Abstract
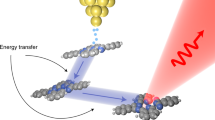
Nature controls the assembly of complex architectures through self-limiting processes; however, few artificial strategies to mimic these processes have been reported to date. Here we demonstrate a system comprising two types of nanocrystal (NC), where the self-limiting assembly of one NC component controls the aggregation of the other. Our strategy uses semiconducting InP/ZnS core–shell NCs (3 nm) as effective assembly modulators and functional nanoparticle surfactants in cucurbit[n]uril-triggered aggregation of AuNCs (5–60 nm), allowing the rapid formation (within seconds) of colloidally stable hybrid aggregates. The resultant assemblies efficiently harvest light within the semiconductor substructures, inducing out-of-equilibrium electron transfer processes, which can now be simultaneously monitored through the incorporated surface-enhanced Raman spectroscopy–active plasmonic compartments. Spatial confinement of electron mediators (for example, methyl viologen (MV2+)) within the hybrids enables the direct observation of photogenerated radical species as well as molecular recognition in real time, providing experimental evidence for the formation of elusive σ–(MV+)2 dimeric species. This approach paves the way for widespread use of analogous hybrids for the long-term real-time tracking of interfacial charge transfer processes, such as the light-driven generation of radicals and catalysis with operando spectroscopies under irreversible conditions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Methods and materials characterization are provided in the Supplementary Information. The data that support the findings of this study are available from the corresponding author on reasonable request.
References
Jiang, R., Li, B., Fang, C. & Wang, J. Metal/semiconductor hybrid nanostructures for plasmon-enhanced applications. Adv. Mater. 26, 5274–5309 (2014).
Lim, E.-K. et al. Nanomaterials for theranostics: recent advances and future challenges. Chem. Rev. 115, 327–394 (2015).
Boles, M., Engel, M. & Talapin, D. Self-assembly of colloidal nanocrystals: from intricate structures to functional materials. Chem. Rev. 116, 11220–11289 (2016).
Boles, M., Ling, D., Hyeon, T. & Talapin, D. The surface science of nanocrystals. Nat. Mater. 15, 141–153 (2016).
Xia, Y. et al. Self-assembly of self-limiting monodisperse supraparticles from polydisperse nanoparticles. Nat. Nanotechnol. 6, 580–587 (2011).
Kalsin, A. et al. Electrostatic self-assembly of binary nanoparticle crystals with a diamond-like lattice. Science 312, 420–424 (2006).
André, A. et al. Toward conductive mesocrystalline assemblies: PbS nanocrystals cross-linked with tetrathiafulvalene dicarboxylate. Chem. Mater. 27, 8105–8115 (2015).
Connolly, S. & Fitzmaurice, D. Programmed assembly of gold nanocrystals in aqueous solution. Adv. Mater. 11, 1202–1205 (1999).
Macfarlane, R., O’Brien, M., Petrosko, S. & Mirkin, C. Nucleic acid-modified nanostructures as programmable atom equivalents: forging a new ‘table of elements’. Angew. Chem. Int. Ed. 52, 5688–5698 (2013).
Wang, Y. et al. Host–guest chemistry with water-soluble gold nanoparticle supraspheres. Nat. Nanotechnol. 12, 170–176 (2017).
Sánchez-Iglesias, A. et al. Hydrophobic interactions modulate self-assembly of nanoparticles. ACS Nano 6, 11059–11065 (2012).
Choueiri, R. M., Klinkova, A., Thérien-Aubin, H., Rubinstein, M. & Kumacheva, E. Structural transitions in nanoparticle assemblies governed by competing nanoscale forces. J. Am. Chem. Soc. 135, 10262–10265 (2013).
Jia, G. et al. Couples of colloidal semiconductor nanorods formed by self-limited assembly. Nat. Mater. 13, 301–307 (2014).
de Q. Silveira, G. et al. Supraparticle nanoassemblies with enzymes. Chem. Mater. 31, 7493–7500 (2019).
Banin, U. & Sitt, A. Colloidal self-assembly: superparticles get complex. Nat. Mater. 11, 1009–1011 (2012).
Yi, C. et al. Self-limiting directional nanoparticle bonding governed by reaction stoichiometry. Science 369, 1369–1374 (2020).
Yao, G. et al. Programming nanoparticle valence bonds with single-stranded DNA encoders. Nat. Mater. 19, 781–788 (2020).
Jehannin, M., Rao, A. & Cölfen, H. New horizons of nonclassical crystallization. J. Am. Chem. Soc. 141, 10120–10136 (2019).
Taylor, R. et al. Precise subnanometer plasmonic junctions for SERS within gold nanoparticle assemblies using cucurbit[n]uril ‘glue’. ACS Nano 5, 3878–3887 (2011).
Barrow, S., Kasera, S., Rowland, M., Del Barrio, J. & Scherman, O. A. Cucurbituril-based molecular recognition. Chem. Rev. 115, 12320–12406 (2015).
Chikkaraddy, R. et al. Single-molecule strong coupling at room temperature in plasmonic nanocavities. Nature 535, 127–130 (2016).
Jing, L. et al. Aqueous based semiconductor nanocrystals. Chem. Rev. 116, 10623–10730 (2016).
Ni, X.-L. et al. Cucurbit[n]uril-based coordination chemistry: from simple coordination complexes to novel poly-dimensional coordination polymers. Chem. Soc. Rev. 42, 9480–9508 (2013).
Ziegler, C. et al. Modern inorganic aerogels. Angew. Chem. Int. Ed. 56, 13200–13221 (2017).
Huang, F., Zhang, H. & Banfield, J. F. Two-stage crystal-growth kinetics observed during hydrothermal coarsening of nanocrystalline ZnS. Nano Lett. 3, 373–378 (2003).
Yang, Z., Wei, J., Sobolev, Y. I. & Grzybowski, B. A. Systems of mechanized and reactive droplets powered by multi-responsive surfactants. Nature 553, 313–318 (2018).
Kowalczyk, B. et al. Charged nanoparticles as supramolecular surfactants for controlling the growth and stability of microcrystals. Nat. Mater. 11, 227–232 (2012).
Liu, X. et al. Reconfigurable ferromagnetic liquid droplets. Science 365, 264–267 (2019).
Cui, M., Emrick, T. & Russell, T. P. Stabilizing liquid drops in nonequilibrium shapes by the interfacial jamming of nanoparticles. Science 342, 460–463 (2013).
Silvera Batista, C. A., Larson, R. G. & Kotov, N. A. Nonadditivity of nanoparticle interactions. Science 350, 176–187 (2015).
Homola, J. Surface plasmon resonance sensors for detection of chemical and biological species. Chem. Rev. 108, 462–493 (2008).
Wang, Y. et al. Self-assembly and structure of directly imaged inorganic-anion monolayers on a gold nanoparticle. J. Am. Chem. Soc. 131, 17412–17422 (2009).
Monk, P. M. S. The Viologens: Physicochemical Properties, Synthesis and Applications of the Salts of 4,4'-Bipyridine (Wiley-VCH, 1999).
Striepe, L. & Baumgartner, T. Viologens and their application as functional materials. Chem. Eur. J. 23, 16924–16940 (2017).
McCune, J. A., Kuehnel, M. F., Reisner, E. & Scherman, O. A. Stimuli-mediated ultrastable radical formation. Chem 6, 1819–1830 (2020).
Trabolsi, A. et al. Radically enhanced molecular recognition. Nat. Chem. 2, 42–49 (2010).
Bruns, C. J. & Stoddart, J. F. The Nature of the Mechanical Bond: From Molecules to Machines (Wiley-VCH, 2016).
Cieślak, A. M. et al. Photo-induced interfacial electron transfer of ZnO nanocrystals to control supramolecular assembly in water. Nanoscale 9, 16128–16132 (2017).
Geraskina, M., Dutton, A., Juetten, M., Wood, S. & Winter, A. The viologen cation radical pimer: a case of dispersion-driven bonding. Angew. Chem. Int. Ed. 56, 9435–9439 (2017).
Kim, H.-J., Jeon, W. S., Ko, Y. H. & Kim, K. Inclusion of methylviologen in cucurbit[7]uril. Proc. Natl Acad. Sci. USA 99, 5007–5011 (2002).
Meisel, D., Mulac, W. A. & Matheson, M. S. Catalysis of methyl viologen radical reactions by polymer-stabilized gold sols. J. Phys. Chem. 85, 179–187 (1981).
Bockman, T. M. & Kochi, J. K. Isolation and oxidation-reduction of methylviologen cation radicals. Novel disproportionation in charge-transfer salts by X-ray crystallography. J. Org. Chem. 55, 4127–4135 (1990).
Norton, J. D. & White, H. S. Effect of comproportionation on the voltammetric reduction of methyl viologen in low ionic strength solutions. J. Electroanal. Chem. 325, 341–350 (1992).
Kim, J. et al. New cucurbituril homologues: syntheses, isolation, characterization and X-ray crystal structures of cucurbit[n]uril (n = 5, 7, and 8). J. Am. Chem. Soc. 122, 540–541 (2000).
Xie, R., Battaglia, D. & Peng, X. Colloidal InP nanocrystals as efficient emitters covering blue to near-infrared. J. Am. Chem. Soc. 129, 15432–15433 (2007).
Kimling, J. et al. Turkevich method for gold nanoparticle synthesis revisited. J. Phys. Chem. B 110, 15700–15707 (2006).
Chai, J.-D. & Head-Gordon, M. Long-range corrected hybrid density functionals with damped atom–atom dispersion corrections. Phys. Chem. Chem. Phys. 10, 6615–6620 (2008).
Marenich, A. V., Cramer, C. J. & Truhlar, D. G. Universal solvation model based on solute electron density and on a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J. Phys. Chem. B 113, 6378–6396 (2009).
Frisch, M. J. et al. Gaussian09 revision E.01 (Gaussian Inc., 2009).
Acknowledgements
We acknowledge financial support from EPSRC grant nos. EP/L027151/1 (NOtCH) and EP/R020965/1 (RaNT). J.H. is thankful for support from the Chinese Scholarship Council and Cambridge Commonwealth, European and International Trust. B.d.N. acknowledges support from the Leverhulme Trust and Isaac Newton Trust. R.C. acknowledges support from Trinity College, Cambridge. S.M.C. thanks Girton College, Cambridge, for a Henslow Research Fellowship. We thank S. J. Barrow, A. S. Groombridge and I. Szabó for helpful discussions. We acknowledge use of the research computing facility at King’s College London, Rosalind (https://rosalind.kcl.ac.uk).
Author information
Authors and Affiliations
Contributions
K.S. and O.A.S. conceived the project and developed the experiments. K.S. developed and prepared the materials. K.S. and J.A.M. carried out the mechanistic studies on self-limiting aggregation. K.S. and O.A.S. proposed the aggregation mechanism. D.D.X. carried out the zeta potential measurements. K.S. and J.H. carried out SERS experiments. K.S., J.H., B.d.N. and J.J.B. analysed the SERS data while K.S. and O.A.S. provided interpretation and proposed a mechanism for the photochemical transformations. T.F. and E.R. carried out the theoretical calculations. K.S. and S.M.C. carried out TEM experiments. R.C. carried out calculations on the optical properties of AuNC aggregates while R.C., J.H., B.d.N. and J.J.B. provided their interpretation. K.S., J.A.M. and O.A.S. analysed the data and wrote the manuscript with input from all co-authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Nanotechnology thanks Hongyu Chen, Zhihong Nie and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information
Supplementary Figs. 1–47, Table 1, discussion, materials and methods, and coordinates for the optimized structure.
Rights and permissions
About this article
Cite this article
Sokołowski, K., Huang, J., Földes, T. et al. Nanoparticle surfactants for kinetically arrested photoactive assemblies to track light-induced electron transfer. Nat. Nanotechnol. 16, 1121–1129 (2021). https://doi.org/10.1038/s41565-021-00949-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-021-00949-6