Abstract
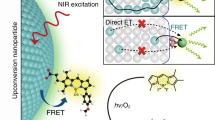
Here, we explore the enhancement of single-molecule emission by a polymeric nanoantenna that can harvest energy from thousands of donor dyes to a single acceptor. In this nanoantenna, the cationic dyes are brought together, in very close proximity, using bulky counterions, thus enabling ultrafast diffusion of excitation energy (≤30 fs) with minimal losses. Our 60 nm nanoparticles containing >10,000 rhodamine-based donor dyes can efficiently transfer energy to 1–2 acceptors, resulting in an antenna effect of ~1,000. Therefore, single Cy5-based acceptors become 25-fold brighter than quantum dots QD655. This unprecedented amplification of the acceptor dye emission enables observation of single molecules at illumination powers (1–10 mW cm−2) that are >10,000-fold lower than typically required in single-molecule measurements. Finally, using a basic set-up, which includes a ×20 air objective and a scalable complementary metal-oxide–semiconductor camera, we could detect single Cy5 molecules by simply shining divergent light on the sample at powers equivalent to sunlight.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
12 January 2018
Owing to a technical error, the wrong version of the Supplementary Information was published for this Article; the equation E = xcoupled donors × ElocalFRET related to Supplementary Table 6 appeared incorrectly. This error has now been corrected.
References
Holzmeister, P., Acuna, G. P., Grohmann, D. & Tinnefeld, P. Breaking the concentration limit of optical single-molecule detection. Chem. Soc. Rev. 43, 1014–1028 (2014).
Sauer, M., Hofkens, J. & Enderlein, J. Handbook of Fluorescence Spectroscopy and Imaging: From Ensemble to Single Molecules (Wiley-VCH, Weinheim, 2011).
Magidson, V. & Khodjakov, A. in Methods in Cell Biology Vol. 114 (eds S. Greenfield & E. W. David) 545–560 (Academic, 2013).
Dixit, R. & Cyr, R. Cell damage and reactive oxygen species production induced by fluorescence microscopy: effect on mitosis and guidelines for non-invasive fluorescence microscopy. Plant J. 36, 280–290 (2003).
Gerhardt, I., Mai, L. J., Lamas-Linares, A. & Kurtsiefer, C. Detection of single molecules illuminated by a light-emitting diode. Sensors 11, 905–916 (2011).
Wei, Q. S. et al. Fluorescent imaging of single nanoparticles and viruses on a smart phone. ACS Nano 7, 9147–9155 (2013).
Prakash, H. P. G. J. Solar Energy: Fundamentals and Applications (Tata McGraw-Hill, New Delhi, 2000).
Novotny, L. & van Hulst, N. Antennas for light. Nat. Photon. 5, 83–90 (2011).
Su, L. et al. Super-resolution localization and defocused fluorescence microscopy on resonantly coupled single-molecule, single-nanorod hybrids. ACS Nano 10, 2455–2466 (2016).
Kinkhabwala, A. et al. Large single-molecule fluorescence enhancements produced by a bowtie nanoantenna. Nat. Photon. 3, 654–657 (2009).
Garcia-Parajo, M. F. Optical antennas focus in on biology. Nat. Photon. 2, 201–203 (2008).
Tinnefeld, P. Single-molecule detection: breaking the concentration barrier. Nat. Nanotech. 8, 480–482 (2013).
Acuna, G. P. et al. Fluorescence enhancement at docking sites of DNA-directed self-assembled nanoantennas. Science 338, 506–510 (2012).
Puchkova, A. et al. DNA origami nanoantennas with over 5000-fold fluorescence enhancement and single-molecule detection at 25 μM. Nano Lett. 15, 8354–8359 (2015).
Yuan, H. F., Khatua, S., Zijlstra, P., Yorulmaz, M. & Orrit, M. Thousand-fold enhancement of single-molecule fluorescence near a single gold nanorod. Angew. Chem. Int. Ed. 52, 1217–1221 (2013).
Scholes, G. D., Fleming, G. R., Olaya-Castro, A. & Van Grondelle, R. Lessons from nature about solar light harvesting. Nat. Chem. 3, 763–774 (2011).
Gartzia-Rivero, L., Banuelos, J. & Lopez-Arbeloa, I. Excitation energy transfer in artificial antennas: from photoactive materials to molecular assemblies. Int. Rev. Phys. Chem. 34, 515–556 (2015).
Tian, Z. Y., Yu, J. B., Wu, C. F., Szymanski, C. & McNeill, J. Amplified energy transfer in conjugated polymer nanoparticle tags and sensors. Nanoscale 2, 1999–2011 (2010).
Yeo, H., Tanaka, K. & Chujo, Y. Effective light-harvesting antennae based on BODIPY-tethered cardo polyfluorenes via rapid energy transferring and low concentration quenching. Macromolecules 46, 2599–2605 (2013).
Thomas Iii, S. W., Joly, G. D. & Swager, T. M. Chemical sensors based on amplifying fluorescent conjugated polymers. Chem. Rev. 107, 1339–1386 (2007).
Galindo, J. F. et al. Dynamics of energy transfer in a conjugated dendrimer driven by ultrafast localization of excitations. J. Am. Chem. Soc. 137, 11637–11644 (2015).
Adronov, A. & Frechet, J. M. J. Light-harvesting dendrimers. Chem. Commun. 1701–1710 (2000).
Yang, J., Yoon, M. C., Yoo, H., Kim, P. & Kim, D. Excitation energy transfer in multiporphyrin arrays with cyclic architectures: towards artificial light-harvesting antenna complexes. Chem. Soc. Rev. 41, 4808–4826 (2012).
Chadha, G., Yang, Q. Z. & Zhao, Y. Self-assembled light-harvesting supercomplexes from fluorescent surface-cross-linked micelles. Chem. Commun. 51, 12939–12942 (2015).
Sun, H. C. et al. Micelle-induced self-assembling protein nanowires: versatile supramolecular scaffolds for designing the light-harvesting system. ACS Nano 10, 421–428 (2016).
Peng, H.-Q. et al. Artificial light-harvesting system based on multifunctional surface-cross-linked micelles. Angew. Chem. Int. Ed. 51, 2088–2092 (2012).
Bhattacharyya, S., Jana, B. & Patra, A. Multichromophoric organic molecules encapsulated in polymer nanoparticles for artificial light harvesting. ChemPhysChem 16, 796–804 (2015).
Winiger, C. B., Li, S. G., Kumar, G. R., Langenegger, S. M. & Haner, R. Long-distance electronic energy transfer in light-harvesting supramolecular polymers. Angew. Chem. Int. Ed. 53, 13609–13613 (2014).
Lin, H. Z. et al. Collective fluorescence blinking in linear J-aggregates assisted by long-distance exciton migration. Nano Lett. 10, 620–626 (2010).
Son, H. J. et al. Light-harvesting and ultrafast energy migration in porphyrin-based metal–organic frameworks. J. Am. Chem. Soc. 135, 862–869 (2013).
Woller, J. G., Hannestad, J. K. & Albinsson, B. Self-assembled nanoscale DNA–porphyrin complex for artificial light harvesting. J. Am. Chem. Soc. 135, 2759–2768 (2013).
Peng, H.-Q. et al. Biological applications of supramolecular assemblies designed for excitation energy transfer. Chem. Rev. 115, 7502–7542 (2015).
Reisch, A. & Klymchenko, A. S. Fluorescent polymer nanoparticles based on dyes: seeking brighter tools for bioimaging. Small 12, 1968–1992 (2016).
Reisch, A. et al. Collective fluorescence switching of counterion-assembled dyes in polymer nanoparticles. Nat. Commun. 5, 4089 (2014).
Genovese, D. et al. Energy transfer processes in dye-doped nanostructures yield cooperative and versatile fluorescent probes. Nanoscale 6, 3022–3036 (2014).
Trofymchuk, K. et al. Exploiting fast exciton diffusion in dye-doped polymer nanoparticles to engineer efficient photoswitching. J. Phys. Chem. Lett. 6, 2259–2264 (2015).
Colby, K. A. et al. Electronic energy migration on different time scales: concentration dependence of the time-resolved anisotropy and fluorescence quenching of lumogen red in poly(methyl methacrylate). J. Phys. Chem. A 114, 3471–3482 (2010).
Shulov, I. et al. Fluorinated counterion-enhanced emission of rhodamine aggregates: ultrabright nanoparticles for bioimaging and light-harvesting. Nanoscale 7, 18198–18210 (2015).
Reisch, A., Runser, A., Arntz, Y., Mely, Y. & Klymchenko, A. S. Charge-controlled nanoprecipitation as a modular approach to ultrasmall polymer nanocarriers: making bright and stable nanoparticles. ACS Nano 9, 5104–5116 (2015).
Roy, R., Hohng, S. & Ha, T. A practical guide to single-molecule FRET. Nat. Methods 5, 507–516 (2008).
Zhao, Q., Young, I. T. & de Jong, J. G. S. Photon budget analysis for fluorescence lifetime imaging microscopy. J. Biomed. Opt. 16, 086007 (2011).
Chen, P. Z. et al. Light-harvesting systems based on organic nanocrystals to mimic chlorosomes. Angew. Chem. Int. Ed. 55, 2759–2763 (2016).
Patra, A., Jana, B. & Bhattacharyya, S. Functionalized dye encapsulated polymer nanoparticle attached with BSA scaffold as efficient antenna materials for artificial light harvesting. Nanoscale 8, 16034–16043 (2016).
Beljonne, D., Curutchet, C., Scholes, G. D. & Silbey, R. J. Beyond Förster resonance energy transfer in biological and nanoscale systems. J. Phys. Chem. B 113, 6583–6599 (2009).
Collini, E. Spectroscopic signatures of quantum-coherent energy transfer. Chem. Soc. Rev. 42, 4932–4947 (2013).
Collini, E. & Scholes, G. D. Coherent intrachain energy migration in a conjugated polymer at room temperature. Science 323, 369–373 (2009).
Hwang, I. & Scholes, G. D. Electronic energy transfer and quantum-coherence in π-conjugated polymers. Chem. Mater. 23, 610–620 (2011).
Engel, G. S. et al. Evidence for wavelike energy transfer through quantum coherence in photosynthetic systems. Nature 446, 782–786 (2007).
Strümpfer, J., Şener, M. & Schulten, K. How quantum coherence assists photosynthetic light-harvesting. J. Phys. Chem. Lett. 3, 536–542 (2012).
Orrit, M. & Bernard, J. Single pentacene molecules detected by fluorescence excitation in a para-terphenyl crystal. Phys. Rev. Lett. 65, 2716–2719 (1990).
Pisoni, D. S. et al. Symmetrical and asymmetrical cyanine dyes. Synthesis, spectral properties, and BSA association study. J. Org. Chem. 79, 5511–5520 (2014).
Karstens, T. & Kobs, K. Rhodamine-B and rhodamine-101 as reference substances for fluorescence quantum yield measurements. J. Phys. Chem. 84, 1871–1872 (1980).
Texier, I. et al. Cyanine-loaded lipid nanoparticles for improved in vivo fluorescence imaging. J. Biomed. Opt. 14, 054005 (2009).
Santra, K. et al. What is the best method to fit time-resolved data? A comparison of the residual minimization and the maximum likelihood techniques as applied to experimental time-correlated, single-photon counting data. J. Phys. Chem. B 120, 2484–2490 (2016).
Lakowicz, J. R. Principles of Fluorescence Spectroscopy (Springer, New York, 2006).
Acknowledgements
This work was supported by the European Research Council ERC Consolidator grant BrightSens 648528. The authors thank C. Ruhlmann from the FRISBI platform (ANR-10-INBS-05) for help with electron microscopy. K.T. was supported by a fellowship from the Ministre de la Recherche (France).
Author information
Authors and Affiliations
Contributions
A.S.K. proposed the concept. A.S.K. and K.T. designed the experiments. K.T. performed most of the experiments and data analysis. A.R. performed electron microscopy and helped with donor NP design and some data analysis. P.D. and F.F. performed time-resolved anisotropy measurements. A.S.K. helped with single-particle microscopy. A.R., Y.M., P.G. and A.S.K. contributed materials and analysis tools. A.S.K. and K.T. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
A European patent application has been filed under no. 17305763.9.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
A correction to this article is available online at https://doi.org/10.1038/s41566-017-0055-6.
Electronic supplementary material
Supplementary Information
Additional information on FRET calculation, spectroscopic, time-resolved and single-particle data, experiment set-up and other details.
Rights and permissions
About this article
Cite this article
Trofymchuk, K., Reisch, A., Didier, P. et al. Giant light-harvesting nanoantenna for single-molecule detection in ambient light. Nature Photon 11, 657–663 (2017). https://doi.org/10.1038/s41566-017-0001-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41566-017-0001-7
This article is cited by
-
Bioinspired polymeric supramolecular columns as efficient yet controllable artificial light-harvesting platform
Nature Communications (2024)
-
Energy funnelling within multichromophore architectures monitored with subnanometre resolution
Nature Chemistry (2021)
-
Picosecond time-resolved photon antibunching measures nanoscale exciton motion and the true number of chromophores
Nature Communications (2021)
-
Zeptomole Imaging of Cytosolic MicroRNA Cancer Biomarkers with A Light-Controlled Nanoantenna
Nano-Micro Letters (2021)
-
Modulation of Aggregation-Induced Emission by Excitation Energy Transfer: Design and Application
Topics in Current Chemistry (2021)