Abstract
Stromal stiffening accompanies malignancy, compromises treatment and promotes tumour aggression. Clarifying the molecular nature and the factors that regulate stromal stiffening in tumours should identify biomarkers to stratify patients for therapy and interventions to improve outcome. We profiled lysyl hydroxylase-mediated and lysyl oxidase-mediated collagen crosslinks and quantified the greatest abundance of total and complex collagen crosslinks in aggressive human breast cancer subtypes with the stiffest stroma. These tissues harbour the highest number of tumour-associated macrophages, whose therapeutic ablation in experimental models reduced metastasis, and decreased collagen crosslinks and stromal stiffening. Epithelial-targeted expression of the crosslinking enzyme, lysyl oxidase, had no impact on collagen crosslinking in PyMT mammary tumours, whereas stromal cell targeting did. Stromal cells in microdissected human tumours expressed the highest level of collagen crosslinking enzymes. Immunohistochemical analysis of biopsies from a cohort of patients with breast cancer revealed that stromal expression of lysyl hydroxylase 2, an enzyme that induces hydroxylysine aldehyde-derived collagen crosslinks and stromal stiffening, correlated significantly with disease specific mortality. The findings link tissue inflammation, stromal cell-mediated collagen crosslinking and stiffening to tumour aggression and identify lysyl hydroxylase 2 as a stromal biomarker.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All gene expression array and RNA-seq data have been deposited in GEO and are publicly available. For the stroma and epithelium specific gene expression analysis, the breast cancer datasets from Finak et al.37 and Gruosso et al.36 have been used (GSE9014, GSE58644, GSE88715 and GSE88847). The association between PLOD2 and DMFS was determined using an online tool (http://xena.ucsc.edu) to download GEO data (GSE2034, GSE5327 and GSE7390) from 683 patients analysed on Affymetrix U133A platform as described in Yau et al.47. The association between PLOD2 gene expression and RFS was determined using an online tool (http://kmplot.com/analysis/) from 1,809 patients analysed on Affymetrix platform (HGU133A and HGU133+2 microarrays)48. Affymetrix ID 202619 or 202620 were used for PLOD2 probes (2014 version) in these analyses. The cBioPortal for Cancer Genomics was used to access the publicly available METABRIC dataset to determine the levels of LOX, PLOD2 and LOXL2 gene expression in patients with breast cancer segregated by ER and HER2 status and to assess gene expression associations of LOX, PLOD2 and LOXL2 with CD16363. Macrophage RNA-seq data have been deposited into GEO (GSE157290). Source data are provided with this paper.
References
Laklai, H. et al. Genotype tunes pancreatic ductal adenocarcinoma tissue tension to induce matricellular fibrosis and tumor progression. Nat. Med. 22, 497–505 (2016).
Acerbi, I. et al. Human breast cancer invasion and aggression correlates with ECM stiffening and immune cell infiltration. Integr. Biol. 7, 1120–1134 (2015).
Dvorak, H. F. Tumors: wounds that do not heal. Similarities between tumor stroma generation and wound healing. N. Engl. J. Med. 315, 1650–1659 (1986).
Conklin, M. W. et al. Aligned collagen is a prognostic signature for survival in human breast carcinoma. Am. J. Pathol. 178, 1221–1232 (2011).
Olive, K. P. et al. Inhibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science 324, 1457–1461 (2009).
Provenzano, P. P. et al. Enzymatic targeting of the stroma ablates physical barriers to treatment of pancreatic ductal adenocarcinoma. Cancer Cell 21, 418–429 (2012).
Netti, P. A., Berk, D. A., Swartz, M. A., Grodzinsky, A. J. & Jain, R. K. Role of extracellular matrix assembly in interstitial transport in solid tumors. Cancer Res. 60, 2497–2503 (2000).
Jain, R. K., Lee, J. J., Hong, D. & Kurzrock, R. Reply to A. Levy et al. J. Clin. Oncol. 31, 396 (2013).
Egeblad, M., Rasch, M. G. & Weaver, V. M. Dynamic interplay between the collagen scaffold and tumor evolution. Curr. Opin. Cell Biol. 22, 697–706 (2010).
Chaudhuri, O. et al. Extracellular matrix stiffness and composition jointly regulate the induction of malignant phenotypes in mammary epithelium. Nat. Mater. 13, 970–978 (2014).
Provenzano, P. P., Inman, D. R., Eliceiri, K. W. & Keely, P. J. Matrix density-induced mechanoregulation of breast cell phenotype, signaling and gene expression through a FAK-ERK linkage. Oncogene 28, 4326–4343 (2009).
Levental, K. R. et al. Matrix crosslinking forces tumor progression by enhancing integrin signaling. Cell 139, 891–906 (2009).
Mekhdjian, A. H. et al. Integrin-mediated traction force enhances paxillin molecular associations and adhesion dynamics that increase the invasiveness of tumor cells into a three-dimensional extracellular matrix. Mol. Biol. Cell 28, 1467–1488 (2017).
Pickup, M. W. et al. Stromally derived lysyl oxidase promotes metastasis of transforming growth factor-beta-deficient mouse mammary carcinomas. Cancer Res. 73, 5336–5346 (2013).
Pickup, M. W., Mouw, J. K. & Weaver, V. M. The extracellular matrix modulates the hallmarks of cancer. EMBO Rep. 15, 1243–1253 (2014).
Provenzano, P. P. et al. Collagen density promotes mammary tumor initiation and progression. BMC Med. 6, 11 (2008).
Eyre, D. R., Weis, M. A. & Wu, J.-J. Advances in collagen cross-link analysis. Methods 45, 65–74 (2008).
Yamauchi M. & Shiiba M. in Post-translational Modifications of Proteins (ed. Kannicht, C.) 95–108 (Humana, 2008).
Yamauchi, M. & Sricholpech, M. Lysine post-translational modifications of collagen. Essays Biochem. 52, 113–133 (2012).
Miller, B. W. et al. Targeting the LOX/hypoxia axis reverses many of the features that make pancreatic cancer deadly: inhibition of LOX abrogates metastasis and enhances drug efficacy. EMBO Mol. Med. 7, 1063–1076 (2015).
Erler, J. T. & Weaver, V. M. Three-dimensional context regulation of metastasis. Clin. Exp. Metastasis 26, 35–49 (2009).
Chen, Y. et al. Lysyl hydroxylase 2 induces a collagen cross-link switch in tumor stroma. J. Clin. Invest. 125, 1147 (2015).
Gilkes, D. M. et al. Procollagen lysyl hydroxylase 2 is essential for hypoxia-induced breast cancer metastasis. Mol. Cancer Res. 11, 456–466 (2013).
Lampi, M. C. & Reinhart-King, C. A. Targeting extracellular matrix stiffness to attenuate disease: from molecular mechanisms to clinical trials. Sci. Transl. Med. https://doi.org/10.1126/scitranslmed.aao0475 (2018).
Grivennikov, S. I., Greten, F. R. & Karin, M. Immunity, inflammation, and cancer. Cell 140, 883–899 (2010).
Mack, M. Inflammation and fibrosis. Matrix Biol. 68–69, 106–121 (2018).
Wick, G. et al. The immunology of fibrosis. Annu. Rev. Immunol. 31, 107–135 (2013).
Ueha, S., Shand, F. H. & Matsushima, K. Cellular and molecular mechanisms of chronic inflammation-associated organ fibrosis. Front. Immunol. 3, 71 (2012).
Sun, X. et al. CCL2-driven inflammation increases mammary gland stromal density and cancer susceptibility in a transgenic mouse model. Breast Cancer Res. 19, 4 (2017).
Qian, B. Z. et al. CCL2 recruits inflammatory monocytes to facilitate breast-tumour metastasis. Nature 475, 222–225 (2011).
Lohela, M. et al. Intravital imaging reveals distinct responses of depleting dynamic tumor-associated macrophage and dendritic cell subpopulations. Proc. Natl Acad. Sci. USA 111, E5086–E5095 (2014).
Ruffell, B. et al. Macrophage IL-10 blocks CD8+ T cell-dependent responses to chemotherapy by suppressing IL-12 expression in intratumoral dendritic cells. Cancer Cell 26, 623–637 (2014).
DeNardo, D. G. et al. Leukocyte complexity predicts breast cancer survival and functionally regulates response to chemotherapy. Cancer Discov. 1, 54–67 (2011).
Nemkov, T., D’Alessandro, A. & Hansen, K. C. Three-minute method for amino acid analysis by UHPLC and high-resolution quadrupole orbitrap mass spectrometry. Amino Acids 47, 2345–2357 (2015).
Oxlund, H., Barckman, M., Ørtoft, G. & Andreassen, T. Reduced concentrations of collagen cross-links are associated with reduced strength of bone. Bone 17, S365–S371 (1995).
Gruosso, T. et al. Spatially distinct tumor immune microenvironments stratify triple-negative breast cancers. J. Clin. Invest. 129, 1785–1800 (2019).
Finak, G. et al. Stromal gene expression predicts clinical outcome in breast cancer. Nat. Med. 14, 518–527 (2008).
Salvador, F. et al. Lysyl oxidase-like protein LOXL2 promotes lung metastasis of breast cancer. Cancer Res. https://doi.org/10.1158/0008-5472.CAN-16-3152 (2017).
Eyre, D. R., Paz, M. A. & Gallop, P. M. Cross-linking in collagen and elastin. Annu. Rev. Biochem. 53, 717–748 (1984).
Lin, E. Y., Nguyen, A. V., Russell, R. G. & Pollard, J. W. Colony-stimulating factor 1 promotes progression of mammary tumors to malignancy. J. Exp. Med. 193, 727–739 (2001).
Strachan, D. C. et al. CSF1R inhibition delays cervical and mammary tumor growth in murine models by attenuating the turnover of tumor-associated macrophages and enhancing infiltration by CD8+ T cells. Oncoimmunology 2, e26968 (2013).
Lin, E. Y. et al. Progression to malignancy in the polyoma middle T oncoprotein mouse breast cancer model provides a reliable model for human diseases. Am. J. Pathol. 163, 2113–2126 (2003).
Novak, M. L. & Koh, T. J. Macrophage phenotypes during tissue repair. J. Leukoc. Biol. 93, 875–881 (2013).
Song, E. et al. Influence of alternatively and classically activated macrophages on fibrogenic activities of human fibroblasts. Cell Immunol. 204, 19–28 (2000).
Fichtner-Feigl, S., Strober, W., Kawakami, K., Puri, R. K. & Kitani, A. IL-13 signaling through the IL-13alpha2 receptor is involved in induction of TGF-beta1 production and fibrosis. Nat. Med. 12, 99–106 (2006).
Takaluoma, K., Lantto, J. & Myllyharju, J. Lysyl hydroxylase 2 is a specific telopeptide hydroxylase, while all three isoenzymes hydroxylate collagenous sequences. Matrix Biol. 26, 396–403 (2007).
Yau, C. et al. A multigene predictor of metastatic outcome in early stage hormone receptor-negative and triple-negative breast cancer. Breast Cancer Res. 12, R85 (2010).
Gyorffy, B. et al. An online survival analysis tool to rapidly assess the effect of 22,277 genes on breast cancer prognosis using microarray data of 1,809 patients. Breast Cancer Res. Treat. 123, 725–731 (2010).
Noy, R. & Pollard, J. W. Tumor-associated macrophages: from mechanisms to therapy. Immunity 41, 49–61 (2014).
Gilkes, D. M., Bajpai, S., Chaturvedi, P., Wirtz, D. & Semenza, G. L. Hypoxia-inducible factor 1 (HIF-1) promotes extracellular matrix remodeling under hypoxic conditions by inducing P4HA1, P4HA2, and PLOD2 expression in fibroblasts. J. Biol. Chem. 288, 10819–10829 (2013).
Wong, C. C. et al. Hypoxia-inducible factor 1 is a master regulator of breast cancer metastatic niche formation. Proc. Natl Acad. Sci. USA 108, 16369–16374 (2011).
Natarajan, S. et al. Collagen remodeling in the hypoxic tumor-mesothelial niche promotes ovarian cancer metastasis. Cancer Res. 79, 2271–2284 (2019).
Lin, E. Y. et al. Macrophages regulate the angiogenic switch in a mouse model of breast cancer. Cancer Res. 66, 11238–11246 (2006).
Ahn, Y., Sanderson, B. W., Klein, O. D. & Krumlauf, R. Inhibition of Wnt signaling by Wise (Sostdc1) and negative feedback from Shh controls tooth number and patterning. Development 137, 3221–3231 (2010).
Hsiao, E. C. et al. Osteoblast expression of an engineered Gs-coupled receptor dramatically increases bone mass. Proc. Natl Acad. Sci. USA 105, 1209–1214 (2008).
Guy, C. T., Cardiff, R. D. & Muller, W. J. Induction of mammary tumors by expression of polyomavirus middle T oncogene: a transgenic mouse model for metastatic disease. Mol. Cell Biol. 12, 954–961 (1992).
Avery, N. C., Sims, T. J. & Bailey, A. J. in Extracellular Matrix Protocols 2nd edn (eds Even-Ram, S. & Artym, V. V.) 103–121 (Humana, 2009).
Clasquin, M. F., Melamud, E. & Rabinowitz, J. D. LCMS data processing with maven: a metabolomic analysis and visualization engine. Curr. Protoc. Bioinform. 37, 14.11.11–14.11.23 (2012).
Ruffell, B. et al. Leukocyte composition of human breast cancer. Proc. Natl Acad. Sci. USA 109, 2796–2801 (2012).
Finak, G. et al. Gene expression signatures of morphologically normal breast tissue identify basal-like tumors. Breast Cancer Res. 8, R58 (2006).
Cerami, E. et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2, 401–404 (2012).
Gao, J. et al. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci. Signal 6, pl1 (2013).
Curtis, C. et al. The genomic and transcriptomic architecture of 2,000 breast tumours reveals novel subgroups. Nature 486, 346–352 (2012).
Berglund, G., Elmstahl, S., Janzon, L. & Larsson, S. A. The Malmo Diet and Cancer Study. Design and feasibility. J. Intern. Med. 233, 45–51 (1993).
Manjer, J. et al. The Malmo Diet and Cancer Study: representativity, cancer incidence and mortality in participants and non-participants. Eur. J. Cancer Prev. 10, 489–499 (2001).
Cohen, D. A. et al. Interobserver agreement among pathologists for semiquantitative hormone receptor scoring in breast carcinoma. Am. J. Clin. Pathol. 138, 796–802 (2012).
Acknowledgements
We thank J. Northcott for writing the ImageJ Macro, L. Korets for mouse husbandry and N. Korets for histology support, as well as K. Lövgren and S. Baker for LH2 immunostaining on patient biopsies. We thank the UCSF Genomics core for their support with the RNA-seq analysis on mouse TAMs and C. Stashko for generating the gene expression heat map. The work was supported by investigator grants through the US National Cancer Institute R33 CA183685 (K.C.H. and V.M.W) and R01CA192914 and CA174929 to (V.M.W), and R01CA222508-01 to (V.M.W. and E.S.H.), as well as US DOD Breast Cancer Research Program (BCRP) grant BC122990 (V.M.W.). Trainee support was provided by US DOD BCRP grant BC130501 (O.M.), US NIH grants TL1 TR001081 and US NIH T32 HL007171 (A.S.B.), and US NIH T32 grant CA 108462 (O.M.). Funding from Eastern Star Scholar-Minnesota Masonic Cancer Center (A.C.N.), the Swedish Research Council (S.B.) and US NIH R01 CA057621 (Z.W.) also supported the work.
Author information
Authors and Affiliations
Contributions
V.M.W., K.C.H., O.M., A.S.B. and A.P.D. conceived the project, prepared figures and wrote the manuscript. A.S.B., T.T.P., T.N. and K.C.H. developed the xAAA method, and A.S.B. and I.Z. performed all LC-MS and LC-PRM experiments. J.N.L. generated the TetO_mLox mouse model. O.M. designed and conducted in vivo experiments using inducible LOX overexpression models. B.R. and L.M.C. designed and conducted anti-CSF1 blocking antibody mouse experiment. O.M. and A.P.D. performed and quantified immunofluorescence, H&E, PS and SHG imaging and analyses on mouse tissue samples. I.A., A.P.D. and J.M.B. performed AFM on human or mouse tissue specimens. I.A. performed SHG imaging on human tissue. E.S.H., Y.-Y.C., and P.K. provided human breast tumour biopsies for xAAA. B.R. and L.C. designed and conducted immunoprofiling on human breast tumour via flow cytometry. O.M. and A.P.D. performed all gene expression analyses with the exception of Fig. 5g–j. T.G., H.K. and M.P. performed gene expression analyses gene expression in microdissected epithelial and stromal compartments of human invasive breast carcinomas. S.B. established and managed the MDCS cohort used for LH2 IHC. Z.W. and S.B. performed LH2 IHC. A.C.N. designed scoring schemes for stromal and neoplastic epithelial LH2 IHC, and A.C.N, A.N., J.G. and A.C. scored all human biopsies. S.B., O.B., A.C.N., O.M. and V.M.W. analysed and interpreted clinical data from LH2 scores. V.M.W., K.C.H., O.M., A.S.B. and A.P.D. wrote the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare the following competing interests: L.M.C. is a paid consultant for Cell Signaling Technologies, Shasqi Inc., and AbbVie Inc., and received reagent and/or research support from Plexxikon Inc., Pharmacyclics, Inc., Acerta Pharma, LLC, Deciphera Pharmaceuticals, LLC, Genentech, Inc., Roche Glycart AG, Syndax Pharmaceuticals Inc., Innate Pharma and NanoString Technologies, and is a member of the scientific advisory boards of Syndax Pharmaceuticals, Carisma Therapeutics, Zymeworks, Inc., Verseau Therapeutics, Cytomix Therapeutics, Inc. and Kineta Inc. T.N. and K.C.H. are co-founders of Omix Technologies.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Partial assignment of MS2 fragmentation spectra of crosslinked amino acid standards.
MS2 fragmentation spectra of commercially available crosslinked amino acid standards with partial assignment of MS2 fragmentation spectra (LNL, DHLNL, dPyr). Protonated forms of precursor ions are denoted by MH+ labels. Colored circles above fragment ions are matched to their suggested fragment ion structure above the full spectra or are listed above the ion if space permits.
Extended Data Fig. 2 Optimized crosslinking amino acid analysis (xAAA) enables accurate measurement of collagen state across a diverse range of tissue types.
a, xAAA workflow schematic. Clinical specimens are hydrolyzed and enriched by solid phase extraction (SPE). b, The enriched hydrolysate is analyzed by LC-SRM on a hybrid quadrupole orbitrap instrument. MS2 spectra is used to accurately identify crosslinked amino acids (xAAs) such as dihydroxy lysinonorleucine (DHLNL) (c) Bar graphs showing quantification of tissue collagen and (d) total xAA measured in human breast, lung, skin, bone, trachea and tendon (pooled n = 3 each tissue). The calculated amino acid crosslink abundancies are normalized to total tissue collagen content (that is hydroxyproline abundance) which is calculated based on dry tissue weight. The final values have been plotted as relative abundance based on peak area. Bar graphs depict relative abundance of Lysine derived-collagen crosslink (LCC) and hydroxylysine-derived collagen crosslink (HLCC) species.
Extended Data Fig. 3 Abundance of LOX-mediated crosslinks does not correlate with patient age.
a, Scatter plot showing the mean and s.e.m. of patient age for normal (n = 4) and tumor tissue (ER+ n = 8, HER2+ n = 6, TN n = 5) samples by subtype. b-f, Scatter plots showing the levels of each LCC and HLCC crosslink measured in normal and tumor breast tissues versus patient age. The total abundance of crosslinks (g) was calculated by summing all individual crosslinks and the total tissue HLCC abundance (h) was calculated by summing DHLNL, Pyr, and d-Pyr. All crosslink values are normalized to total collagen content (that is hydroxyproline abundance) and dry tissue weight and are plotted as log2 transformed normalized peak areas from LC-MS data. The best fit line and its slope and r2 value are displayed on each plot. All n values represent biologically independent human tissue specimens.
Extended Data Fig. 4 Validation of LOX overexpression in doxycycline (DOX) treated MMTV-rtTA+/−; TetO_mLox+/− mice.
a–c, Representative dot plots (n = 2) of GFP expression in mouse mammary epithelial cells analyzed by flow cytometry. GFP and mLox are encoded on the same mRNA transcript with independent translation initiation sites. a, GFP expression is induced in DOX-treated MMTV-rtTA+/−; TetO_mLox+/− mice. b, c, GFP expression is not induced in control mice lacking the MMTV-rtTA promoter (b) or control mice not treated with DOX (c). To select for mammary epithelial cells were gated out cells positive for anti-mouse CD45-APC, anti-mouse CD31-APC, anti-mouse Ter119-APC antibodies. d, Quantification of Lox gene expression in whole mouse mammary gland by RT-qPCR in control MMTV-rtTA−/−; TetO_mLox+/− mice (n = 2) and LOX overexpressing MMTV-rtTA+/−; TetO_mLox+/− (n = 2) mice. e, Western blot of whole tumor lysate from control (n = 4) and epithelial LOX overexpressing (n = 6) PyMT tumors. Scatter plots with mean ±s.e.m. quantify optical density of each band normalized to E-cadherin. Western blot for these lysates was run once. Statistical analyses were performed using two-tailed Mann-Whitney U test Pro-LOX **p = 0.0095, Cleaved LOX *p = 0.0381. All n values represent biologically independent mouse tissue specimens.
Extended Data Fig. 5 Design of inducible lysyl oxidase expression in vivo using a TetOFF system in the background of MMTV-PyMT model.
a, PCR results and sequence of PCR primers of N- and C-terminal ends to confirm TetO_mLox transgene incorporation. b, Diagram of inducible TetOFF system. TetO_mLox transgene is reversibly turned off or on in the presence or absence of doxycycline, respectively.
Extended Data Fig. 6 PyMT collagen crosslinking abundance does not increase between 8- and 11-week tumors.
a–f, Scatter plots showing individual and mean values ±s.e.m. of the levels of total collagen crosslinks (a), total HLCCs (b), LNL (c), HLNL (d), DHLNL (e), and Pyr (f) in 8-week (n = 10) and 11-week-old (n = 9) PyMT tumors. Quantity of crosslinks per tissue was calculated normalizing crosslinks to total collagen content (that is, hydroxyproline abundance) and dry tissue weight. Values were plotted as log2 transformed normalized peak areas as quantified from LC-MS data. Statistical analysis was performed using two-tailed unpaired t-test (a) p = 0.6149 (b) p = 0.7451 (c) p = 0.7235 (d) p = 0.1766 (e) p = 0.7563 (f) p = 0.4687. All n values represent biologically independent mouse tumor specimens.
Extended Data Figure 7 Early profibrotic macrophages express M2a markers and are lost upon anti-CSF1 treatment.
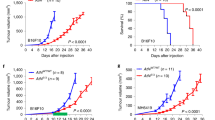
a, Scatter plot showing individual values and mean ±s.e.m. of the number of F4-80 positive cells per field of view in IgG1 treated (n = 6) or anti-CSF1 treated (n = 4) PyMT tumors from 8-week-old mice. b, c, Scatter plots showing individual values and mean ±s.e.m. of the percentage of F4/80 positive cells expressing markers MHCII and CD86 (b) and markers CD163 and RELMα (c) in IgG1 treated (n = 6) or anti-CSF1 treated (n = 4–5) PyMT tumors from 8-week-old mice. d–g, Scatter plots showing individual values and mean ±s.e.m. of the number of cells per field of view (FOV) of other non-macrophage immune populations CD11b+ F4-80− (d), MHCIIhi F4-80- (e), CD45+ CD4+ (f), and CD45+ CD8+ (g) in IgG1 (n = 4 e, g; n = 5 d, f) and anti-CSF1 (n = 6) treated mice. Statistical analysis (a-g) was performed using two-tailed Mann-Whitney U test (a) *p = 0.019 (b) MHCIIhi p = 0.7190, CD86+ p = 0.4286 (c) CD163+ *p = 0.0303, RELMα+ p = 0.0381 (d) p = 0.9719 (e) p = 0.1524 (f) p = 0.3074 (g) p = 1.000. h, Heat map of selected differentially expressed genes from macrophages sorted from 8-week and 11-week PyMT tumors. i, Representative images from 8-week-old IgG1 treated and anti-CSF1 treated PyMT mice stained for cytokeratin 8/18 (green), PDGFRα (magenta), vimentin (yellow), and DAPI (blue). Scale bar is 100 µm. j, Scatter plot showing mean ±s.e.m. of fibroblasts (vimentin+ and PDGFRα+) per field of view in 8-week-old IgG1 treated (n = 6) and anti-CSF1 (n = 5) treated PyMT mice. All n for (a-j) values represent biologically independent mouse tissue specimens. k–m, Scatter plot depicting the Spearman correlation of CD163 gene expression with LOX (k), PLOD2 (l), and LOXL2 (*p = 0.0143) (m) in human breast tumors (n = 1904 biologically independent human tissue specimens).
Extended Data Figure 8 ECM Stiffness for individual specimens.
a, Scatter plot showing mean and individual values of atomic force microscopy (AFM) microindentation measurements of individual human samples categorized as normal breast tissue (black circles) or invasive ductal carcinoma (IDC; red squares). b, Scatter plot showing mean and individual values of AFM microindentation measurements of individual mouse mammary tumors from doxycycline treated MMTV-PyMT+/−; Col1a1-tTA+/−; TetO-mLox control mice (DOX-ind PyMT Con; black circles) or water treated mice overexpressing stromal LOX (Water PyMT LOX OX; red squares). c, Scatter plot showing mean and individual values of AFM microindentation measurements of individual mouse mammary gland tumors from 8-week-old IgG1 treated (IgG Control; red squares) and anti-CSF1 treated (aCSF1 Ab; black circles) PyMT tumors.
Extended Data Figure 9 PLOD2 correlations with distant metastasis free survival and probability of relapse free survival.
a, Log2 scaled data graphs showing relative PLOD2 gene expression levels in patients stratified by estrogen and epidermal growth factor receptor two (ER/HER2) status (ER+ HER2- n = 314; ER- or + /HER2+ n = 73; ER-/HER2- n = 133). Whiskers denote minima and maxima, box denotes 25th and 75th percentiles and median. Statistical analysis was performed to compare PLOD2 expression levels among subtypes using Kruskal–Wallis one-way ANOVA (****p < 0.0001) and two-tailed Mann-Whitney U test without adjustment for multiple comparisons for individual comparisons (****p < 0.0001). b, Line graphs showing distant metastasis-free survival (DMFS) for patients with estrogen receptor positive and epidermal growth factor receptor two negative breast tumors (ER +/− HER2-; low n = 157 & high = 157). c, Line graph of DMFS for patients with estrogen receptor negative or negative and epidermal growth factor receptor two positive breast tumors (ER- or +/HER2+; low n = 36 & high = 37). d, Line graph of DMFS for patients with estrogen receptor negative epidermal growth factor receptor negative breast tumors (ER- HER2-; low n = 66 & high = 67). Statistical analyses were performed to compare PLOD2 and DMFS for each subtype or Log-rank (Mantel-Cox) test (b) p = 0.8051 (c) *p = 0.0144 (d) p = **0.0050. e-g, Kaplan-Meier curves indicating the probability of relapse-free survival assessed in ER+ /PR + (e), HER2+ (f), and triple negative (g) patients with breast cancer up to 16 years after diagnosis. A correlation between PLOD2 (gene encoding LH2) expression and relapse-free survival (RFS) has been determined using an online tool (http://kmplot.com/analysis/) as described in the methods. Top and bottom panels represent two distinct Affymetrix PLOD2 probes (202619 and 202620) from the same database. For Kaplan-Meier curves, statistical analyses were performed using LogRank test.
Extended Data Figure 10 Assessment of LOX and neoplastic epithelial LH2 expression in breast cancer.
a, Neoplastic epithelial LOX staining of the Biomax BC081116c TMA was assessed by a researcher with an intensity score and stratified as negative/low, moderate, or high. b, Stromal LOX staining of the Biomax BC081116c TMA was assessed by a researcher with an intensity score and stratified as negative/low, moderate, or high. Scale bar is 500 µm. c, Tumor samples from incident breast cancer cases were collected, and a tissue microarray (TMA) including two 1-mm cores from each tumor was constructed. Neoplastic epithelial LH2 staining was assessed by a pathologist with semi-quantitative intensity score and stratified as negative, low, or moderate/high. Scale bars are 500 µm. See Supplementary Table 2 for patient information. d, Clinical correlation of neoplastic epithelial LH2 intensity score with tumor grades. e, Kaplan-Meier curves indicating cumulative breast cancer specific survival (BCSS) based on epithelial LH2 intensity assessed in patients with breast cancer up to 10 years after diagnosis (LH2 negative n = 271, weak n = 112, moderate/high n = 77). f, BCSS curves by epithelial LH2 intensity including only axillary lymph node negative patients (LH2 negative n = 175, weak n = 67, moderate/high n = 50). g, BCSS curves by epithelial LH2 intensity including only axillary lymph node positive patients (LH2 negative n = 84, weak n = 42, moderate/high n = 26). For tumor grade and LH2 intensity score, statistical analysis was performed using a linear-by-linear association (***p < 0.001). For Kaplan-Meier curves, statistical analyses were using LogRank test (e) *p = 0.025 (f) *p = 0.038 (g) p = 0.165.
Supplementary information
Supplementary Information
Supplementary Tables 1–3 and Figs. 1–4.
Source data
Source Data Fig. 1
Plasmid map for TetO_mLox.
Source Data Fig. 2
Unprocessed western blots.
Rights and permissions
About this article
Cite this article
Maller, O., Drain, A.P., Barrett, A.S. et al. Tumour-associated macrophages drive stromal cell-dependent collagen crosslinking and stiffening to promote breast cancer aggression. Nat. Mater. 20, 548–559 (2021). https://doi.org/10.1038/s41563-020-00849-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-020-00849-5