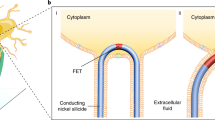
Abstract
Optical methods for modulating cellular behaviour are promising for both fundamental and clinical applications. However, most available methods are either mechanically invasive, require genetic manipulation of target cells or cannot provide subcellular specificity. Here, we address all these issues by showing optical neuromodulation with free-standing coaxial p-type/intrinsic/n-type silicon nanowires. We reveal the presence of atomic gold on the nanowire surfaces, likely due to gold diffusion during the material growth. To evaluate how surface gold impacts the photoelectrochemical properties of single nanowires, we used modified quartz pipettes from a patch clamp and recorded sustained cathodic photocurrents from single nanowires. We show that these currents can elicit action potentials in primary rat dorsal root ganglion neurons through a primarily atomic gold-enhanced photoelectrochemical process.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Famm, K., Litt, B., Tracey, K. J., Boyden, E. S. & Slaoui, M. A jump-start for electroceuticals. Nature 496, 159–161 (2013).
Vetter, R. J., Williams, J. C., Hetke, J. F., Nunamaker, E. A. & Kipke, D. R. Chronic neural recording using silicon-substrate microelectrode arrays implanted in cerebral cortex. IEEE Trans. Biomed. Eng. 51, 896–904 (2004).
Zhou, W., Dai, X. C. & Lieber, C. M. Advances in nanowire bioelectronics. Rep. Prog. Phys. 80, 016701 (2017).
Ghezzi, D. et al. A hybrid bioorganic interface for neuronal photoactivation. Nat. Commun. 2, 166 (2011).
Ghezzi, D. et al. A polymer optoelectronic interface restores light sensitivity in blind rat retinas. Nat. Photon. 7, 400–406 (2013).
Mathieson, K. et al. Photovoltaic retinal prosthesis with high pixel density. Nat. Photon. 6, 391–397 (2012).
Carvalho-de-Souza, J. L. et al. Photosensitivity of neurons enabled by cell-targeted gold nanoparticles. Neuron 86, 207–217 (2015).
Jiang, Y. W. et al. Heterogeneous silicon rnesostructures for lipid-supported bioelectric interfaces. Nat. Mater. 15, 1023–1030 (2016).
Packer, A. M., Roska, B. & Hausser, M. Targeting neurons and photons for optogenetics. Nat. Neurosci. 16, 805–815 (2013).
Boyden, E. S., Zhang, F., Bamberg, E., Nagel, G. & Deisseroth, K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat. Neurosci. 8, 1263–1268 (2005).
Zimmerman, J., Parameswaran, R. & Tian, B. Z. Nanoscale semiconductor devices as new biomaterials. Biomater. Sci. 2, 619–626 (2014).
Rogers, J. A., Someya, T. & Huang, Y. G. Materials and mechanics for stretchable electronics. Science 327, 1603–1607 (2010).
Hwang, S. W. et al. A physically transient form of silicon electronics. Science 337, 1640–1644 (2012).
Straub, B., Meyer, E. & Fromherz, P. Recombinant maxi-K channels on transistor, a prototype of iono-electronic interfacing. Nat. Biotechnol. 19, 121–124 (2001).
Chiappini, C. et al. Biodegradable silicon nanoneedles delivering nucleic acids intracellularly induce localized in vivo neovascularization. Nat. Mater. 14, 532–539 (2015).
Priolo, F., Gregorkiewicz, T., Galli, M. & Krauss, T. F. Silicon nanostructures for photonics and photovoltaics. Nat. Nanotech. 9, 19–32 (2014).
Xie, P., Xiong, Q. H., Fang, Y., Qing, Q. & Lieber, C. M. Local electrical potential detection of DNA by nanowire-nanopore sensors. Nat. Nanotech. 7, 119–125 (2012).
Yan, R. et al. Nanowire-based single-cell endoscopy. Nat. Nanotech. 7, 191–196 (2012).
Zimmerman, J. F. et al. Cellular uptake and dynamics of unlabeled freestanding silicon nanowires. Sci. Adv. 2, e1601039 (2016).
Zhang, A. Q. & Lieber, C. M. Nano-bioelectronics. Chem. Rev. 116, 215–257 (2016).
Tian, B. Z. et al. Coaxial silicon nanowires as solar cells and nanoelectronic power sources. Nature 449, 885–888 (2007).
Su, Y. D. et al. Single-nanowire photoelectrochemistry. Nat. Nanotech. 11, 609–612 (2016).
Hannon, J. B., Kodambaka, S., Ross, F. M. & Tromp, R. M. The influence of the surface migration of gold on the growth of silicon nanowires. Nature 440, 69–71 (2006).
Luo, Z. Q. et al. Atomic gold-enabled three-dimensional lithography for silicon mesostructures. Science 348, 1451–1455 (2015).
Seibt, M. et al. Gettering in silicon photovoltaics: current state and future perspectives. Phys. Stat. Solidi A Appl. Mater. Sci. 203, 696–713 (2006).
Yuan, G. B. et al. Understanding the origin of the low performance of chemically grown silicon nanowires for solar energy conversion. Angew. Chem. Int. Ed. 50, 2334–2338 (2011).
Kim, S. K. et al. Tuning light absorption in core/shell silicon nanowire photovoltaic devices through morphological design. Nano Lett. 12, 4971–4976 (2012).
Garnett, E. C. et al. Dopant profiling and surface analysis of silicon nanowires using capacitance–voltage measurements. Nat. Nanotech. 4, 311–314 (2009).
Liu, Z. et al. Anomalous high capacitance in a coaxial single nanowire capacitor. Nat. Commun. 3, 879 (2012).
Vogel, E. M. Technology and metrology of new electronic materials and devices. Nat. Nanotech. 2, 25–32 (2007).
Mogyoros, I., Kiernan, M. C., Burke, D. & Bostock, H. Strength-duration properties of sensory and motor axons in amyotrophic lateral sclerosis. Brain 121, 851–859 (1998).
Walter, M. G. et al. Solar water splitting cells. Chem. Rev. 110, 6446–6473 (2010).
Warren, E. L., McKone, J. R., Atwater, H. A., Graya, H. B. & Lewis, N. S. Hydrogen-evolution characteristics of Ni-Mo-coated, radial junction, n+p-silicon microwire array photocathodes. Energy Environ. Sci. 5, 9653–9661 (2012).
Sachsenhauser, M., Sharp, I. D., Stutzmann, M. & Garrido, J. A. Surface state mediated electron transfer across the n-type SiC/electrolyte interface. J. Phys. Chem. C 120, 6524–6533 (2016).
Lee, J. H., Zhang, A. Q., You, S. S. & Lieber, C. M. Spontaneous internalization of cell penetrating peptide-modified nanowires into primary neurons. Nano Lett. 16, 1509–1513 (2016).
Liu, C., Colon, B. C., Ziesack, M., Silver, P. A. & Nocera, D. G. Water splitting–biosynthetic system with CO2 reduction efficiencies exceeding photosynthesis. Science 352, 1210–1213 (2016).
Sakimoto, K. K., Wong, A. B. & Yang, P. D. Self-photosensitization of nonphotosynthetic bacteria for solar-to-chemical production. Science 351, 74–77 (2016).
Silva, G. A. Neuroscience nanotechnology: progress, opportunities and challenges. Nat. Rev. Neurosci. 7, 65–74 (2006).
Acknowledgements
We thank F. Shi at the University of Illinois Chicago for her help in collecting the EDS data. This work is supported by the Air Force Office of Scientific Research (AFOSR FA9550-14-1-0175, FA9550-15-1-0285), the National Science Foundation (NSF CAREER, DMR-1254637; NSF MRSEC, DMR 1420709), the Alfred P. Sloan Foundation Fellowship (FG-2016-6805), the Searle Scholars Foundation, the National Institute of Health (NIH GM030376, NIH F30AI138156, and NS101488), MSTP Training Grant (T32GM007281) and the Paul and Daisy Soros Foundation.
Author information
Authors and Affiliations
Contributions
R.P. grew SiNWs for all experiments and performed all neuron electrophysiology experiments. R.P. and M.J.B. analysed neuron electrophysiology data. Y.J. performed and analysed STEM data. Y.J., J.Y. and R.P. prepared samples for, performed and analysed APT experiments. K.K. performed and analysed XPS experiments. R.P. and A.P. prepared samples for and performed SEM on neuron/SiNW samples. R.P. and M.J.B. performed and analysed LIVE/DEAD and fluorescence microscopy experiments. J.L.C.-d.-S. and R.P. set up equipment for all neuron electrophysiology experiments, temperature recordings and photocurrent recordings. R.P., J.F.Z. and J.L.C.-d.-S. developed the single NW photocurrent recording method. R.P. performed and analysed all photocurrent and temperature recordings. E.J.A. provided support and input on all experiments. B.T. and F.B. directed the research. R.P. and B.T. co-wrote the paper. All authors read and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Table 1, Supplementary Figures 1–11.
Life Sciences Reporting Summary
Life Sciences Reporting Summary
Rights and permissions
About this article
Cite this article
Parameswaran, R., Carvalho-de-Souza, J.L., Jiang, Y. et al. Photoelectrochemical modulation of neuronal activity with free-standing coaxial silicon nanowires. Nature Nanotech 13, 260–266 (2018). https://doi.org/10.1038/s41565-017-0041-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-017-0041-7
This article is cited by
-
Janus microparticles-based targeted and spatially-controlled piezoelectric neural stimulation via low-intensity focused ultrasound
Nature Communications (2024)
-
Non-Faradaic optoelectrodes for safe electrical neuromodulation
Nature Communications (2024)
-
Monolithic silicon for high spatiotemporal translational photostimulation
Nature (2024)
-
Tetherless Optical Neuromodulation: Wavelength from Orange-red to Mid-infrared
Neuroscience Bulletin (2024)
-
Plasmonics for neuroengineering
Communications Materials (2023)