Abstract
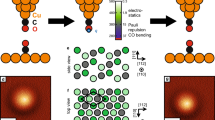
Atomic force microscopy is an impressive tool with which to directly resolve the bonding structure of organic compounds1,2,3,4,5. The methodology usually involves chemical passivation of the probe-tip termination by attaching single molecules or atoms such as CO or Xe (refs 1,6,7,8,9). However, these probe particles are only weakly connected to the metallic apex, which results in considerable dynamic deflection. This probe particle deflection leads to pronounced image distortions, systematic overestimation of bond lengths, and in some cases even spurious bond-like contrast features, thus inhibiting reliable data interpretation8–12. Recently, an alternative approach to tip passivation has been used in which slightly indenting a tip into oxidized copper substrates and subsequent contrast analysis allows for the verification of an oxygen-terminated Cu tip13,14,15. Here we show that, due to the covalently bound configuration of the terminal oxygen atom, this copper oxide tip (CuOx tip) has a high structural stability, allowing not only a quantitative determination of individual bond lengths and access to bond order effects, but also reliable intermolecular bond characterization. In particular, by removing the previous limitations of flexible probe particles, we are able to provide conclusive experimental evidence for an unusual intermolecular N–Au–N three-centre bond. Furthermore, we demonstrate that CuOx tips allow the characterization of the strength and configuration of individual hydrogen bonds within a molecular assembly.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gross, L., Mohn, F., Moll, N., Liljeroth, P. & Meyer, G. The chemical structure of a molecule resolved by atomic force microscopy. Science 325, 1110–1114 (2009).
Mohn, F., Gross, L., Moll, N. & Meyer, G. Imaging the charge distribution within a single molecule. Nat. Nanotech. 7, 227–231 (2012).
de Oteyza, D. G. et al. Direct imaging of covalent bond structure in single-molecule chemical reactions. Science 340, 1434–1437 (2013).
Zhang, J. et al. Real-space identification of intermolecular bonding with atomic force microscopy. Science 342, 611–614 (2013).
Pavliček, N. et al. Synthesis and characterization of triangulene. Nat. Nanotech. 12, 308–311 (2017).
Bartels, L. et al. Dynamics of electron-induced manipulation of individual CO molecules on Cu(110). Phys. Rev. Lett. 80, 2004–2007 (1998).
Mohn, F., Schuler, B., Gross, L. & Meyer, G. Different tips for high-resolution atomic force microscopy and scanning tunneling microcopy of single molecules. Appl. Phys. Lett. 102, 073109 (2013).
Gross, L. et al. Bond-order discrimination by atomic force microscopy. Science 337, 1326–1329 (2012).
Weymouth, A. J., Hofmann, T. & Giessibl, F. J. Quantifying molecular stiffness and interaction with lateral force microscopy. Science 343, 1120–1122 (2014).
Pavliček, N. et al. Atomic force microscopy reveals bistable configurations of dibenzo[a,h]thianthrene and their interconversion pathway. Phys. Rev. Lett. 108, 086101 (2012).
Hapala, P. et al. Mechanism of high-resolution STM/AFM imaging with functionalized tips. Phys. Rev. B 90, 085421 (2014).
Hämäläinen, S. K. et al. Intermolecular contrast in atomic force microscopy images without intermolecular bonds. Phys. Rev. Lett. 113, 186102 (2014).
Bamidele, J. et al. Chemical tip fingerprinting in scanning probe microscopy of an oxidized Cu(110) surface. Phys. Rev. B 86, 155422 (2012).
Mönig, H. et al. Understanding scanning tunneling microscopy contrast mechanisms on metal oxides: a case study. ACS Nano 7, 10233–10244 (2013).
Mönig, H. et al. Submolecular imaging by noncontact atomic force microscopy with an oxygen atom rigidly connected to a metallic probe. ACS Nano 10, 1201–1209 (2016).
Liu, S., Lu, Y.-J., Kappes, M. M. & Ibers, J. A. The structure of the C60 molecule: X-ray crystal structure determination of a twin at 110 K. Science 254, 408–410 (1991).
Sweetman, A. M. et al. Mapping the force field of a hydrogen-bonded assembly. Nat. Commun. 5, 3931 (2014).
Wang, C.-G., Cheng, Z.-H., Qiu, X.-H. & Ji, W. Unusually high electron density in an intermolecular non-bonding region: role of metal substrate. Chin. Chem. Lett. 28, 759–764 (2017).
Van der Lit, J. et al. Modeling the self-assembly of organic molecules in 2D molecular layers with different structures. J. Phys. Chem. C 120, 318–323 (2016).
Van der Lit, J., Di Cicco, F., Hapala, P., Jelinek, P. & Swart, I. Submolecular resolution imaging of molecules by atomic force microscopy: the influence of the electrostatic force. Phys. Rev. Lett. 116, 096102 (2016).
Weiss, C., Wagner, C., Temirov, R. & Tautz, F. S. Direct imaging of intermolecular bonds in scanning tunneling microscopy. J. Am. Chem. Soc. 132, 11864–11865 (2010).
Han, Z. et al. Imaging the halogen bond in self-assembled halogenbenzenes on silver. Science 358, 206–210 (2017).
Rohlfing, M., Temirov, R. & Tautz, F. S. Adsorption structure and scanning tunneling data of a prototype organic-inorganic interface: PTCDA on Ag(111). Phys. Rev. B 76, 115421 (2007).
Desiraju, G. R. A bond by any other name. Angew. Chem. Int. Ed. 50, 52–60 (2011).
Kelly, R. E. A., Lee, Y. J. & Kantorovich, L. N. Homopairing possibilities of the DNA base adenine. J. Phys. Chem. B 109, 11933–11939 (2005).
Schneiderbauer, M., Emmrich, M., Weymouth, A. J. & Giessibl, F. J. CO tip functionalization inverts atomic force microscopy contrast via short-range electrostatic forces. Phys. Rev. Lett. 112, 166102 (2014).
Ellner, M. et al. The electric field of CO tips and its relevance for atomic force microscopy. Nano Lett. 16, 1974–1980 (2016).
Giessibl, F. J. High-speed force sensor for force microscopy and profilometry utilizing a quartz tuning fork. Appl. Phys. Lett. 73, 3956–3958 (1998).
SPIP V5.1.11 Scanning probe image processor (Hørsholm: Image Metrology); http://www.imagemet.com.
Kern, K. et al. Long-range spatial self-organization in the adsorbate-induced restructuring of surfaces: Cu(100)-(2×1)O. Phys. Rev. Lett. 67, 855–858 (1991).
Stephens, P. J., Devlin, F. J., Chabalowski, C. F. & Frisch, M. J. Ab initio calculation of vibrational absorption and circular dichroism spectra using density functional force fields. J. Phys. Chem. 98, 11623–11627 (1994).
Turbomole V6.3 (University of Karlsruhe and Forschungszentrum Karlsruhe, Turbomole, 2009); http://www.turbomole.com.
Weigend, F. & Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: design and assessment of accuracy. Phys. Chem. Chem. Phys. 7, 3297–3305 (2005).
Grimme, S. J. Semiempirical GGA-type density functional constructed with a long-range dispersion correction. Comput. Chem. 27, 1787–1799 (2006).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Dion, M., Rydberg, H., Schröder, E., Langreth, D. C. & Lundqvist, B. I. Van der Waals density functional for general geometries. Phys. Rev. Lett. 92, 246401 (2004).
Klimeš, J., Bowler, D. R. & Michaelides, A. Van der Waals density functionals applied to solids. Phys. Rev. B 83, 195131 (2011).
Klimeš, J., Bowler, D. & Michaelides, A. Chemical accuracy for the van der Waals density functional. J. Phys. Condens. Matter 22, 022201 (2010).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Monkhorst, H. J. & Pack, J. D. Special points for Brillouin-zone integrations. Phys. Rev. B 13, 5188–5192 (1976).
Soler, J. M. et al. The SIESTA method for ab initio order-N materials simulation. J. Phys. Condens. Matter 14, 2745 (2002).
Acknowledgements
This work was supported by the Deutsche Forschungsgemeinschaft through collaborative research centres TRR 61 and SFB 858, and through projects MO 2345/4-1, AM 460/2-1, and FU 299/19. W.J. thanks the National Science Foundation of China for support through grant no. 11622437. D. Yesilpinar is thanked for technical support.
Author information
Authors and Affiliations
Contributions
H.M. conceived the experiments. H.M., O.D.A., A.T. and L.L. performed the experiments. H.M., S.A. and O.D.A. analysed the data. S.A., Z.H., W.J. and M.R. performed the DFT simulations. M.C. and C.A.S. synthesized the DBTH molecules. S.A. and H.F. contributed significantly to the scientific discussion of the project. All authors discussed the results and commented on the manuscript, which was drafted by H.M.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–10
Rights and permissions
About this article
Cite this article
Mönig, H., Amirjalayer, S., Timmer, A. et al. Quantitative assessment of intermolecular interactions by atomic force microscopy imaging using copper oxide tips. Nature Nanotech 13, 371–375 (2018). https://doi.org/10.1038/s41565-018-0104-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-018-0104-4
This article is cited by
-
On-surface synthesis of ballbot-type N-heterocyclic carbene polymers
Nature Chemistry (2023)
-
Discovery and construction of surface kagome electronic states induced by p-d electronic hybridization in Co3Sn2S2
Nature Communications (2023)
-
Single hydrogen atom manipulation for reversible deprotonation of water on a rutile TiO2 (110) surface
Communications Chemistry (2021)
-
Polymerization of silanes through dehydrogenative Si–Si bond formation on metal surfaces
Nature Chemistry (2021)
-
Constructing covalent organic nanoarchitectures molecule by molecule via scanning probe manipulation
Nature Chemistry (2021)