Abstract
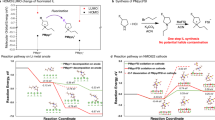
Metal fluorides, promising lithium-ion battery cathode materials, have been classified as conversion materials due to the reconstructive phase transitions widely presumed to occur upon lithiation. We challenge this view by studying FeF3 using X-ray total scattering and electron diffraction techniques that measure structure over multiple length scales coupled with density functional theory calculations, and by revisiting prior experimental studies of FeF2 and CuF2. Metal fluoride lithiation is instead dominated by diffusion-controlled displacement mechanisms, and a clear topological relationship between the metal fluoride F− sublattices and that of LiF is established. Initial lithiation of FeF3 forms FeF2 on the particle’s surface, along with a cation-ordered and stacking-disordered phase, A-LixFeyF3, which is structurally related to α-/β-LiMn2+Fe3+F6 and which topotactically transforms to B- and then C-LixFeyF3, before forming LiF and Fe. Lithiation of FeF2 and CuF2 results in a buffer phase between FeF2/CuF2 and LiF. The resulting principles will aid future developments of a wider range of isomorphic metal fluorides.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The authors declare that all data supporting the findings of this study are included within the paper and its Supplementary Information files. Source data are available from the corresponding authors upon reasonable request.
References
Cabana, J., Monconduit, L., Larcher, D. & Palacín, M. R. Beyond intercalation-based Li-ion batteries: the state of the art and challenges of electrode materials reacting through conversion reactions. Adv. Mater. 22, E170–E192 (2010).
Amatucci, G. G. & Pereira, N. Fluoride based electrode materials for advanced energy storage devices. J. Fluor. Chem. 128, 243–262 (2007).
Grenier, A. et al. Solid fluoride electrolytes and their composite with carbon: issues and challenges for rechargeable solid state fluoride-ion batteries. J. Phys. Chem. C 121, 24962–24970 (2017).
Thieu, D. T. et al. CuF2 as reversible cathode for fluoride ion batteries. Adv. Funct. Mater. 27, 1701051 (2017).
Zhang, L., Anji Reddy, M. & Fichtner, M. Development of tysonite-type fluoride conducting thin film electrolytes for fluoride ion batteries. Solid State Ion. 272, 39–44 (2015).
Anji Reddy, M. & Fichtner, M. Batteries based on fluoride shuttle. J. Mater. Chem. 21, 17059–17062 (2011).
Gschwind, F. et al. Fluoride ion batteries: theoretical performance, safety, toxicity, and a combinatorial screening of new electrodes. J. Fluor. Chem. 182, 76–90 (2016).
Davis, V. K. et al. Room-temperature cycling of metal fluoride electrodes: liquid electrolytes for high-energy fluoride ion cells. Science 362, 1144–1148 (2018).
Reddy, M. A. & Fichtner, M. in Advanced Fluoride-Based Materials for Energy Conversion (ed. Groult, H.) 51–76 (Elsevier, 2015).
Yamakawa, N., Jiang, M., Key, B. & Grey, C. P. Identifying the local structures formed during lithiation of the conversion material, iron fluoride, in a Li ion battery: a solid-state NMR, X-ray diffraction, and pair distribution function analysis study. J. Am. Chem. Soc. 131, 10525–10536 (2009).
Zhang, W. et al. In situ electrochemical XAFS studies on an iron fluoride high-capacity cathode material for rechargeable lithium batteries. J. Phys. Chem. C 117, 11498–11505 (2013).
Kim, S.-W., Seo, D.-H., Gwon, H., Kim, J. & Kang, K. Fabrication of FeF3 nanoflowers on CNT branches and their application to high power lithium rechargeable batteries. Adv. Mater. 22, 5260–5264 (2010).
Tan, H. J. et al. Electrochemical cycling and lithium insertion in nanostructured FeF3 cathodes. J. Electrochem. Soc. 161, A445–A449 (2014).
Badway, F., Cosandey, F., Pereira, N. & Amatucci, G. G. Carbon metal fluoride nanocomposites. J. Electrochem. Soc. 150, A1318–A1327 (2003).
Li, L. et al. Origins of large voltage hysteresis in high-energy-density metal fluoride lithium-ion battery conversion electrodes. J. Am. Chem. Soc. 138, 2838–2848 (2016).
Liu, P., Vajo, J. J., Wang, J. S., Li, W. & Liu, J. Thermodynamics and kinetics of the Li/FeF3 reaction by electrochemical analysis. J. Phys. Chem. C 116, 6467–6473 (2012).
Li, L. et al. Visualization of electrochemically driven solid-state phase transformations using operando hard X-ray spectro-imaging. Nat. Commun. 6, 6883 (2015).
Zhou, M., Zhao, L., Kitajou, A., Okada, S. & Yamaki, J.-I. Mechanism on exothermic heat of FeF3 cathode in Li-ion batteries. J. Power Sources 203, 103–108 (2012).
Badway, F., Pereira, N., Cosandey, F. & Amatucci, G. G. Carbon-metal fluoride nanocomposites: structure and electrochemistry of FeF3: C. J. Electrochem. Soc. 150, A1209–A1218 (2003).
Geddes, H. S., Blade, H., McCabe, J. F., Hughes, L. P. & Goodwin, A. L. Structural characterisation of amorphous solid dispersions via metropolis matrix factorisation of pair distribution function data. Chem. Commun. 55, 13346–13349 (2019).
West, A. R. & Bruce, P. G. Tetragonal-packed crystal structures. Acta Crystallogr. B 38, 1891–1896 (1982).
Yu, H. C. et al. Designing the next generation high capacity battery electrodes. Energy Environ. Sci. 7, 1760–1768 (2014).
Müller, U. Symmetry Relationships between Crystal Structures: Applications of Crystallographic Group Theory in Crystal Chemistry (Oxford Univ. Press, 2013).
Oszlanyi, G. & Suto, A. Ab initio structure solution by charge flipping. Acta Crystallogr. A 60, 134–141 (2004).
Rauch, E. et al. Automatic crystal orientation and phase mapping in TEM by precession diffraction. Microsc. Anal. 22, S5–S8 (2008).
Courbion, G., Jacoboni, C. & de Pape, R. The dimorphism of LiMnFeF6: a new kind of cationic order in the structural type Na2SiF6. J. Solid State Chem. 45, 127–134 (1982).
Doe, R. E., Persson, K. A., Meng, Y. S. & Ceder, G. First-principles investigation of the Li−Fe−F phase diagram and equilibrium and nonequilibrium conversion reactions of iron fluorides with lithium. Chem. Mater. 20, 5274–5283 (2008).
Goodenough, J. B. & Park, K.-S. The Li-ion rechargeable battery: a perspective. J. Am. Chem. Soc. 135, 1167–1176 (2013).
Tawa, S., Matsumoto, K. & Hagiwara, R. Reaction pathways of iron trifluoride investigated by operation at 363 K using an ionic liquid electrolyte. J. Electrochem. Soc. 166, A2105–A2110 (2019).
van der Ven, A., Garikipati, K., Kim, S. & Wagemaker, M. The role of coherency strains on phase stability in LixFePO4: needle crystallites minimize coherency strain and overpotential. J. Electrochem. Soc. 156, A949 (2009).
Karki, K. et al. Revisiting conversion reaction mechanisms in lithium batteries: lithiation-driven topotactic transformation in FeF2. J. Am. Chem. Soc. 140, 17915–17922 (2018).
Xiao, A. W. et al. Understanding the conversion mechanism and performance of monodisperse FeF2 nanocrystal cathodes. Nat. Mater. 19, 644–654 (2020).
Wang, F. et al. Conversion reaction mechanisms in lithium ion batteries: study of the binary metal fluoride electrodes. J. Am. Chem. Soc. 133, 18828–18836 (2011).
Jain, A. et al. Commentary: the materials project: a materials genome approach to accelerating materials innovation. APL Mater. 1, 011002 (2013).
Wang, F. et al. Tracking lithium transport and electrochemical reactions in nanoparticles. Nat. Commun. 3, 1201 (2012).
Wang, F. et al. Ternary metal fluorides as high-energy cathodes with low cycling hysteresis. Nat. Commun. 6, 6668 (2015).
Cosandey, F., Al-Sharab, J. F., Badway, F., Amatucci, G. G. & Stadelmann, P. EELS spectroscopy of iron fluorides and FeFx/C nanocomposite electrodes used in Li-ion batteries. Microsc. Microanal. 13, 87–95 (2007).
Sun, Y. et al. In situ chemical synthesis of lithium fluoride/metal nanocomposite for high capacity prelithiation of cathodes. Nano Lett. 16, 1497–1501 (2016).
Hori, H. & Okada, S. Reconversion reaction of LiF/Fe composite thin film cathodes for lithium-ion battery. Electrochemistry 83, 909–913 (2015).
Li, T., Chen, Z. X., Ai, X. P., Cao, Y. L. & Yang, H. X. LiF/Fe nanocomposite as a lithium-rich and high capacity conversion cathode material for Li-ion batteries. J. Power Sources 217, 54–58 (2012).
Hua, X. et al. Comprehensive study of the CuF2 conversion reaction mechanism in a lithium ion battery. J. Phys. Chem. C 118, 15169–15184 (2014).
Huang, Q. et al. Fading mechanisms and voltage hysteresis in FeF2–NiF2 solid solution cathodes for lithium and lithium-ion batteries. Small 15, 1804670 (2019).
Pereira, N., Badway, F., Wartelsky, M., Gunn, S. & Amatucci, G. G. Iron oxyfluorides as high capacity cathode materials for lithium batteries. J. Electrochem. Soc. 156, A407 (2009).
Hammersley, A. FIT2D V9.129 Reference Manual V3.1 ESRF Internal Report 98HA01T (ESRF, 1998).
Basham, M. et al. Data Analysis WorkbeNch (DAWN). J. Synchrotron Radiat. 22, 853–858 (2015).
Qiu, X., Thompson, J. W. & Billinge, S. L. J. PDFgetX2: a GUI-driven program to obtain the pair distribution function from X-ray powder diffraction data. Appl. Crystallogr. 37, 678 (2004).
Toby, B. H. & von Dreele, R. B. GSAS-II: the genesis of a modern open-source all purpose crystallography software package. J. Appl. Crystallogr. 46, 544–549 (2013).
Farrow, C. L. et al. PDFfit2 and PDFgui: computer programs for studying nanostructure in crystals. J. Phys. Condens. Matter 19, 335219 (2007).
Proffen, T. & Neder, R. B. DISCUS, a program for diffuse scattering and defect structure simulations – update. J. Appl. Crystallogr. 32, 838–839 (1999).
Treacy, M. M. J., Newsam, J. M. & Deem, M. W. A general recursion method for calculating diffracted intensities from crystals containing planar faults. Proc. Math. Phys. Eng. Sci. 433, 499–520 (1991).
Pickard, C. J. & Needs, R. J. High-pressure phases of silane. Phys. Rev. Lett. 97, 045504 (2006).
Pickard, C. J. & Needs, R. J. Ab initio random structure searching. J. Phys. Condens. Matter 23, 053201 (2011).
Clark, S. J. et al. First principles methods using CASTEP. Z. Kristallogr. Cryst. Mater. 220, 567–570 (2005).
Lee, D. D. & Seung, H. S. Learning the parts of objects by non-negative matrix factorization. Nature 401, 788–791 (1999).
Egami, T. & Billinge, S. J. L. Underneath the Bragg Peaks Structural Analysis of Complex Materials (Pergamon, 2012).
Acknowledgements
X.H. is supported by funding from the Engineering and Physical Sciences Research Council (EPSRC) Doctoral Prize, Adolphe Merkle and the Swiss National Science Foundation (Program NRP70 no. 153990) and European Commission via Marie Skłodowska-Curie actions (MSCA) (grant 798169). A.S.E. acknowledges financial support from the Royal Society. E.C.-M. acknowledges funding from the European Commission via MSCA (grant 747449) and RTI2018-094550-A-l00 from the Ministerio de Ciencia e Innovación. H.S.G. is supported by EPSRC via Industrial Cooperative Awards in Science and Technology studentship. Z.L. is supported by the Faraday Institution (grant number FIRG017). C.J.P. is supported by the Royal Society through a Royal Society Wolfson Research Merit award and by EPSRC grant EP/P022596/1. A.L.G. acknowledges funding from the European Research Council (grant 788144). This research was supported as part of the North Eastern Center for Chemical Energy Storage, an Energy Frontier Research Center funded by the US Department of Energy, Office of Science, and Office of Basic Energy Sciences under award number DE-SC0001294. Work done at Argonne and use of the Advanced Photon Source, an Office of Science User Facility operated for the US Department of Energy Office of Science by Argonne National Laboratory, was supported by the US Department of Energy under contract no. DE-AC02-06CH11357. Work done at Diamond Light Source was under proposal EE17315-1. We thank G. Ceder and other North Eastern Center for Chemical Energy Storage members for many stimulating discussions concerning fluoride-based conversion reactions and on the origins of structural hysteresis. We also acknowledge help from S. Dutton, T. Dean, A. Docker, M. Leskes and D. Keeble.
Author information
Authors and Affiliations
Contributions
X.H., E.C.-M. and R.R. planned the project with C.P.G.; E.C.-M. and R.R. prepared the pristine materials with help from N.P. and G.G.A.; X.H., E.C.-M., R.R. and W.M. performed the electrochemistry tests and prepared samples for ex situ characterization; E.C.-M. and R.R. performed high-resolution X-ray diffraction measurements; A.S.E. acquired and analysed the TEM data with support from P.A.M.; E.C.-M. performed the magnetic measurements and analysed the data; X.H., R.R. and K.M.W. acquired PDF data with support from U.S. and K.W.C.; X.H. performed analyses of the electrochemistry, X-ray diffraction and PDF data; Z.L. and C.J.P. performed the DFT calculations; X.H., H.S.G. and A.L.G. performed the NMF analysis; X.H. and C.P.G. wrote the manuscript with input from all the coauthors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Materials thanks Gleb Yushin and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 NMR, XRD and PDF results of n-FeF3 in the first cycle.
a) Galvanostatic profile of n-FeF3’s first cycle with distinct reaction processes indicated by different colour backgrounds. The 6Li MAS NMR chemical shifts (red) from our previous study10 are labelled at their respective state of charge with the main phase identification indicated. Black squares mark the states of charge where ex situ X-ray total scattering experiments were performed to acquire b) XRD and c) PDF patterns. Green, blue and pink dotted lines indicate unique Bragg or PDF features from FeF3, LixFeyF3 and FeF2, respectively. The red arrow highlights a drastic decrease in the samples’ particle sizes upon charge. The evolution of the phase mole fractions obtained from the PDF refinement is shown in d) with the deduced step-by-step mechanism diagram indicated on the left, which echoes with the simplified scheme shown in Fig. 5d.
Extended Data Fig. 2 Li-Fe-F phase diagram.
The reference phases in the phase diagram are labelled and indicated by light blue circles so as to show the positions of A- and B-LixFeyF3, whose Fe concentration is off-stoichiometric. The reaction pathways associated with the FeF3 and FeF2 systems are respectively marked by using green and pink dashed arrows. Each reaction process is also labelled with its respective roman numeral used to label the equations in the manuscript.
Supplementary information
Supplementary Information
Supplementary Sections 1–11, Figs. 1–24 and Tables 1 and 2.
Supplementary Video 1
NMF analysis of the discharged FeF2.
Supplementary Video 2
NMF analysis of the discharged CuF2.
Computational Data 1
DFT calculation data.
Rights and permissions
About this article
Cite this article
Hua, X., Eggeman, A.S., Castillo-Martínez, E. et al. Revisiting metal fluorides as lithium-ion battery cathodes. Nat. Mater. 20, 841–850 (2021). https://doi.org/10.1038/s41563-020-00893-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-020-00893-1
This article is cited by
-
Converting intercalation-type cathode in spent lithium-ion batteries into conversion-type cathode
Nano Research (2024)
-
Tactics to optimize conversion-type metal fluoride/sulfide/oxide cathodes toward advanced lithium metal batteries
Nano Research (2023)
-
Enabling the reversibility of anhydrous copper(II) fluoride cathodes for rechargeable lithium batteries via fluorinated high-concentration electrolytes
Science China Materials (2023)
-
Thermal synthesis of conversion-type bismuth fluoride cathodes for high-energy-density Li-ion batteries
Communications Chemistry (2022)
-
The challenges and opportunities of battery-powered flight
Nature (2022)