Abstract
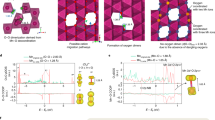
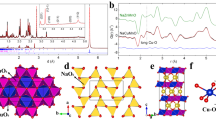
Lattice oxygen redox offers an unexplored way to access superior electrochemical properties of transition metal oxides (TMOs) for rechargeable batteries. However, the reaction is often accompanied by unfavourable structural transformations and persistent electrochemical degradation, thereby precluding the practical application of this strategy. Here we explore the close interplay between the local structural change and oxygen electrochemistry during short- and long-term battery operation for layered TMOs. The substantially distinct evolution of the oxygen-redox activity and reversibility are demonstrated to stem from the different cation-migration mechanisms during the dynamic de/intercalation process. We show that the π stabilization on the oxygen oxidation initially aids in the reversibility of the oxygen redox and is predominant in the absence of cation migrations; however, the π-interacting oxygen is gradually replaced by σ-interacting oxygen that triggers the formation of O–O dimers and structural destabilization as cycling progresses. More importantly, it is revealed that the distinct cation-migration paths available in the layered TMOs govern the conversion kinetics from π to σ interactions. These findings constitute a step forward in unravelling the correlation between the local structural evolution and the reversibility of oxygen electrochemistry and provide guidance for further development of oxygen-redox layered electrode materials.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All relevant experimental and theoretical data within the article will be provided by the corresponding author on reasonable request.
References
Grimaud, A. et al. Activating lattice oxygen redox reactions in metal oxides to catalyse oxygen evolution. Nat. Chem. 9, 457–465 (2017).
Grimaud, A., Hong, W. T., Shao-Horn, Y. & Tarascon, J. M. Anionic redox processes for electrochemical devices. Nat. Mater. 15, 121–126 (2016).
Sathiya, M. et al. Reversible anionic redox chemistry in high-capacity layered-oxide electrodes. Nat. Mater. 12, 827–835 (2013).
Assat, G. & Tarascon, J.-M. Fundamental understanding and practical challenges of anionic redox activity in Li-ion batteries. Nat. Energy 3, 373–386 (2018).
Li, B. & Xia, D. Anionic redox in rechargeable lithium batteries. Adv. Mater. 29, 1701054 (2017).
Luo, K. et al. Charge-compensation in 3d-transition-metal-oxide intercalation cathodes through the generation of localized electron holes on oxygen. Nat. Chem. 8, 684–691 (2016).
Seo, D.-H. et al. The structural and chemical origin of the oxygen redox activity in layered and cation-disordered Li-excess cathode materials. Nat. Chem. 8, 692–697 (2016).
Song, J.-H. et al. Anionic redox activity regulated by transition metal in lithium-rich layered oxides. Adv. Energy Mater. 10, 2001207 (2020).
Xie, Y., Saubanère, M. & Doublet, M. L. Requirements for reversible extra-capacity in Li-rich layered oxides for Li-ion batteries. Energy Environ. Sci. 10, 266–274 (2017).
Gent, W. E. et al. Coupling between oxygen redox and cation migration explains unusual electrochemistry in lithium-rich layered oxides. Nat. Commun. 8, 2091 (2017).
Hong, J. et al. Metal–oxygen decoordination stabilizes anion redox in Li-rich oxides. Nat. Mater. 18, 256–265 (2019).
Ku, K. et al. A new lithium diffusion model in layered oxides based on asymmetric but reversible transition metal migration. Energy Environ. Sci. 13, 1269–1278 (2020).
Eum, D. et al. Voltage decay and redox asymmetry mitigation by reversible cation migration in lithium-rich layered oxide electrodes. Nat. Mater. 19, 419–427 (2020).
Ben Yahia, M., Vergnet, J., Saubanère, M. & Doublet, M.-L. Unified picture of anionic redox in Li/Na-ion batteries. Nat. Mater. 18, 496–502 (2019).
Vergnet, J., Saubanère, M., Doublet, M.-L. & Tarascon, J.-M. The structural stability of P2-layered Na-based electrodes during anionic redox. Joule 4, 420–434 (2020).
Assat, G. et al. Fundamental interplay between anionic/cationic redox governing the kinetics and thermodynamics of lithium-rich cathodes. Nat. Commun. 8, 2219 (2017).
Assat, G., Delacourt, C., Corte, D. A. D. & Tarascon, J.-M. Editors’ choice—practical assessment of anionic redox in Li-rich layered oxide cathodes: a mixed blessing for high energy Li-ion batteries. J. Electrochem. Soc. 163, A2965–A2976 (2016).
McCalla, E. et al. Visualization of O–O peroxo-like dimers in high-capacity layered oxides for Li-ion batteries. Science 350, 1516–1521 (2015).
Chen, H. & Islam, M. S. Lithium extraction mechanism in Li-rich Li2MnO3 involving oxygen hole formation and dimerization. Chem. Mater. 28, 6656–6663 (2016).
Chen, Z., Li, J. & Zeng, X. C. Unraveling oxygen evolution in Li-rich oxides: a unified modeling of the intermediate peroxo/superoxo-like dimers. J. Am. Chem. Soc. 141, 10751–10759 (2019).
Rong, X. et al. Structure-induced reversible anionic redox activity in Na layered oxide cathode. Joule 2, 125–140 (2018).
Mortemard de Boisse, B. Highly reversible oxygen-redox chemistry at 4.1 V in Na4/7−x[□1/7Mn6/7]O2 (□: Mn vacancy). Adv. Energy Mater. 8, 1800409 (2018).
Du, K. et al. Exploring reversible oxidation of oxygen in a manganese oxide. Energy Environ. Sci. 9, 2575–2577 (2016).
House, R. A. et al. Superstructure control of first-cycle voltage hysteresis in oxygen-redox cathodes. Nature 577, 502–508 (2020).
Cao, X. et al. Restraining oxygen loss and suppressing structural distortion in a newly Ti-substituted layered oxide P2-Na0.66Li0.22Ti0.15Mn0.63O2. ACS Energy Lett. 4, 2409–2417 (2019).
Somerville, J. W. et al. Nature of the ‘Z’-phase in layered Na-ion battery cathodes. Energy Environ. Sci. 12, 2223–2232 (2019).
Singh, G., López del Amo, J. M., Galceran, M., Pérez-Villar, S. & Rojo, T. Structural evolution during sodium deintercalation/intercalation in Na2/3[Fe1/2Mn1/2]O2. J. Mater. Chem. A 3, 6954–6961 (2015).
Hong, J. et al. Structural evolution of layered Li1.2Ni0.2Mn0.6O2 upon electrochemical cycling in a Li rechargeable battery. J. Mater. Chem. 20, 10179–10186 (2010).
Huang, Q. et al. Tailoring alternating heteroepitaxial nanostructures in Na-ion layered oxide cathodes via an in-situ composition modulation route. Nano Energy 44, 336–344 (2018).
Rong, X. et al. Anionic redox reaction-induced high-capacity and low-strain cathode with suppressed phase transition. Joule 3, 503–517 (2019).
Sudayama, T. et al. Multiorbital bond formation for stable oxygen-redox reaction in battery electrodes. Energy Environ. Sci. 13, 1492–1500 (2020).
Kitchaev, D. A., Vinckeviciute, J. & Van der Ven, A. Delocalized metal-oxygen π-redox is the origin of anomalous nonhysteretic capacity in Li-ion and Na-ion cathode materials. J. Am. Chem. Soc. 143, 1908–1916 (2021).
Freire, M. et al. A new active Li–Mn–O compound for high energy density Li-ion batteries. Nat. Mater. 15, 173–177 (2016).
Julien, C. M., Ait-Salah, A., Mauger, A. & Gendron, F. Magnetic properties of lithium intercalation compounds. Ionics 12, 21–32 (2006).
Cramer, C. J., Tolman, W. B., Theopold, K. H. & Rheingold, A. L. Variable character of O–O and M–O bonding in side-on (η2) 1:1 metal complexes of O2. Proc. Natl Acad. Sci. USA 100, 3635–3640 (2003).
Das, T. K., Couture, M., Ouellet, Y., Guertin, M. & Rousseau, D. L. Simultaneous observation of the O–O and Fe–O2 stretching modes in oxyhemoglobins. Proc. Natl Acad. Sci. USA 98, 479–484 (2001).
Krebs, C., Edmondson, D. E. & Huynh, B. H. Demonstration of peroxodiferric intermediate in M-Ferritin ferroxidase reaction using rapid Freeze-Quench Mössbauer, Resonance Raman, and XAS spectroscopies. Methods Enzymol. 354, 436–454 (2002).
Grimaud, A. et al. Activation of surface oxygen sites on an iridium-based model catalyst for the oxygen evolution reaction. Nat. Energy 2, 16189 (2016).
Schweinar, K. et al. Lattice oxygen exchange in rutile IrO2 during the oxygen evolution reaction. J. Phys. Chem. Lett. 11, 5008–5014 (2020).
Arciniegas Jaimes, D. M. et al. Effect of B-site order–disorder in the structure and magnetism of the new perovskite family La2MnB′O6 with B′ = Ti, Zr, and Hf. Inorg. Chem. 60, 4935–4944 (2021).
Wang, Y. L. et al. High stability of electro-transport and magnetism against the A-site cation disorder in SrRuO3. Sci. Rep. 6, 27840 (2016).
Zhuo, Z. et al. Spectroscopic signature of oxidized oxygen states in peroxides. J. Phys. Chem. Lett. 9, 6378–6384 (2018).
Yang, W. & Devereaux, T. P. Anionic and cationic redox and interfaces in batteries: advances from soft X-ray absorption spectroscopy to resonant inelastic scattering. J. Power Sources 389, 188–197 (2018).
Malavasi, L., Galinetto, P., Mozzati, M. C., Azzoni, C. B. & Flor, G. Raman spectroscopy of AMn2O4 (A = Mn, Mg and Zn) spinels. Phys. Chem. Chem. Phys. 4, 3876–3880 (2002).
McCloskey, B. D., Bethune, D. S., Shelby, R. M., Girishkumar, G. & Luntz, A. C. Solvents’ critical role in nonaqueous lithium–oxygen battery electrochemistry. J. Phys. Chem. Lett. 2, 1161–1166 (2011).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Jain, A. et al. A high-throughput infrastructure for density functional theory calculations. Comput. Mater. Sci. 50, 2295–2310 (2011).
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (no. NRF-2019M3E6A1064522), and Creative Materials Discovery Program through the NRF funded by the Ministry of Science, ICT and Future Planning (grant no. NRF-2017M3D1A1039553). This research was supported by the Institute for Basic Science (grant no. IBS-R006-A2) and Korea Electric Power Corporation (grant no. R20XO02-31).
Author information
Authors and Affiliations
Contributions
D.E. and K.K. conceived the basic idea for this project and led the research. D.E. performed the material synthesis, electrochemical testing and structural/spectroscopic characterization including HRPD, XANES, STXM and Raman analyses. B.K. conducted the DFT calculations. B.K., J.-H.S., H.P. and H.-Y.J. discussed and provided fundamental ideas for the overall study. S.J. Kim and S.-P.C. performed the STEM measurements. S.J. Kim contributed to the interpretation of the results. J.P. and S.J. Kang measured and processed the DEMS data. M.H.L. and J.H.H. provided constructive advice for the experimental design for the structural characterization. H.-Y.J. and Y.K. assisted with processing of the SQUID and DEMS results, respectively. S.K.P., J.K., K.O. and D.-H.K. offered valuable comments on this project. D.E. and K.K. wrote the paper. K.K. supervised all aspects of the research.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Materials thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Notes 1–9, Figs. 1–29, Tables 1–8 and refs. 1–12.
Rights and permissions
About this article
Cite this article
Eum, D., Kim, B., Song, JH. et al. Coupling structural evolution and oxygen-redox electrochemistry in layered transition metal oxides. Nat. Mater. 21, 664–672 (2022). https://doi.org/10.1038/s41563-022-01209-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-022-01209-1
This article is cited by
-
Highly reversible transition metal migration in superstructure-free Li-rich oxide boosting voltage stability and redox symmetry
Nature Communications (2024)
-
Freedom of chemical space
Nature Sustainability (2024)
-
Phase segregation and nanoconfined fluid O2 in a lithium-rich oxide cathode
Nature Materials (2024)
-
Structurally robust lithium-rich layered oxides for high-energy and long-lasting cathodes
Nature Communications (2024)
-
Electrochemomechanical failure in layered oxide cathodes caused by rotational stacking faults
Nature Materials (2024)