Abstract
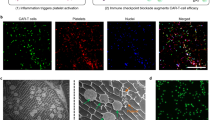
Cancer recurrence after surgical resection remains a significant cause of treatment failure. Here, we have developed an in situ formed immunotherapeutic bioresponsive gel that controls both local tumour recurrence after surgery and development of distant tumours. Briefly, calcium carbonate nanoparticles pre-loaded with the anti-CD47 antibody are encapsulated in the fibrin gel and scavenge H+ in the surgical wound, allowing polarization of tumour-associated macrophages to the M1-like phenotype. The released anti-CD47 antibody blocks the ‘don’t eat me’ signal in cancer cells, thereby increasing phagocytosis of cancer cells by macrophages. Macrophages can promote effective antigen presentation and initiate T cell mediated immune responses that control tumour growth. Our findings indicate that the immunotherapeutic fibrin gel ‘awakens’ the host innate and adaptive immune systems to inhibit both local tumour recurrence post surgery and potential metastatic spread.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the plots within this paper and other findings of this study are available from the corresponding author upon reasonable request.
References
Turajlic, S. & Swanton, C. Metastasis as an evolutionary process. Science 352, 169–175 (2016).
Wang, C. et al. In situ activation of platelets with checkpoint inhibitors for post-surgical cancer immunotherapy. Nat. Biomed. Eng. 1, 0011 (2017).
Wang, C. et al. In situ formed reactive oxygen species-responsive scaffold with gemcitabine and checkpoint inhibitor for combination therapy. Sci. Transl. Med. 10, eaan3682 (2018).
Tohme, S. et al. Neutrophil extracellular traps promote the development and progression of liver metastases after surgical stress. Cancer Res. 76, 1367–1380 (2016).
Baker, D., Masterson, T., Pace, R., Constable, W. & Wanebo, H. The influence of the surgical wound on local tumor recurrence. Surgery 106, 525–532 (1989).
Grivennikov, S. I., Greten, F. R. & Karin, M. Immunity, inflammation and cancer. Cell. 140, 883–899 (2010).
Vakkila, J. & Lotze, M. T. Inflammation and necrosis promote tumour growth. Nat. Rev. Immunol. 4, 641–648 (2004).
Albain, K. S. et al. Radiotherapy plus chemotherapy with or without surgical resection for stage III non-small-cell lung cancer: a phase III randomised controlled trial. The Lancet 374, 379–386 (2009).
Kwon, E. D. et al. Elimination of residual metastatic prostate cancer after surgery and adjunctive cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) blockade immunotherapy. Proc. Natl Acad. Sci. USA 96, 15074–15079 (1999).
Stephan, S. B. et al. Biopolymer implants enhance the efficacy of adoptive T-cell therapy. Nat. Biotechnol. 33, 97–101 (2015).
Pardoll, D. M. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 12, 252–264 (2012).
Wang, C., Ye, Y., Hu, Q., Bellotti, A. & Gu, Z. Tailoring biomaterials for cancer immunotherapy: emerging trends and future outlook. Adv. Mater. 29, 1606036 (2017).
Chen, Q. et al. Photothermal therapy with immune-adjuvant nanoparticles together with checkpoint blockade for effective cancer immunotherapy. Nat. Commun. 7, 13193 (2016).
Gordon, S. Alternative activation of macrophages. Nat. Rev. Immunol. 3, 23–35 (2003).
Calandra, T. & Roger, T. Macrophage migration inhibitory factor: a regulator of innate immunity. Nat. Rev. Immunol. 3, 791–800 (2003).
Subramanian, S., Parthasarathy, R., Sen, S., Boder, E. T. & Discher, D. E. Species- and cell type-specific interactions between CD47 and human SIRPα. Blood 107, 2548–2556 (2006).
Edris, B. et al. Antibody therapy targeting the CD47 protein is effective in a model of aggressive metastatic leiomyosarcoma. Proc. Natl Acad. Sci. USA 109, 6656–6661 (2012).
Michaels, A. D. et al. CD47 blockade as an adjuvant immunotherapy for resectable pancreatic cancer. Clin. Cancer Res. 24, 1415–1425 (2018).
Ring, N. G. et al. Anti-SIRPα antibody immunotherapy enhances neutrophil and macrophage antitumor activity. Proc. Natl Acad. Sci. USA 114, 10578–10585 (2017).
Jaiswal, S. et al. CD47 is upregulated on circulating hematopoietic stem cells and leukemia cells to avoid phagocytosis. Cell 138, 271–285 (2009).
Kershaw, M. H. & Smyth, M. J. Making macrophages eat cancer. Science 341, 41–42 (2013).
Sockolosky, J. T. et al. Durable antitumor responses to CD47 blockade require adaptive immune stimulation. Proc. Natl Acad. Sci. USA 113, 2646–2654 (2016).
Huang, Y., Ma, Y., Gao, P. & Yao, Z. Targeting CD47: the achievements and concerns of current studies on cancer immunotherapy. J. Thorac. Dis. 9, E168–E174 (2017).
Herberman, R. R., Ortaldo, J. R. & Bonnard, G. D. Augmentation by interferon of human natural and antibody-dependent cell-mediated cytotoxicity. Nature 277, 221–223 (1979).
Lin, E. Y. & Pollard, J. W. Tumor-associated macrophages press the angiogenic switch in breast cancer. Cancer Res. 67, 5064–5066 (2007).
Mantovani, A., Sozzani, S., Locati, M., Allavena, P. & Sica, A. Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 23, 549–555 (2002).
Neubert, N. J. et al. T cell–induced CSF1 promotes melanoma resistance to PD1 blockade. Sci. Transl. Med. 10, eaan3311 (2018).
Lawrence, T. & Natoli, G. Transcriptional regulation of macrophage polarization: enabling diversity with identity. Nat. Rev. Immunol. 11, 750–761 (2011).
Wang, Y.-C. et al. Notch signaling determines the M1 versus M2 polarization of macrophages in antitumor immune responses. Cancer Res. 70, 4840–4849 (2010).
Chanmee, T., Ontong, P., Konno, K. & Itano, N. Tumor-associated macrophages as major players in the tumor microenvironment. Cancers 6, 1670–1690 (2014).
Baer, C. et al. Suppression of microRNA activity amplifies IFN-γ-induced macrophage activation and promotes anti-tumour immunity. Nat. Cell Biol. 18, 790–802 (2016).
Lewis, C. E., Harney, A. S. & Pollard, J. W. The multifaceted role of perivascular macrophages in tumors. Cancer Cell. 30, 18–25 (2016).
Pyonteck, S. M. et al. CSF-1R inhibition alters macrophage polarization and blocks glioma progression. Nat. Med. 19, 1264–1272 (2013).
Martin, P. Wound healing—aiming for perfect skin regeneration. Science 276, 75–81 (1997).
Zhao, Y. et al. A preloaded amorphous calcium carbonate/doxorubicin@silica nanoreactor for ph‐responsive delivery of an anticancer drug. Angew. Chem. Int. Ed. 54, 919–922 (2015).
Lu, Y., Aimetti, A. A., Langer, R. & Gu, Z. Bioresponsive materials. Nat. Rev. Mater. 2, 16075 (2017).
Mi, P. et al. A pH-activatable nanoparticle with signal-amplification capabilities for non-invasive imaging of tumour malignancy. Nat. Nanotech. 11, 724–730 (2016).
Credo, R., Curtis, C. & Lorand, L. Ca2+-related regulatory function of fibrinogen. Proc. Natl Acad. Sci. USA 75, 4234–4237 (1978).
Chen, Q. et al. A self-assembled albumin-based nanoprobe for in vivo ratiometric photoacoustic pH imaging. Adv. Mater. 27, 6820–6827 (2015).
Colegio, O. R. et al. Functional polarization of tumour-associated macrophages by tumour-derived lactic acid. Nature 513, 559–563 (2014).
Liu, Q. et al. Nanoparticle-mediated trapping of wnt family member 5A in tumor microenvironments enhances immunotherapy for B-Raf proto-oncogene mutant melanoma. ACS Nano 12, 1250–1261 (2018).
Rodell, C. B. et al. TLR7/8-agonist-loaded nanoparticles promote the polarization of tumour-associated macrophages to enhance cancer immunotherapy. Nat. Biomed. Eng. 2, 578 (2018).
Meyer, M. A. et al. Breast and pancreatic cancer interrupt IRF8-dependent dendritic cell development to overcome immune surveillance. Nat. Commun. 9, 1250 (2018).
Velu, V. et al. Enhancing SIV-specific immunity in vivo by PD-1 blockade. Nature 458, 206–210 (2009).
Tumeh, P. C. et al. PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 515, 568–571 (2014).
Zou, W., Wolchok, J. D. & Chen, L. PD-L1 (B7-H1) and PD-1 pathway blockade for cancer therapy: mechanisms, response biomarkers and combinations. Sci. Transl. Med. 8, 328rv324 (2016).
Ribas, A. & Wolchok, J. D. Cancer immunotherapy using checkpoint blockade. Science 359, 1350–1355 (2018).
Nishiyama, N. et al. Novel cisplatin-incorporated polymeric micelles can eradicate solid tumors in mice. Cancer Res. 63, 8977–8983 (2003).
Chen, Q. et al. An albumin-based theranostic nano-agent for dual-modal imaging guided photothermal therapy to inhibit lymphatic metastasis of cancer post surgery. Biomaterials 35, 9355–9362 (2014).
Lee, E. J. et al. Nanocage‐therapeutics prevailing phagocytosis and immunogenic cell death awakens immunity against cancer. Adv. Mater. 30, 1705581 (2018).
Acknowledgements
This work was supported by grants from start-up packages from UNC/NC state and UCLA, the Jonsson Comprehensive Cancer Center at UCLA, the Alfred P. Sloan Foundation (Sloan Research Fellowship), the National Key R&D Program of China (2017YFA0205600), the Program for Guangdong Introducing Innovative and Enterpreneurial Teams (2017ZT07S054) and the National Natural Science Foundation of China (51728301). The authors thank L. Huang at UNC at Chapel Hill for providing the B16F10-Luc-GFP.
Author information
Authors and Affiliations
Contributions
Q.C., G.D. and Z.G. conceived and designed the experiments. Q.C., C.W., X.Z., G.C., Q.H., Ji.W., D.W., Y.Z., H.L., Y.L., G.Y. and X.Z. performed the experiments and analysed data. Q.C., G.C., C.J., Ju.W., G.D. and Z.G. co-wrote the paper. All authors discussed the results and implications and edited the manuscript at all stages.
Corresponding author
Ethics declarations
Competing interests
Z.G. and Q.C. have applied for patents related to this study. Z.G. is a scientific co-founder of ZenCapsule Inc.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Chen, Q., Wang, C., Zhang, X. et al. In situ sprayed bioresponsive immunotherapeutic gel for post-surgical cancer treatment. Nature Nanotech 14, 89–97 (2019). https://doi.org/10.1038/s41565-018-0319-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-018-0319-4
This article is cited by
-
Hypoxia-induced ZEB1 promotes cervical cancer immune evasion by strengthening the CD47-SIRPα axis
Cell Communication and Signaling (2024)
-
In-situ-sprayed therapeutic hydrogel for oxygen-actuated Janus regulation of postsurgical tumor recurrence/metastasis and wound healing
Nature Communications (2024)
-
Macrophage metabolism, phenotype, function, and therapy in hepatocellular carcinoma (HCC)
Journal of Translational Medicine (2023)
-
Nanomaterials in tumor immunotherapy: new strategies and challenges
Molecular Cancer (2023)
-
Phase-transition nanodroplets with immunomodulatory capabilities for potentiating mild magnetic hyperthermia to inhibit tumour proliferation and metastasis
Journal of Nanobiotechnology (2023)