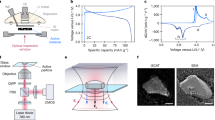
Abstract
The quality of the solid–electrolyte interphase is crucial for the performance of most battery chemistries, but its formation dynamics during operation are not well understood due to a lack of reliable operando characterization techniques. Herein, we report a dynamic, non-invasive, operando reflection interference microscope to enable the real-time imaging of the solid–electrolyte interphase during its formation and evolution processes with high sensitivity. The stratified structure of the solid–electrolyte interphase formed during four distinct steps includes the emergence of a permanent inner inorganic layer enriched in LiF, a transient assembly of an interfacial electrified double layer and a consequent emergence of a temporary outer organic-rich layer whose presence is reversible with electrochemical cycling. Reflection interference microscope imaging reveals an inverse correlation between the thicknesses of two interphasial subcomponents, implying that the permanent inorganic-rich inner layer dictates the organic-rich outer layer formation and lithium nucleation. The real-time visualization of solid–electrolyte interphase dynamics provides a powerful tool for the rational design of battery interphases.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All the data used to plot the figures are available via zenodo.org.
References
Goodenough, J. B. & Park, K. S. The Li-ion rechargeable battery: a perspective. J. Am. Chem. Soc. 135, 1167–1176 (2013).
Dunn, B., Kamath, H. & Tarascon, J. M. Electrical energy storage for the grid: a battery of choices. Science 334, 928–935 (2011).
Grande, L. et al. The lithium/air battery: still an emerging system or a practical reality? Adv. Mater. 27, 784–800 (2015).
Lu, Y. Y. et al. Stable cycling of lithium metal batteries using high transference number electrolytes. Adv. Energy Mater. 5, 1402073 (2015).
Arico, A. S., Bruce, P., Scrosati, B., Tarascon, J. M. & Van Schalkwijk, W. Nanostructured materials for advanced energy conversion and storage devices. Nat. Mater. 4, 366–377 (2005).
Sun, Y. M., Liu, N. A. & Cui, Y. Promises and challenges of nanomaterials for lithium-based rechargeable batteries. Nat. Energy 1, 16071 (2016).
Cheng, X. B. & Zhang, Q. Dendrite-free lithium metal anodes: stable solid electrolyte interphases for high-efficiency batteries. J. Mater. Chem. A 3, 7207–7209 (2015).
Tarascon, J. M. & Armand, M. Issues and challenges facing rechargeable lithium batteries. Nature 414, 359–367 (2001).
Guo, Y. P., Li, H. Q. & Zhai, T. Y. Reviving lithium-metal anodes for next-generation high-energy batteries. Adv. Mater. 29, 1700007 (2017).
Yuan, S. Y. et al. Graphene-supported nitrogen and boron rich carbon layer for improved performance of lithium-sulfur batteries due to enhanced chemisorption of lithium polysulfides. Adv. Energy Mater. 6, 1501733 (2016).
Janek, J. & Zeier, W. G. A solid future for battery development. Nat. Energy 1, 16141 (2016).
Luntz, A. C., Voss, J. & Reuter, K. Interfacial challenges in solid-state Li ion batteries. J. Phys. Chem. Lett. 6, 4599–4604 (2015).
Cao, R. G., Xu, W., Lv, D. P., Xiao, J. & Zhang, J. G. Anodes for rechargeable lithium-sulfur batteries. Adv. Energy Mater. 5, 1402273 (2015).
He, P., Zhang, T., Jiang, J. & Zhou, H. S. Lithium-air batteries with hybrid electrolytes. J. Phys. Chem. Lett. 7, 1267–1280 (2016).
Huang, C.-J. et al. Decoupling the origins of irreversible coulombic efficiency in anode-free lithium metal batteries. Nat. Commun. 12, 1452. (2021).
Qiao, Y. et al. A high-energy-density and long-life initial-anode-free lithium battery enabled by a Li2O sacrificial agent. Nat. Energy 6, 653–662 (2021).
Xu, K. Nonaqueous liquid electrolytes for lithium-based rechargeable batteries. Chem. Rev. 104, 4303–4417 (2004).
Peled, E. The electrochemical behavior of alkali and alkaline earth metals in nonaqueous battery systems—the solid electrolyte interphase model. J. Electrochem. Soc. 126, 2047 (1979).
Gauthier, M. et al. Electrode–electrolyte interface in Li-ion batteries: current understanding and new insights. J. Phys. Chem. Lett. 6, 4653–4672 (2015).
An, S. J. et al. The state of understanding of the lithium-ion-battery graphite solid electrolyte interphase (SEI) and its relationship to formation cycling. Carbon 105, 52–76 (2016).
Orsini, F. et al. In situ scanning electron microscopy (SEM) observation of interfaces within plastic lithium batteries. J. Power Sources 76, 19–29 (1998).
Mehdi, B. L. et al. Observation and quantification of nanoscale processes in lithium batteries by operando electrochemical (S)TEM. Nano Lett. 15, 2168–2173 (2015).
Nie, M. Y. et al. Lithium ion battery graphite solid electrolyte interphase revealed by microscopy and spectroscopy. J. Phys. Chem. C 117, 1257–1267 (2013).
Wang, L. et al. Identifying the components of the solid–electrolyte interphase in Li-ion batteries. Nat. Chem. 11, 789–796 (2019).
Ai, Q. et al. Lithium-conducting covalent-organic-frameworks as artificial solid-electrolyte-interphase on silicon anode for high performance lithium ion batteries. Nano Energy 72, 104657 (2020).
Zeng, X. et al. Electrolyte design for in situ construction of highly Zn2+‐conductive solid electrolyte interphase to enable high‐performance aqueous Zn‐ion batteries under practical conditions. Adv. Mater. 33, 2007416 (2021).
Nanda, J. et al. Unraveling the nanoscale heterogeneity of solid electrolyte interphase using tip-enhanced Raman spectroscopy. Joule 3, 2001–2019 (2019).
Chen, D. et al. Origin of extra capacity in the solid electrolyte interphase near high-capacity iron carbide anodes for Li ion batteries. Energy Environ. Sci. 13, 2924–2937 (2020).
Qiu, H. et al. Zinc anode-compatible in-situ solid electrolyte interphase via cation solvation modulation. Nat. Commun. 10, 5374 (2019).
Wood, K. N. et al. Operando X-ray photoelectron spectroscopy of solid electrolyte interphase formation and evolution in Li2S-P2S5 solid-state electrolytes. Nat. Commun. 9, 2490 (2018).
Cheng, D. et al. Unveiling the stable nature of the solid electrolyte interphase between lithium metal and LiPON via cryogenic electron microscopy. Joule 4, 2484–2500 (2020).
Cao, C. et al. Solid electrolyte interphase on native oxide-terminated silicon anodes for Li-ion batteries. Joule 3, 762–781 (2019).
Zhou, Y. et al. Real-time mass spectrometric characterization of the solid–electrolyte interphase of a lithium-ion battery. Nat. Nanotechnol. 15, 224–230 (2020).
Li, Y. Z. et al. Atomic structure of sensitive battery materials and interfaces revealed by cryo-electron microscopy. Science 358, 506–510 (2017).
Wang, X. F. et al. New insights on the structure of electrochemically deposited lithium metal and its solid electrolyte interphases via cryogenic TEM. Nano Lett. 17, 7606–7612 (2017).
Wan, J. et al. Ultra-thin solid electrolyte interphase evolution and wrinkling processes in molybdenum disulfide-based lithium-ion batteries. Nat. Commun. 10, 3265 (2019).
von Cresce, A., Russell, S. M., Baker, D. R., Gaskell, K. J. & Xu, K. In situ and quantitative characterization of solid electrolyte interphases. Nano Lett. 14, 1405–1412 (2014).
Pathak, R. et al. Fluorinated hybrid solid-electrolyte-interphase for dendrite-free lithium deposition. Nat. Commun. 11, 93 (2020).
Liu, T. et al. In situ quantification of interphasial chemistry in Li-ion battery. Nat. Nanotechnol. 14, 50–56 (2019).
Nie, M. et al. Role of solution structure in solid electrolyte interphase formation on graphite with LiPF6 in propylene carbonate. J. Phys. Chem. C 117, 25381–25389 (2013).
Qian, J. et al. Dendrite-free Li deposition using trace-amounts of water as an electrolyte additive. Nano Energy 15, 135–144 (2015).
Shen, C. et al. Li2O-reinforced solid electrolyte interphase on three-dimensional sponges for dendrite-free lithium deposition. Front. Chem. 6, 517 (2018).
Terborg, L. et al. Ion chromatographic determination of hydrolysis products of hexafluorophosphate salts in aqueous solution. Anal. Chim. Acta 714, 121–126 (2012).
Shi, Y. et al. Electrochemical impedance imaging on conductive surfaces. Anal. Chem. 93, 12320–12328 (2021).
Foley, K. J., Shan, X. & Tao, N. J. Surface impedance imaging technique. Anal. Chem. 80, 5146–5151 (2008).
Aurbach, D., Daroux, M., Faguy, P. & Yeager, E. Identification of surface films formed on lithium in propylene carbonate solutions. J. Electrochem. Soc. 134, 1611 (1987).
Xing, L. et al. Theoretical investigations on oxidative stability of solvents and oxidative decomposition mechanism of ethylene carbonate for lithium ion battery use. J. Phys. Chem. B 113, 16596–16602 (2009).
Kanamura, K. et al. Oxidation of propylene carbonate containing LiBF4 or LiPF6 on LiCoO2 thin film electrode for lithium batteries. Electrochim. Acta 47, 433–439 (2001).
von Cresce, A. et al. In situ and quantitative characterization of solid electrolyte interphases. Nano Lett. 14, 1405–1412 (2014).
Aurbach, D. et al. The study of electrolyte solutions based on ethylene and diethyl carbonates for rechargeable Li batteries: I. Li metal anodes. J. Electrochem. Soc. 142, 2873 (1995).
Parimalam, B. S., MacIntosh, A. D., Kadam, R. & Lucht, B. L. Decomposition reactions of anode solid electrolyte interphase (SEI) components with LiPF6. J. Phys. Chem. C 121, 22733–22738 (2017).
Li, Y. et al. Atomic structure of sensitive battery materials and interfaces revealed by cryo–electron microscopy. Science 358, 506–510 (2017).
Huang, W., Wang, H., Boyle, D. T., Li, Y. & Cui, Y. Resolving nanoscopic and mesoscopic heterogeneity of fluorinated species in battery solid-electrolyte interphases by cryogenic electron microscopy. ACS Energy Lett. 5, 1128–1135 (2020).
Wang, X. et al. New insights on the structure of electrochemically deposited lithium metal and its solid electrolyte interphases via cryogenic TEM. Nano Lett. 17, 7606–7612 (2017).
Wang, A., Kadam, S., Li, H., Shi, S. & Qi, Y. Review on modeling of the anode solid electrolyte interphase (SEI) for lithium-ion batteries. npj Comput. Mater. 4, 15 (2018).
Leung, K. & Jungjohann, K. J. Spatial heterogeneities and onset of passivation breakdown at lithium anode interfaces. J. Phys. Chem. C 121, 20188–20196 (2017).
Zhang, Y. et al. Dendrite-free lithium deposition with self-aligned nanorod structure. Nano Lett. 14, 6889–6896 (2014).
Kasse, R. M. et al. Understanding additive controlled lithium morphology in lithium metal batteries. J. Mater. Chem. A 8, 16960–16972 (2020).
Acknowledgements
X.S. acknowledges the funding support from the start-up fund at the University of Houston, the University of Houston’s Center for Carbon Management in Energy, UL Research Institutes, Beyond Bits Technology, the US Department of Agriculture’s Small Business Innovation Research programme (awards no. 2022-70012-36900 and no. 2019-33610-29769) and University Training and Research for Fossil Energy Applications (US Department of Energy DE-FE-0032092). The work performed at Pacific Northwest National Laboratory (PNNL) was supported by the Assistant Secretary for Energy Efficiency and Renewable Energy, and Vehicle Technologies Office of the US Department of Energy through the Advanced Battery Materials Research programme under contract no. DE-AC05-76RL01830. PNNL is operated by Battelle for the US Department of Energy under contract no. DE-AC05-76RL01830. Y.Y. acknowledges the funding support from the US Department of Energy’s Office of Energy Efficiency and Renewable Energy under the Vehicle Technologies Program under contract no. DE-EE0008864. K.X. acknowledges the support from the Joint Center for Energy Storage Research, an energy hub funded by the US Department of Energy’s Basic Energy Sciences programme. X.S. thanks Z. Zhu from PNNL for his insightful discussions of SEI formation, and thanks P. Ruchhoeft for providing the thermal evaporator for the electrode fabrication.
Author information
Authors and Affiliations
Contributions
X.S., W.X., Y.Y. and K.X. conceived the idea and designed the research. X.S. supervised the research. G.F. carried out and managed most of the experiments, including performing all the RIM measurements on SEI formation and Li nucleation, characterizing the SEI chemical compositions using XPS and conducting atomic force microscopy and profilometer calibrations. G.F. analysed the data and prepared the figures. H.J. prepared the electrolytes, electrochemical depositions of SEI and Li on Cu foils and samples for XPS depth profiling. Y.S. helped with RIM instrument development and data analysis. M.H.E. performed the XPS depth profiling experiment and provided insight to understand the data. X.Y., Y.Z., Y.L. and C.Y. provided suggestions on the experiments. Y.L. provided insight on understanding the RIM response. X.S., G.F., W.X., Y.Y. and K.X. discussed the data and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
X.S. has equity interest in Pani Clean, Inc. Y.Y. has equity interest in LiBeyond, LLC and Solid Design Instruments, LLC. Y.L. has equity interest in LiBeyond, LLC. The University of Houston reviewed and approved their relationship in compliance with its conflict-of-interest policy. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Nanotechnology thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–21, Tables 1 and 2 and Discussion Sections 1–9.
Supplementary Video 1
The formation dynamics of the LiF-rich SEI layer (thickness map) during the CV scanning process in the potential window of 2.2–1.4 V.
Supplementary Video 2
The formation dynamics of the organic-rich SEI layer (thickness map) during the CV scanning process in the potential window of 0.8–0.1 V.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Feng, G., Jia, H., Shi, Y. et al. Imaging solid–electrolyte interphase dynamics using operando reflection interference microscopy. Nat. Nanotechnol. 18, 780–789 (2023). https://doi.org/10.1038/s41565-023-01316-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-023-01316-3
This article is cited by
-
Three-dimensional operando optical imaging of particle and electrolyte heterogeneities inside Li-ion batteries
Nature Nanotechnology (2023)