Abstract
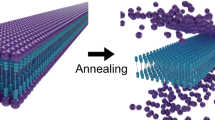
Small-molecule self-assembly is an established route for producing high-surface-area nanostructures with readily customizable chemistries and precise molecular organization. However, these structures are fragile, exhibiting molecular exchange, migration and rearrangement—among other dynamic instabilities—and are prone to dissociation upon drying. Here we show a small-molecule platform, the aramid amphiphile, that overcomes these dynamic instabilities by incorporating a Kevlar-inspired domain into the molecular structure. Strong, anisotropic interactions between aramid amphiphiles suppress molecular exchange and elicit spontaneous self-assembly in water to form nanoribbons with lengths of up to 20 micrometres. Individual nanoribbons have a Young’s modulus of 1.7 GPa and tensile strength of 1.9 GPa. We exploit this stability to extend small-molecule self-assembly to hierarchically ordered macroscopic materials outside of solvated environments. Through an aqueous shear alignment process, we organize aramid amphiphile nanoribbons into arbitrarily long, flexible threads that support 200 times their weight when dried. Tensile tests of the dry threads provide a benchmark for Young’s moduli (between ~400 and 600 MPa) and extensibilities (between ~0.6 and 1.1%) that depend on the counterion chemistry. This bottom-up approach to macroscopic materials could benefit solid-state applications historically inaccessible by self-assembled nanomaterials.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data generated and analysed during this study are available from the corresponding author on reasonable request.
References
Whitesides, G. M., Mathias, J. P. & Seto, C. T. Molecular self-assembly and nanochemistry: a chemical strategy for the synthesis of nanostructures. Science 254, 1312–1319 (1991).
Aida, T., Meijer, E. & Stupp, S. I. Functional supramolecular polymers. Science 335, 813–817 (2012).
Zhang, S. et al. A self-assembly pathway to aligned monodomain gels. Nat. Mater. 9, 594–601 (2010).
Koutsopoulos, S., Unsworth, L. D., Nagai, Y. & Zhang, S. Controlled release of functional proteins through designer self-assembling peptide nanofiber hydrogel scaffold. Proc. Natl Acad. Sci. USA 106, 4623–4628 (2009).
Tantakitti, F. et al. Energy landscapes and functions of supramolecular systems. Nat. Mater. 15, 469–476 (2016).
Ortony, J. H. et al. Internal dynamics of a supramolecular nanofibre. Nat. Mater. 13, 812–816 (2014).
Schief, W., Touryan, L., Hall, S. & Vogel, V. Nanoscale topographic instabilities of a phospholipid monolayer. J. Phys. Chem. B 104, 7388–7393 (2000).
da Silva, R. M. et al. Super-resolution microscopy reveals structural diversity in molecular exchange among peptide amphiphile nanofibres. Nat. Commun. 7, 11561 (2016).
Wimley, W. C. & Thompson, T. E. Transbilayer and interbilayer phospholipid exchange in dimyristoylphosphatidylcholine/dimyristoylphosphatidylethanolamine large unilamellar vesicles. Biochemistry 30, 1702–1709 (1991).
Ortony, J. H. et al. Water dynamics from the surface to the interior of a supramolecular nanostructure. J. Am. Chem. Soc. 139, 8915–8921 (2017).
Yuan, D., Shi, J., Du, X., Zhou, N. & Xu, B. Supramolecular glycosylation accelerates proteolytic degradation of peptide nanofibrils. J. Am. Chem. Soc. 137, 10092–10095 (2015).
Toledano, S., Williams, R. J., Jayawarna, V. & Ulijn, R. V. Enzyme-triggered self-assembly of peptide hydrogels via reversed hydrolysis. J. Am. Chem. Soc. 128, 1070–1071 (2006).
Freeman, R. et al. Reversible self-assembly of superstructured networks. Science 362, 808–813 (2018).
Williams, R. J. et al. Enzyme-assisted self-assembly under thermodynamic control. Nat. Nanotechnol. 4, 19–24 (2009).
Hashim, P., Bergueiro, J., Meijer, E. & Aida, T. Supramolecular polymerization: a conceptual expansion for innovative materials. Prog. Polym. Sci. 105, 101250 (2020).
Xu, Y. et al. Nanostructured polymer films with metal-like thermal conductivity. Nat. Commun. 10, 1771 (2019).
Tuller, H. L. Ionic conduction in nanocrystalline materials. Solid State Ion. 131, 143–157 (2000).
Sherrington, D. C. & Taskinen, K. A. Self-assembly in synthetic macromolecular systems via multiple hydrogen bonding interactions. Chem. Soc. Rev. 30, 83–93 (2001).
Dobb, M., Johnson, D. & Saville, B. Supramolecular structure of a high-modulus polyaromatic fiber (Kevlar 49). J. Polym. Sci. Polym. Phys. Ed. 15, 2201–2211 (1977).
Seyler, H., Storz, C., Abbel, R. & Kilbinger, A. F. A facile synthesis of aramide–peptide amphiphiles. Soft Matter 5, 2543–2545 (2009).
Claussen, R. C., Rabatic, B. M. & Stupp, S. I. Aqueous self-assembly of unsymmetric peptide bolaamphiphiles into nanofibers with hydrophilic cores and surfaces. J. Am. Chem. Soc. 125, 12680–12681 (2003).
Yang, M. et al. Dispersions of aramid nanofibers: a new nanoscale building block. ACS Nano 5, 6945–6954 (2011).
Schleuss, T. W. et al. Hockey-puck micelles from oligo(p-benzamide)-b-PEG rod–coil block copolymers. Angew. Chem. Int. Ed. 45, 2969–2975 (2006).
Bohle, A. et al. Hydrogen-bonded aggregates of oligoaramide−poly(ethylene glycol) block copolymers. Macromolecules 43, 4978–4985 (2010).
Abbel, R., Schleuss, T. W., Frey, H. & Kilbinger, A. F. M. Rod-length dependent aggregation in a series of oligo(p-benzamide)-block-poly(ethylene glycol) rod-coil copolymers. Macromol. Chem. Phys. 206, 2067–2074 (2005).
Johansson, A., Kollman, P., Rothenberg, S. & McKelvey, J. Hydrogen bonding ability of the amide group. J. Am. Chem. Soc. 96, 3794–3800 (1974).
Dixon, D. A., Dobbs, K. D. & Valentini, J. J. Amide-water and amide-amide hydrogen bond strengths. J. Phys. Chem. 98, 13435–13439 (1994).
Kline, S. R. Reduction and analysis of SANS and USANS data using IGOR Pro. J. Appl. Crystallogr. 39, 895–900 (2006).
Nallet, F., Laversanne, R. & Roux, D. Modelling X-ray or neutron scattering spectra of lyotropic lamellar phases: interplay between form and structure factors. J. Phys. II 3, 487–502 (1993).
Mertens, H. D. & Svergun, D. I. Structural characterization of proteins and complexes using small-angle X-ray solution scattering. J. Struct. Biol. 172, 128–141 (2010).
Yokoi, H., Kinoshita, T. & Zhang, S. Dynamic reassembly of peptide RADA16 nanofiber scaffold. Proc. Natl Acad. Sci. USA 102, 8414–8419 (2005).
Hartgerink, J. D., Beniash, E. & Stupp, S. I. Self-assembly and mineralization of peptide-amphiphile nanofibers. Science 294, 1684–1688 (2001).
Cravotto, G. & Cintas, P. Molecular self-assembly and patterning induced by sound waves. The case of gelation. Chem. Soc. Rev. 38, 2684–2697 (2009).
Gorelik, T. E., van de Streek, J., Kilbinger, A. F., Brunklaus, G. & Kolb, U. Ab-initio crystal structure analysis and refinement approaches of oligo p-benzamides based on electron diffraction data. Acta Crystallogr. B Struct. Sci. 68, 171–181 (2012).
Gorelik, T. et al. H-bonding schemes of di- and tri-p-benzamides assessed by a combination of electron diffraction, X-ray powder diffraction and solid-state NMR. CrystEngComm 12, 1824–1832 (2010).
Wang, J., Liu, K., Xing, R. & Yan, X. Peptide self-assembly: thermodynamics and kinetics. Chem. Soc. Rev. 45, 5589–5604 (2016).
Barth, A. Infrared spectroscopy of proteins. Biochim. Biophys. Acta Bioenerg. 1767, 1073–1101 (2007).
Zandomeneghi, G., Krebs, M. R., McCammon, M. G. & Fändrich, M. FTIR reveals structural differences between native β‐sheet proteins and amyloid fibrils. Protein Sci. 13, 3314–3321 (2004).
Matayoshi, E. D., Wang, G. T., Krafft, G. A. & Erickson, J. Novel fluorogenic substrates for assaying retroviral proteases by resonance energy transfer. Science 247, 954–958 (1990).
Wu, B., Heidelberg, A. & Boland, J. J. Mechanical properties of ultrahigh-strength gold nanowires. Nat. Mater. 4, 525–529 (2005).
Smith, J. F., Knowles, T. P., Dobson, C. M., MacPhee, C. E. & Welland, M. E. Characterization of the nanoscale properties of individual amyloid fibrils. Proc. Natl Acad. Sci. USA 103, 15806–15811 (2006).
Knowles, T. P. et al. Role of intermolecular forces in defining material properties of protein nanofibrils. Science 318, 1900–1903 (2007).
Lamour, G., Kirkegaard, J. B., Li, H., Knowles, T. P. & Gsponer, J. Easyworm: an open-source software tool to determine the mechanical properties of worm-like chains. Source Code Biol. Med. 9, 16 (2014).
Huang, Y. Y., Knowles, T. P. & Terentjev, E. M. Strength of nanotubes, filaments, and nanowires from sonication‐induced scission. Adv. Mater. 21, 3945–3948 (2009).
Nassar, R., Wong, E., Gsponer, J. & Lamour, G. Inverse correlation between amyloid stiffness and size. J. Am. Chem. Soc. 141, 58–61 (2019).
Peng, Z. et al. High tensile strength of engineered β-solenoid fibrils via sonication and pulling. Biophys. J. 113, 1945–1955 (2017).
Santos, H. M., Lodeiro, C. & Capelo-Martínez, J.-L. in Ultrasound in Chemistry: Analytical Applications 1–16 (Wiley Online Library, 2009).
Lamour, G. et al. Mapping the broad structural and mechanical properties of amyloid fibrils. Biophys. J. 112, 584–594 (2017).
Zhao, X. et al. Molecular self-assembly and applications of designer peptide amphiphiles. Chem. Soc. Rev. 39, 3480–3498 (2010).
Niece, K. L., Hartgerink, J. D., Donners, J. J. & Stupp, S. I. Self-assembly combining two bioactive peptide-amphiphile molecules into nanofibers by electrostatic attraction. J. Am. Chem. Soc. 125, 7146–7147 (2003).
Angeloni, N. L. et al. Regeneration of the cavernous nerve by Sonic hedgehog using aligned peptide amphiphile nanofibers. Biomaterials 32, 1091–1101 (2011).
Fink, L., Steiner, A., Szekely, O., Szekely, P. & Raviv, U. Structure and interactions between charged lipid membranes in the presence of multivalent ions. Langmuir 35, 9694–9703 (2019).
Knowles, T. P. & Buehler, M. J. Nanomechanics of functional and pathological amyloid materials. Nat. Nanotechnol. 6, 469–479 (2011).
Bradbury, R. & Nagao, M. Effect of charge on the mechanical properties of surfactant bilayers. Soft Matter 12, 9383–9390 (2016).
Takahashi, Y., Ozaki, Y., Takase, M. & Krigbaum, W. Crystal structure of poly(p‐benzamide). J. Polym. Sci. B Polym. Phys. 31, 1135–1143 (1993).
Paramonov, S. E., Jun, H.-W. & Hartgerink, J. D. Self-assembly of peptide−amphiphile nanofibers: the roles of hydrogen bonding and amphiphilic packing. J. Am. Chem. Soc. 128, 7291–7298 (2006).
Russell, P. Photonic crystal fibers. Science 299, 358–362 (2003).
Lindemann, W. R., Christoff-Tempesta, T. & Ortony, J. H. A global minimization toolkit for batch-fitting and χ2 cluster analysis of CW-EPR spectra. Biophys. J. 119, 1937–1945 (2020).
Acknowledgements
We thank E. Deiss-Yehiely and C. Settens for their helpful input. We thank R. Allen and L. Hopkins for contributing graphics shown in the figures. We acknowledge J. Tian and S. Kallakuri for contributions to synthesis of early stage AAs that led to the molecular designs incorporated in this report. This material is based upon work supported by the National Science Foundation under grant no. CHE-1945500. This work was supported in part by the Professor Amar G. Bose Research Grant Program, the Abdul Latif Jameel Water and Food Systems Lab, and the MIT Center for Environmental Health Sciences under NIH Center grant P30-ES002109. D.-Y.K. acknowledges the support of the National Research Foundation of Korea’s Basic Science Research Program and Chonbuk National University Fellowship Program. T.C.-T. and W.R.L. acknowledge the support of the National Science Foundation Graduate Research Fellowship Program under grant no. 1122374. T.C.-T. acknowledges the support of the Martin Family Society of Fellows for Sustainability. G.L. acknowledges support from the Université d’Evry-Paris Saclay. This work made use of the MRSEC Shared Experimental Facilities at MIT supported by the National Science Foundation under award number DMR-14-19807 and the MIT Department of Chemistry Instrumentation Facility. X-ray scattering measurements were performed at beamline 12-ID-B of the Advanced Photon Source, a US Department of Energy Office of Science User Facility operated for the US Department of Energy Office of Science by Argonne National Laboratory under contract no. DE-AC02-06CH11357. This work was performed in part at the Harvard University Center for Nanoscale Systems cryo-TEM facility, a member of the National Nanotechnology Coordinated Infrastructure Network, which is supported by the National Science Foundation under award no. 1541959.
Author information
Authors and Affiliations
Contributions
T.C.-T., D.-Y. K. and J.H.O. conceived and designed the experiments. D.-Y.K. and T.C.-T. synthesized materials with assistance from W.R.L. and A.J.L.; T.C.-T. and D.-Y.K. performed chemical characterization of all samples. T.C.-T. performed conventional TEM and cryo-TEM. Y.C. and T.C.-T. performed SEM. G.L. performed AFM and statistical topographical analyses. G.L. performed sonication-induced scission measurements, imaging with AFM and TEM, and analysis of data. A.J.L. performed FRET measurements and analysis of the data. X.Z., T.C.-T. and Y.C. performed solution X-ray scattering and analysis of the data. A.J.L. and Y.C. conceptualized nanoribbon thread processing, and Y.C. and M.G. prepared nanoribbon threads. M.G. performed tensile testing of nanoribbon threads and analysis of the data. T.C.-T. and Y.C. performed X-ray scattering of solid-state nanoribbon threads and analysis of the data. J.H.O., T.C.-T and Y.C. cowrote the manuscript. J.H.O. provided project administration, funding acquisition and supervision. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Nanotechnology thanks the anonymous reviewers for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–37, Schemes 1–5 and Sections 1–3.
Rights and permissions
About this article
Cite this article
Christoff-Tempesta, T., Cho, Y., Kim, DY. et al. Self-assembly of aramid amphiphiles into ultra-stable nanoribbons and aligned nanoribbon threads. Nat. Nanotechnol. 16, 447–454 (2021). https://doi.org/10.1038/s41565-020-00840-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-020-00840-w
This article is cited by
-
The reformation of catalyst: From a trial-and-error synthesis to rational design
Nano Research (2024)
-
Supramolecular framework membrane for precise sieving of small molecules, nanoparticles and proteins
Nature Communications (2023)
-
Focus on using nanopore technology for societal health, environmental, and energy challenges
Nano Research (2022)
-
Domain-selective thermal decomposition within supramolecular nanoribbons
Nature Communications (2021)