Abstract
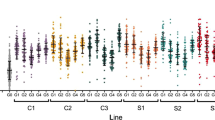
The dengue, Zika and chikungunya viruses are transmitted by the mosquito Aedes aegypti and pose a substantial threat to global public health. Current vaccines and mosquito control strategies have limited efficacy, so novel interventions are needed1,2. Wolbachia are bacteria that inhabit insect cells and have been found to reduce viral infection—a phenotype that is referred to as viral ‘blocking’3. Although not naturally found in A. aegypti4, Wolbachia were stably introduced into this mosquito in 20114,5 and were shown to reduce the transmission potential of dengue, Zika and chikungunya6,7. Subsequent field trials showed Wolbachia’s ability to spread through A. aegypti populations and reduce the local incidence of dengue fever8. Despite these successes, the evolutionary stability of viral blocking is unknown. Here, we utilized artificial selection to reveal genetic variation in the mosquito that affects Wolbachia-mediated dengue blocking. We found that mosquitoes exhibiting weaker blocking also have reduced fitness, suggesting the potential for natural selection to maintain blocking. We also identified A. aegypti genes that affect blocking strength, shedding light on a possible mechanism for the trait. These results will inform the use of Wolbachia as biocontrol agents against mosquito-borne viruses and direct further research into measuring and improving their efficacy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Raw data are deposited in the Dryad online repository (https://doi.org/10.5061/dryad.6vv10h0) and sequence data are available via the European Nucleotide Archive (accession number: PRJEB33044).
References
Moyes, C. L. et al. Contemporary status of insecticide resistance in the major Aedes vectors of arboviruses infecting humans. PLoS Negl. Trop. Dis. 11, e0005625 (2017).
Global Strategy for Dengue Prevention and Control, 2012–2020 (World Health Organization, 2012).
Teixeira, L., Ferreira, A. & Ashburner, M. The bacterial symbiont Wolbachia induces resistance to RNA viral infections in Drosophila melanogaster. PLoS Biol. 6, e2 (2008).
Gloria-Soria, A., Chiodo, T. G. & Powell, J. R. Lack of evidence for natural Wolbachia infections in Aedes aegypti (Diptera: Culicidae). J. Med. Entomol. 55, 1354–1356 (2018).
Walker, T. et al. The wMel Wolbachia strain blocks dengue and invades caged Aedes aegypti populations. Nature 476, 450–453 (2011).
Moreira, L. A. et al. A Wolbachia symbiont in Aedes aegypti limits infection with dengue, chikungunya, and Plasmodium. Cell 139, 1268–1278 (2009).
Aliota, M. T., Peinado, S. A., Velez, I. D. & Osorio, J. E. The wMel strain of Wolbachia reduces transmission of Zika virus by Aedes aegypti. Sci. Rep. 6, 28792 (2016).
O’Neill, S. L. et al. Scaled deployment of Wolbachia to protect the community from Aedes transmitted arboviruses. Gates Open Res. 2, 36 (2018).
Terradas, G., Allen, S. L., Chenoweth, S. F. & McGraw, E. A. Family level variation in Wolbachia-mediated dengue virus blocking in Aedes aegypti. Parasit. Vectors 10, 622 (2017).
Longdon, B. et al. The causes and consequences of changes in virulence following pathogen host shifts. PLoS Pathog. 11, e1004728 (2015).
Bull, J. J. & Turelli, M. Wolbachia versus dengue: evolutionary forecasts. Evol. Med. Public Health 2013, 197–207 (2013).
Ant, T. H., Herd, C. S., Geoghegan, V., Hoffmann, A. A. & Sinkins, S. P. The Wolbachia strain wAu provides highly efficient virus transmission blocking in Aedes aegypti. PLoS Pathog. 14, e1006815 (2018).
Hoffmann, A. A., Ross, P. A. & Rašić, G. Wolbachia strains for disease control: ecological and evolutionary considerations. Evol. Appl. 8, 751–768 (2015).
Pan, X. et al. The bacterium Wolbachia exploits host innate immunity to establish a symbiotic relationship with the dengue vector mosquito Aedes aegypti. ISME J. 12, 277–288 (2018).
Caragata, E. P. et al. Dietary cholesterol modulates pathogen blocking by Wolbachia. PLoS Pathog. 9, e1003459 (2013).
Bhattacharya, T., Newton, I. L. G. & Hardy, R. W. Wolbachia elevates host methyltransferase expression to block an RNA virus early during infection. PLoS Pathog. 13, e1006427 (2017).
Pan, X. et al. Wolbachia induces reactive oxygen species (ROS)-dependent activation of the Toll pathway to control dengue virus in the mosquito Aedes aegypti. Proc. Natl Acad. Sci. USA 109, E23–E31 (2012).
Terradas, G., Joubert, D. A. & McGraw, E. A. The RNAi pathway plays a small part in Wolbachia-mediated blocking of dengue virus in mosquito cells. Sci. Rep. 7, 43847 (2017).
Amuzu, H. E. & McGraw, E. A. Wolbachia-based dengue virus inhibition is not tissue-specific in Aedes aegypti. PLoS Negl. Trop. Dis. 10, e0005145 (2016).
Frankham, R. Are responses to artificial selection for reproductive fitness characters consistently asymmetrical? Genet. Res. Camb. 56, 35–42 (1990).
Ohm, J. R. et al. Fitness consequences of altered feeding behavior in immune-challenged mosquitoes. Parasit. Vectors 9, 113 (2016).
Rueda, L. M., Patel, K. J., Axtell, R. C. & Stinner, R. E. Temperature-dependent development and survival rates of Culex quinquefasciatus and Aedes aegypti (Diptera: Culicidae). J. Med. Entomol. 27, 892–898 (1990).
Halbleib, J. M. & Nelson, W. J. Cadherins in development: cell adhesion, sorting, and tissue morphogenesis. Genes Dev. 20, 3199–3214 (2006).
Colpitts, T. M. et al. Use of a tandem affinity purification assay to detect interactions between West Nile and dengue viral proteins and proteins of the mosquito vector. Virology 417, 179–187 (2011).
Lindsey, A. R. I., Bhattacharya, T., Newton, I. L. G. & Hardy, R. W. Conflict in the intracellular lives of endosymbionts and viruses: a mechanistic look at Wolbachia-mediated pathogen-blocking. Viruses 10, 141 (2018).
Sheehan, K. B., Martin, M., Lesser, C. F., Isberg, R. R. & Newton, I. L. Identification and characterization of a candidate Wolbachia pipientis type IV effector that interacts with the actin cytoskeleton. mBio 7, e00622-16 (2016).
Hughes, G. L. et al. Wolbachia infections in Anopheles gambiae cells: transcriptomic characterization of a novel host–symbiont interaction. PLoS Pathog. 7, e1001296 (2011).
Frentiu, F. D. et al. Limited dengue virus replication in field-collected Aedes aegypti mosquitoes infected with Wolbachia. PLoS Negl. Trop. Dis. 8, e2688 (2014).
Ferguson, N. M. et al. Modeling the impact on virus transmission of Wolbachia-mediated blocking of dengue virus infection of Aedes aegypti. Sci. Transl. Med. 7, 279ra237 (2015).
Hoffmann, A. A. et al. Successful establishment of Wolbachia in Aedes populations to suppress dengue transmission. Nature 476, 454–457 (2011).
Ye, Y. H. et al. Evolutionary potential of the extrinsic incubation period of dengue virus in Aedes aegypti. Evolution 70, 2459–2469 (2016).
Ritchie, S. A. et al. An explosive epidemic of DENV-3 in Cairns, Australia. PLoS ONE 8, e68137 (2013).
Ye, Y. H. et al. Comparative susceptibility of mosquito populations in North Queensland, Australia to oral infection with dengue virus. Am. J. Trop. Med. Hyg. 90, 422–430 (2014).
Jupatanakul, N., Sim, S. & Dimopoulos, G. The insect microbiome modulates vector competence for arboviruses. Viruses 6, 4294–4313 (2014).
Kawecki, T. J. et al. Experimental evolution. Trends Ecol. Evol. 27, 547–560 (2012).
Zheng, M. L., Zhang, D. J., Damiens, D. D., Lees, R. S. & Gilles, J. R. Standard operating procedures for standardized mass rearing of the dengue and chikungunya vectors Aedes aegypti and Aedes albopictus (Diptera: Culicidae)—II—egg storage and hatching. Parasit. Vectors 8, 348 (2015).
Joshi, V., Mourya, D. T. & Sharma, R. C. Persistence of dengue-3 virus through transovarial transmission passage in successive generations of Aedes aegypti mosquitoes. Am. J. Trop. Med. Hyg. 67, 158–161 (2002).
Yeap, H. L. et al. Assessing quality of life-shortening Wolbachia-infected Aedes aegypti mosquitoes in the field based on capture rates and morphometric assessments. Parasit. Vectors 7, 58 (2014).
Dobson, S. L. & Rattanadechakul, W. A novel technique for removing Wolbachia infections from Aedes albopictus (Diptera: Culicidae). J. Med. Entomol. 38, 844–849 (2001).
McMeniman, C. J. et al. Stable introduction of a life-shortening Wolbachia infection into the mosquito Aedes aegypti. Science 323, 141–144 (2009).
Kriventseva, E. V. et al. OrthoDB v10: sampling the diversity of animal, plant, fungal, protist, bacterial and viral genomes for evolutionary and functional annotations of orthologs. Nucleic Acids Res. 47, D807–D811 (2018).
Dutton, T. J. & Sinkins, S. P. Strain-specific quantification of Wolbachia density in Aedes albopictus and effects of larval rearing conditions. Insect Mol. Biol. 13, 317–322 (2004).
Dutra, H. L. et al. The influence of larval competition on Brazilian Wolbachia-infected Aedes aegypti mosquitoes. Parasit. Vectors 9, 282 (2016).
Ross, P. A., Endersby, N. M., Yeap, H. L. & Hoffmann, A. A. Larval competition extends developmental time and decreases adult size of wMelPop Wolbachia-infected Aedes aegypti. Am. J. Trop. Med. Hyg. 91, 198–205 (2014).
Joy, T. K., Arik, A. J., Corby-Harris, V., Johnson, A. A. & Riehle, M. A. The impact of larval and adult dietary restriction on lifespan, reproduction and growth in the mosquito Aedes aegypti. Exp. Gerontol. 45, 685–690 (2010).
Mains, J. W., Mercer, D. R. & Dobson, S. L. Digital image analysis to estimate numbers of Aedes eggs oviposited in containers. J. Am. Mosq. Control Assoc. 24, 496–501 (2008).
Kozłowski, J. Measuring fitness in life history studies. Trends Ecol. Evol. 8, 84–85 (1993).
Jha, A. R. et al. Shared genetic signals of hypoxia adaptation in Drosophila and in high-altitude human populations. Mol. Biol. Evol. 33, 501–517 (2016).
Acknowledgements
We thank E. Kerton, M. Jones, E. Kennedy, H. Amuzu, G. Terradas, M. Novelo Canto, S. Mole, D. Guy, L. Jimenez, C. Hammer and C. Koh for technical support throughout the experiment. We thank D. Marshall, C. Godfray and K. King for helpful discussions on the manuscript. This work was supported by a grant (APP1103804) from the National Health and Medical Research Council of Australia to E.A.M.
Author information
Authors and Affiliations
Contributions
S.A.F. and E.A.M. conceived and designed the experiments and wrote the manuscript. S.A.F. planned and performed the experiments, including mosquito rearing and infection, selection, RNA and DNA collection and analysis, antibiotic treatment, and life-history measurements. L.T.S. measured candidate gene expression. S.A.F. performed statistical analysis and interpreted the data. S.L.A., A.S., S.F.C. and I.A. collaboratively processed and statistically analysed the genomic data. S.A.F. interpreted the results. J.R.O. and S.A.F. designed the Leslie matrix models to assess mosquito fitness. J.R.O. ran the models. S.A.F. statistically analysed and interpreted the data.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–3, Tables 1–2 and captions for supplementary files.
Supplementary File 1
Realized heritability of Wolbachia-mediated dengue blocking.
Supplementary File 2
A summary of SNPs in genes that are significantly differentiated in the A. aegypti genome between the high and low-blocking populations.
Supplementary File 3
A summary of SNPs in genes that are significantly differentiated in the A. aegypti genome between the random and low-blocking populations.
Rights and permissions
About this article
Cite this article
Ford, S.A., Allen, S.L., Ohm, J.R. et al. Selection on Aedes aegypti alters Wolbachia-mediated dengue virus blocking and fitness. Nat Microbiol 4, 1832–1839 (2019). https://doi.org/10.1038/s41564-019-0533-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-019-0533-3
This article is cited by
-
Cross-tissue and generation predictability of relative Wolbachia densities in the mosquito Aedes aegypti
Parasites & Vectors (2022)
-
Attempts to use breeding approaches in Aedes aegypti to create lines with distinct and stable relative Wolbachia densities
Heredity (2022)
-
Genetic stability of Aedes aegypti populations following invasion by wMel Wolbachia
BMC Genomics (2021)
-
Combating mosquito-borne diseases using genetic control technologies
Nature Communications (2021)
-
Two New Strains of Wolbachia Affecting Natural Avocado Thrips
Indian Journal of Microbiology (2021)