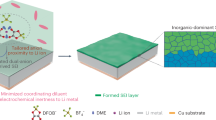
Abstract
For halide perovskites that are susceptible to photolysis and ion migration, iodide-related defects, such as iodine (I2) and iodine vacancies, are inevitable. Even a small number of these defects can trigger self-accelerating chemical reactions, posing serious challenges to the durability of perovskite solar cells. Fortunately, before I2 can damage the perovskites under illumination, they generally diffuse over a long distance. Therefore, detrimental I2 can be captured by interfacial materials with strong iodide/polyiodide (Ix−) affinities, such as fullerenes and perfluorodecyl iodide. However, fullerenes in direct contact with perovskites fail to confine Ix− ions within the perovskite layer but cause detrimental iodine vacancies. Perfluorodecyl iodide, with its directional Ix− affinity through halogen bonding, can both capture and confine Ix−. Therefore, inverted perovskite solar cells with over 10 times improved ultraviolet irradiation and thermal-light stabilities (under 85 °C and 1 sun illumination), and 1,000 times improved reverse-bias stability (under ISOS-V ageing tests) have been developed.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding authors on reasonable request. Source data are provided with this paper.
References
Kojima, A., Teshima, K., Shirai, Y. & Miyasaka, T. Organometal halide perovskites as visible-light sensitizers for photovoltaic cells. J. Am. Chem. Soc. 131, 6050–6051 (2009).
Mitzi, D. B., Field, C. A., Harrison, W. T. A. & Guloy, A. M. Conducting tin halides with a layered organic-based perovskite structure. Nature 369, 467–469 (1994).
Zhao, Y. et al. Inactive (PbI2)2RbCl stabilizes perovskite films for efficient solar cells. Science 377, 531–534 (2022).
Fei, C. et al. Lead-chelating hole-transport layers for efficient and stable perovskite minimodules. Science 380, 823–829 (2023).
Gu, H. et al. Design optimization of bifacial perovskite minimodules for improved efficiency and stability. Nat. Energy 8, 675–684 (2023).
National Renewable Energy Laboratory, Best Research-Cell Efficiency Chart (2023); https://www.nrel.gov/pv/cell-efficiency.html
Green, M. A. et al. Solar cell efficiency tables (Version 63). Prog. Photovoltaics Res. Appl. 32, 3–13 (2024).
Phung, N. et al. Photoprotection in metal halide perovskites by ionic defect formation. Joule 6, 2152–2174 (2022).
Eames, C. et al. Ionic transport in hybrid lead iodide perovskite solar cells. Nat. Commun. 6, 7497 (2015).
Yuan, Y. & Huang, J. Ion migration in organometal trihalide perovskite and its impact on photovoltaic efficiency and stability. Acc. Chem. Res. 49, 286–293 (2016).
Deng, Y. et al. Defect compensation in formamidinium–caesium perovskites for highly efficient solar mini-modules with improved photostability. Nat. Energy 6, 633–641 (2021).
Xie, H. et al. Decoupling the effects of defects on efficiency and stability through phosphonates in stable halide perovskite solar cells. Joule 5, 1246–1266 (2021).
Lin, D. et al. Ion migration accelerated reaction between oxygen and metal halide perovskites in light and its suppression by cesium incorporation. Adv. Energy Mater. 11, 2002552 (2021).
Tan, S. et al. Shallow iodine defects accelerate the degradation of α-phase formamidinium perovskite. Joule 4, 2426–2442 (2020).
Aristidou, N. et al. Fast oxygen diffusion and iodide defects mediate oxygen-induced degradation of perovskite solar cells. Nat. Commun. 8, 15218 (2017).
Wang, S. et al. Accelerated degradation of methylammonium lead iodide perovskites induced by exposure to iodine vapour. Nat. Energy 2, 16195 (2017).
Kato, Y. et al. Silver iodide formation in methyl ammonium lead iodide perovskite solar cells with silver top electrodes. Adv. Mater. Interfaces 2, 1500195 (2015).
Khenkin, M. V. et al. Consensus statement for stability assessment and reporting for perovskite photovoltaics based on ISOS procedures. Nat. Energy 5, 35–49 (2020).
Ni, Z. et al. Evolution of defects during the degradation of metal halide perovskite solar cells under reverse bias and illumination. Nat. Energy 7, 65–73 (2022).
Razera, R. A. Z. et al. Instability of p–i–n perovskite solar cells under reverse bias. J. Mater. Chem. A 8, 242–250 (2020).
Kang, D. H., Kim, S. Y., Lee, J. W. & Park, N. G. Efficient surface passivation of perovskite films by a post-treatment method with a minimal dose. J. Mater. Chem. A 9, 3441–3450 (2021).
Udalova, N. N. et al. New features of photochemical decomposition of hybrid lead halide perovskites by laser irradiation. ACS Appl. Mater. Interfaces 12, 12755–12762 (2020).
Petrov, A. A. et al. A new formation strategy of hybrid perovskites via room temperature reactive polyiodide melts. Mater. Horiz. 4, 625–632 (2017).
Pearson, R. G. & Songstad, J. Application of the principle of hard and soft acids and bases to organic chemistry. J. Am. Chem. Soc. 89, 1827–1836 (1967).
Svensson, P. H. & Kloo, L. Synthesis, structure, and bonding in polyiodide and metal iodide–iodine systems. Chem. Rev. 103, 1649–1684 (2003).
Boschloo, G. & Hagfeldt, A. Characteristics of the iodide/triiodide redox mediator in dye-sensitized solar cells. Acc. Chem. Res. 42, 1819–1826 (2009).
Xu, J. et al. Perovskite–fullerene hybrid materials suppress hysteresis in planar diodes. Nat. Commun. 6, 7081 (2015).
Metrangolo, P. et al. Halogen bonding in perovskite solar cells: a new tool for improving solar energy conversion. Angew. Chem. Int. Ed. 61, e202114793 (2022).
Abate, A. et al. Halogen-bond driven self-assembly of perfluorocarbon monolayers on silicon nitride. J. Mater. Chem. A 7, 24445–24453 (2019).
Abate, A. et al. Supramolecular halogen bond passivation of organic–inorganic halide perovskite solar cells. Nano Lett. 14, 3247–3254 (2014).
Liu, F. et al. Is excess PbI2 beneficial for perovskite solar cell performance? Adv. Energy Mater. 6, 1502206 (2016).
Teymourinia, H. et al. Interfacial passivation of perovskite solar cells by reactive ion scavengers. ACS Appl. Energy Mater. 4, 1078–1084 (2021).
Akbulatov, A. F. et al. Effect of electron-transport material on light-induced degradation of inverted planar junction perovskite solar cells. Adv. Energy Mater. 7, 1700476 (2017).
Hu, J. et al. Tracking the evolution of materials and interfaces in perovskite solar cells under an electric field. Commun. Mater. 3, 39 (2022).
Ball, M. L., Millic, J. V. & Loo, Y.-L. The emerging role of halogen bonding in hybrid perovskite photovoltaics. Chem. Mater. 34, 2495–2502 (2022).
Kim, G. Y. et al. Large tunable photoeffect on ion conduction in halide perovskites and implications for photodecomposition. Nat. Mater. 17, 445–449 (2018).
Canil, L. et al. Tuning halide perovskite energy levels. Energ. Environ. Sci. 14, 1429–1438 (2021).
Mei, A. et al. Stabilizing perovskite solar cells to IEC61215:2016 standards with over 9000-h operational tracking. Joule 4, 2646–2660 (2020).
Wolff, C. M. et al. Perfluorinated self-assembled monolayers enhance the stability and efficiency of inverted perovskite solar cells. ACS Nano 14, 16156–16156 (2020).
Acknowledgements
Y. Yuan acknowledges financial support from National Key Research and Development Program of China (2022YFB3803300), financial support from National Natural Science Foundation of China (52273202, 51673218). J. He acknowledges financial support from National Natural Science Foundation of China (62275275). L.Y. acknowledges financial support from National Natural Science Foundation of China (62104261). B.Y. acknowledges financial support from National Natural Science Foundation of China (62004066). H.H. acknowledges financial support from National Natural Science Foundation of China (11874427). Y. Yuan acknowledges the Major Scientific and Technological Project of Changsha (kq2301002), the Key Project of the Natural Science Program of Xinjiang Uygur Autonomous Region (2023D01D03) and the Innovation-Driven Project of Central South University (2020CX006). Y.W. acknowledges financial support from the Natural Science Foundation of Hunan Province (2021JJ40709, 2022JJ20080).
Author information
Authors and Affiliations
Contributions
Y. Yuan conducted the project, conceived the ideas, designed the experiments and analysed the data. J. Huang supervised the project and analysed the data. J. He supervised the Raman spectral and absorption spectral studies. X.R. fabricated the perovskite films, and studied the iodide/polyiodide capture effect of Lewis acids. J.W. and X.R. fabricated the solar cells, and obtained the J–V curves, the EQE and the NMR specta. X.R. and J.W. measured the activation energies of ion migration, the reverse-bias stability and the working stability of solar cells. X.R., Y.L. and Y.W. carried out the ultraviolet–visible absorption spectrum measurements, X.R. and B.Y. carried out the SEM measurements, J.W., H.X. and Y.G. carried out the XPS test and analysis. X.R. and H.H. carried out the Raman spectral tests. Y. Yan carried out the density functional theory calculations. Y. Yuan, J. Huang and J. He wrote the paper; all authors revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Materials thanks Antonio Abate, Jin-Wook Lee and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Notes 1–7, Figs. 1–33 and Tables 1–5.
Source data
Source Data Fig. 1
Statistical source data in Fig. 1c
Source Data Fig. 2
Unprocessed NMR data in Fig. 2b; raw data used for fitting in Fig. 2d–f; statistical source data in Fig. 2g.
Source Data Fig. 3
Unprocessed J–V data in Fig. 3b; statistical source data in Fig. 3c,d; unprocessed J–V data in Fig. 3g.
Source Data Fig. 4
Statistical source data in Fig. 4a; unprocessed J–V data in Fig. 4b; statistical source data in Fig. 4c–f.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ren, X., Wang, J., Lin, Y. et al. Mobile iodides capture for highly photolysis- and reverse-bias-stable perovskite solar cells. Nat. Mater. (2024). https://doi.org/10.1038/s41563-024-01876-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41563-024-01876-2