Optics (W. H. Freeman, 2005).
Pawley, J. Handbook of Biological Confocal Microscopy (Springer, 2010).
Eggeling, C., Willig, K. I., Sahl, S. J. & Hell, S. W. Lens-based fluorescence nanoscopy. Q. Rev. Biophys. 48, 178–243 (2015).
Hell, S. W. & Wichmann, J. Breaking the diffraction resolution limit by stimulated emission: stimulated-emission-depletion fluorescence microscopy. Opt. Lett. 19, 780–782 (1994).
Willig, K. I., Rizzoli, S. O., Westphal, V., Jahn, R. & Hell, S. W. STED microscopy reveals that synaptotagmin remains clustered after synaptic vesicle exocytosis. Nature 440, 935–939 (2006).
Rust, M. J., Bates, M. & Zhuang, X. Sub-diffraction-limit imaging by stochastic optical reconstruction microscopy (STORM). Nat. Methods 3, 793 (2006).
Heilemann, M. et al. Subdiffraction-resolution fluorescence imaging with conventional fluorescent probes. Angew. Chem. Int. Ed. 47, 6172–6176 (2008).
Betzig, E. et al. Imaging intracellular fluorescent proteins at nanometer resolution. Science 313, 1642–1645 (2006).
Gustafsson, M. G. Surpassing the lateral resolution limit by a factor of two using structured illumination microscopy. J. Microsc. 198, 82–87 (2000).
Heintzmann, R. & Cremer, C. G. Laterally Modulated Excitation Microscopy: Improvement of Resolution by using a Diffraction Grating Vol. 3568 EBO (SPIE, 1999).
Sahl, S. J., Hell, S. W. & Jakobs, S. Fluorescence nanoscopy in cell biology. Nat. Rev. Mol. Cell Biol. 18, 685–701 (2017).
Yeh, L.-H., Chowdhury, S., Repina, N. A. & Waller, L. Speckle-structured illumination for 3D phase and fluorescence computational microscopy. Biomed. Opt. Express 10, 3635–3653 (2019).
Mudry, E. et al. Structured illumination microscopy using unknown speckle patterns. Nat. Photon. 6, 312–315 (2012).
Ayuk, R. et al. Structured illumination fluorescence microscopy with distorted excitations using a filtered blind-SIM algorithm. Opt. Lett. 38, 4723–4726 (2013).
Jost, A. et al. Optical sectioning and high resolution in single-slice structured illumination microscopy by thick slice blind-SIM reconstruction. PLoS ONE 10, e0132174 (2015).
Heintzmann, R., Jovin, T. M. & Cremer, C. Saturated patterned excitation microscopy—a concept for optical resolution improvement. J. Opt. Soc. Am. A 19, 1599–1609 (2002).
Gustafsson, M. G. L. Nonlinear structured-illumination microscopy: wide-field fluorescence imaging with theoretically unlimited resolution. Proc. Natl Acad. Sci. USA 102, 13081–13086 (2005).
Rego, E. H. et al. Nonlinear structured-illumination microscopy with a photoswitchable protein reveals cellular structures at 50-nm resolution. Proc. Natl Acad. Sci. USA 109, E135–E143 (2012).
Li, D. et al. Extended-resolution structured illumination imaging of endocytic and cytoskeletal dynamics. Science 349, aab3500 (2015).
Cragg, G. E. & So, P. T. C. Lateral resolution enhancement with standing evanescent waves. Opt. Lett. 25, 46–48 (2000).
Chung, E., Kim, D. & So, P. T. Extended resolution wide-field optical imaging: objective-launched standing-wave total internal reflection fluorescence microscopy. Opt. Lett. 31, 945–947 (2006).
Kner, P., Chhun, B. B., Griffis, E. R., Winoto, L. & Gustafsson, M. G. L. Super-resolution video microscopy of live cells by structured illumination. Nat. Methods 6, 339–342 (2009).
Grandin, H. M., Stadler, B., Textor, M. & Voros, J. Waveguide excitation fluorescence microscopy: a new tool for sensing and imaging the biointerface. Biosens. Bioelectron. 21, 1476–1482 (2006).
Agnarsson, B., Ingthorsson, S., Gudjonsson, T. & Leosson, K. Evanescent-wave fluorescence microscopy using symmetric planar waveguides. Opt. Express 17, 5075–5082 (2009).
Agnarsson, B., Jonsdottir, A. B., Arnfinnsdottir, N. B. & Leosson, K. On-chip modulation of evanescent illumination and live-cell imaging with polymer waveguides. Opt. Express 19, 22929–22935 (2011).
Diekmann, R. et al. Chip-based wide field-of-view nanoscopy. Nat. Photon. 11, 322–328 (2017).
Helle, Ø. I., Coucheron, D. A., Tinguely, J.-C., Øie, C. I. & Ahluwalia, B. S. Nanoscopy on-a-chip: super-resolution imaging on the millimeter scale. Opt. Express 27, 6700–6710 (2019).
Tinguely, J.-C., Helle, Ø. I. & Ahluwalia, B. S. Silicon nitride waveguide platform for fluorescence microscopy of living cells. Opt. Express 25, 27678–27690 (2017).
Fu, Y., Ye, T., Tang, W. & Chu, T. Efficient adiabatic silicon-on-insulator waveguide taper. Photon. Res. 2, A41–A44 (2014).
Harris, N. C. et al. Efficient, compact and low loss thermo-optic phase shifter in silicon. Opt. Express 22, 10487–10493 (2014).
van de Linde, S. et al. Direct stochastic optical reconstruction microscopy with standard fluorescent probes. Nat. Protoc. 6, 991–1009 (2011).
Wei, F. & Liu, Z. Plasmonic structured illumination microscopy. Nano Lett. 10, 2531–2536 (2010).
Wei, F. et al. Wide field super-resolution surface imaging through plasmonic structured illumination microscopy. Nano Lett. 14, 4634–4639 (2014).
Archetti, A. et al. Waveguide-PAINT offers an open platform for large field-of-view super-resolution imaging. Nat. Commun. 10, 1267 (2019).
Liu, X. et al. Fluorescent nanowire ring illumination for wide-field far-field subdiffraction imaging. Phys. Rev. Lett. 118, 076101 (2017).
Schneider, J. et al. Ultrafast, temporally stochastic STED nanoscopy of millisecond dynamics. Nat. Methods 12, 827–830 (2015).
Santi, P. A. Light sheet fluorescence microscopy: a review. J. Histochem. Cytochem. 59, 129–138 (2011).
Cnossen, J. et al. Localization microscopy at doubled precision with patterned illumination. Nat. Methods 17, 59–63 (2020).
Otterstrom, N. T., Behunin, R. O., Kittlaus, E. A., Wang, Z. & Rakich, P. T. A silicon Brillouin laser. Science 360, 1113–1116 (2018).
Gaeta, A. L., Lipson, M. & Kippenberg, T. J. Photonic-chip-based frequency combs. Nat. Photon. 13, 158–169 (2019).
Orth, A. & Crozier, K. Gigapixel fluorescence microscopy with a water immersion microlens array. Opt. Express 21, 2361–2368 (2013).
Løvhaugen, P., Ahluwalia, B. S., Huser, T. R. & Hellesø, O. G. Serial Raman spectroscopy of particles trapped on a waveguide. Opt. Express 21, 2964–2970 (2013).
Helle, Ø. I., Ahluwalia, B. S. & Hellesø, O. G. Optical transport, lifting and trapping of micro-particles by planar waveguides. Opt. Express 23, 6601–6612 (2015).
Ahluwalia, B. S., Løvhaugen, P. & Hellesø, O. G. Waveguide trapping of hollow glass spheres. Opt. Lett. 36, 3347–3349 (2011).
Wang, X. et al. Enhanced cell sorting and manipulation with combined optical tweezer and microfluidic chip technologies. Lab Chip 11, 3656–3662 (2011).
Yurtsever, G., Weiss, N., Kalkman, J., van Leeuwen, T. G. & Baets, R. Ultra-compact silicon photonic integrated interferometer for swept-source optical coherence tomography. Opt. Lett. 39, 5228–5231 (2014).
Prieto, F. et al. An integrated optical interferometric nanodevice based on silicon technology for biosensor applications. Nanotechnology 14, 907 (2003).
Müller, M., Mönkemöller, V., Hennig, S., Hübner, W. & Huser, T. Open-source image reconstruction of super-resolution structured illumination microscopy data in ImageJ. Nat. Commun. 7, 10980 (2016).
Ovesný, M., Křížek, P., Borkovec, J., Švindrych, Z. & Hagen, G. M. ThunderSTORM: a comprehensive ImageJ plug-in for PALM and STORM data analysis and super-resolution imaging. Bioinformatics 30, 2389–2390 (2014).
Mönkemöller, V. et al. Primary rat LSECs preserve their characteristic phenotype after cryopreservation. Sci. Rep. 8, 14657–14657 (2018).
Acknowledgements
We thank R. Heintzmann and S. Das for help with the SIM reconstruction algorithms and design of the fibre-optical phase modulator, respectively. We also acknowledge C. I. Øie and D. Wolfson for help with biological specimens and T. Huser for fruitful conceptual discussions. This work was supported by the European Research Council (grant no. 336716 to B.S.A.).
Author information
Authors and Affiliations
Contributions
B.S.A. supervised and conceived this project and provided funding for the project. Ø.I.H. built the set-up, performed the experiments and analysed the data. J.-C.T., F.T.D. and O.G.H. designed the waveguide chip and the mask for fabrication. All authors contributed to chip design. Ø.I.H., F.T.D. and J.-C.T. characterized the waveguides. F.T.D. and Ø.I.H. fabricated the on-chip thermo-optics for phase modulation. M.L. performed all the simulations of cSIM. Ø.I.H. and B.S.A. mainly wrote the manuscript and all authors commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
B.SA. and O.G.H. have applied for patent GB1705660.7 on SIM-on-chip. B.S.A. and Ø.I.H. are founders of Chip NanoImaging (https://www.chipnano.com/) that is commercializing SIM-on-chip technology. The other authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–17 and Notes 1–6.
Rights and permissions
About this article
Cite this article
Helle, Ø.I., Dullo, F.T., Lahrberg, M. et al. Structured illumination microscopy using a photonic chip. Nat. Photonics 14, 431–438 (2020). https://doi.org/10.1038/s41566-020-0620-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41566-020-0620-2
This article is cited by
-
Superresolution structured illumination microscopy reconstruction algorithms: a review
Light: Science & Applications (2023)
-
Transmission structured illumination microscopy with tunable frequency illumination using tilt mirror assembly
Scientific Reports (2023)
-
A sub-wavelength Si LED integrated in a CMOS platform
Nature Communications (2023)
-
Far-field super-resolution chemical microscopy
Light: Science & Applications (2023)
-
Single planar photonic chip with tailored angular transmission for multiple-order analog spatial differentiator
Nature Communications (2022)