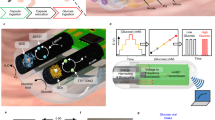
Abstract
A promising class of materials for applications that rely on electron transfer for signal generation are the n-type semiconducting polymers. Here we demonstrate the integration of an n-type conjugated polymer with a redox enzyme for the autonomous detection of glucose and power generation from bodily fluids. The reversible, mediator-free, miniaturized glucose sensor is an enzyme-coupled organic electrochemical transistor with a detection range of six orders of magnitude. This n-type polymer is also used as an anode and paired with a polymeric cathode in an enzymatic fuel cell to convert the chemical energy of glucose and oxygen into electrical power. The all-polymer biofuel cell shows a performance that scales with the glucose content in the solution and a stability that exceeds 30 days. Moreover, at physiologically relevant glucose concentrations and from fluids such as human saliva, it generates enough power to operate an organic electrochemical transistor, thus contributes to the technological advancement of self-powered micrometre-scale sensors and actuators that run on metabolites produced in the body.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data and materials availability
All the data needed to evaluate the conclusions in the paper are present in the paper and/or the Supplementary Materials. Additional data related to this paper may be requested from the authors.
References
Pappa, A.-M. et al. Organic electronics for point-of-care metabolite monitoring. Trends Biotechnol. 36, 45–59 (2018).
Gifford, R. Continuous glucose monitoring: 40 years, what we’ve learned and what’s next. ChemPhysChem 14, 2032–2044 (2013).
Witkowska Nery, E., Kundys, M., Jeleń, P. S. & Jönsson-Niedziółka, M. Electrochemical glucose sensing: is there still room for improvement? Anal. Chem. 88, 11271–11282 (2016).
Wang, J. Electrochemical glucose biosensors. Chem. Rev. 108, 814–825 (2008).
Heller, A. & Feldman, B. Electrochemical glucose sensors and their applications in diabetes management. Chem. Rev. 108, 2482–2505 (2008).
Wu, Y. & Hu, S. Biosensors based on direct electron transfer in redox proteins. Microchim. Acta 159, 1–17 (2007).
Zhang, W. & Li, G. Third-generation biosensors based on the direct electron transfer of proteins. Anal. Sci. 20, 603–609 (2004).
Zhu, C., Yang, G., Li, H., Du, D. & Lin, Y. Electrochemical sensors and biosensors based on nanomaterials and nanostructures. Anal. Chem. 87, 230–249 (2015).
Kissinger, P. T. Engineering the bioelectronic interface: applications to analyte biosensing and protein detection. J. Am. Chem. Soc. 132, 1444–1444 (2010).
Banica, F. G. Chemical Sensors and Biosensors: Fundamentals and Applications (Wiley, 2012).
Simon, D. T., Gabrielsson, E. O., Tybrandt, K. & Berggren, M. Organic bioelectronics: bridging the signaling gap between biology and technology. Chem. Rev. 116, 13009–13041 (2016).
Luz, R. A. S., Pereira, A. R., de Souza, J. C. P., Sales, F. C. P. F. & Crespilho, F. N. Enzyme biofuel cells: thermodynamics, kinetics and challenges in applicability. ChemElectroChem 1, 1751–1777 (2014).
Minteer, S. D., Atanassov, P., Luckarift, H. R. & Johnson, G. R. New materials for biological fuel cells. Mater. Today 15, 166–173 (2012).
Aghahosseini, H. et al. Glucose-based biofuel cells: nanotechnology as a vital science in biofuel cells performance. Nanochem. Res. 1, 183–204 (2016).
Kwon, C. H. et al. High-power hybrid biofuel cells using layer-by-layer assembled glucose oxidase-coated metallic cotton fibers. Nat. Commun. 9, 4479 (2018).
Wen, D. & Eychmüller, A. Enzymatic biofuel cells on porous nanostructures. Small 12, 4649–4661 (2016).
Giovannitti, A. et al. The role of the side chain on the performance of n-type conjugated polymers in aqueous electrolytes. Chem. Mater. 30, 2945–2953 (2018).
Inal, S., Rivnay, J., Suiu, A.-O., Malliaras, G. G. & McCulloch, I. Conjugated polymers in bioelectronics. Acc. Chem. Res. 51, 1368–1376 (2018).
Spyropoulos, G. D., Gelinas, J. N. & Khodagholy, D. Internal ion-gated organic electrochemical transistor: a building block for integrated bioelectronics. Sci. Adv. 5, eaau7378 (2019).
Pappa, A. M. et al. Direct metabolite detection with an n-type accumulation mode organic electrochemical transistor. Sci. Adv. 4, eaat0911 (2019).
Liao, C., Zhang, M., Niu, L., Zheng, Z. & Yan, F. Highly selective and sensitive glucose sensors based on organic electrochemical transistors with graphene-modified gate electrodes. J. Mater. Chem. B 1, 3820–3829 (2013).
Piro, B. et al. Fabrication and use of organic electrochemical transistors for sensing of metabolites in aqueous media. Appl. Sci. 8, 928 (2018).
Lineweaver, H. & Burk, D. The determination of enzyme dissociation constants. J. Am. Chem. Soc. 56, 658–666 (1934).
Trefz, D. et al. Electrochemical investigations of the n-type semiconducting polymer P(NDI2OD-T2) and its monomer: new insights in the reduction behavior. J. Phys. Chem. C 119, 22760–22771 (2015).
Wooten, M., Karra, S., Zhang, M. & Gorski, W. On the direct electron transfer, sensing, and enzyme activity in the glucose oxidase/carbon nanotubes system. Anal. Chem. 86, 752–757 (2014).
Zhang, Y. et al. Visualizing the solid–liquid interface of conjugated copolymer films using fluorescent liposomes. ACS Appl. Bio Mater. 1, 1348–1354 (2018).
Giovannitti, A. et al. N-type organic electrochemical transistors with stability in water. Nat. Commun. 7, 13066 (2016).
Giussani, E., Fazzi, D., Brambilla, L., Caironi, M. & Castiglioni, C. Molecular level investigation of the film structure of a high electron mobility copolymer via vibrational spectroscopy. Macromolecules 46, 2658–2670 (2013).
Navarrete, J. T. L. & Zerbi, G. Lattice dynamics and vibrational spectra of polythiophene. II. Effective coordinate theory, doping induced, and photoexcited spectra. J. Chem. Phys. 94, 965–970 (1991).
Mitraka, E. et al. Oxygen-induced doping on reduced PEDOT. J. Mater. Chem. A 5, 4404–4412 (2017).
Singh, S. K., Crispin, X. & Zozoulenko, I. V. Oxygen reduction reaction in conducting polymer PEDOT: density functional theory study. J. Phys. Chem. C 121, 12270–12277 (2017).
Winther-Jensen, B., Winther-Jensen, O., Forsyth, M. & MacFarlane, D. R. High rates of oxygen reduction over a vapor phase–polymerized PEDOT electrode. Science 321, 671–674 (2008).
Mitraka, E. et al. Electrocatalytic production of hydrogen peroxide with poly(3,4-ethylenedioxythiophene) electrodes. Adv. Sustain. Syst. 3, 1800110 (2019).
Eckermann, A. L., Feld, D. J., Shaw, J. A. & Meade, T. J. Electrochemistry of redox-active self-assembled monolayers. Coord. Chem. Rev. 254, 1769–1802 (2010).
Salimi, A., Sharifi, E., Noorbakhsh, A. & Soltanian, S. Immobilization of glucose oxidase on electrodeposited nickel oxide nanoparticles: direct electron transfer and electrocatalytic activity. Biosens. Bioelectron. 22, 3146–3153 (2007).
Laviron, E. General expression of the linear potential sweep voltammogram in the case of diffusionless electrochemical systems. J. Electroanal. Chem. Interfacial Electrochem. 101, 19–28 (1979).
Liu, J. et al. Enhancing molecular n-type doping of donor–acceptor copolymers by tailoring side chains. Adv. Mater. 30, 1704630 (2018).
Wang, S. et al. A chemically doped naphthalenediimide–bithiazole polymer for n-type organic thermoelectrics. Adv. Mater. 30, 1801898 (2018).
Cho, C. et al. Completely organic multilayer thin film with thermoelectric power factor rivaling inorganic tellurides. Adv. Mater. 27, 2996–3001 (2015).
Rivnay, J. et al. Unconventional face-on texture and exceptional in-plane order of a high mobility n-type polymer. Adv. Mater. 22, 4359–4363 (2010).
Miglbauer, E., Wójcik, P. J. & Głowacki, E. D. Single-compartment hydrogen peroxide fuel cells with poly(3,4-ethylenedioxythiophene) cathodes. Chem. Commun. 54, 11873–11876 (2018).
Savva, A. et al. Influence of water on the performance of organic electrochemical transistors. Chem. Mater. 31, 927–937 (2019).
Wustoni, S. & Savva, A. & Sun, R. & Bihar, E. & Inal, S. Enzyme-free detection of glucose with a hybrid conductive gel electrode. Adv. Mater. Interfaces 6, 1800928 (2019).
Savva, A. & Wustoni, S. & Inal. Ionic-to-electronic coupling efficiency in PEDOT:PSS films operated in aqueous electrolytes. J. Mater. Chem. C 6, 12023–12030 (2018).
Bihar, E. et al. A fully inkjet-printed disposable glucose sensor on paper. npj Flexible Electron. 2, 30 (2018).
Acknowledgements
The authors thank E. Bihar (KAUST) for her guidance during the experiments with saliva samples. The authors also thank J. Surgailis, J. I. de Oliveira Filho, L. B. Ospina and T. Cecilia-Hidalgo (KAUST) for spontaneous help. D.O., I.M. and S.I. gratefully acknowledge financial support from the KAUST Office of Sponsored Research (OSR) under Award no. OSR-2016-CRG5-3003. G.N. and S.I. acknowledge support from KAUST Sensors Initiative OSR Award no. REP/1/2719. Figures 1a and 4a were created by H. Hwang, scientific illustrator at KAUST.
Author information
Authors and Affiliations
Contributions
S.I. conceived the research, designed the experiments and supervised the work. D.O, G.N. and A.S performed the OECT and biofuel cell experiments. D.O. fabricated the devices and performed the UV–vis–near infrared studies. S.W. designed the electropolymerization experiments. X.C, I.P.M. and I.M. provided the n-type materials. P. M. F. J. C. and T.P. helped with the biofuel cell experiments. A.S. and D.O. performed the QCM-D experiments. A.G. performed and E.D.F supervised the Raman spectroscopy measurements. D.O. and G.N. wrote the manuscript with S.I. All the authors were involved in the discussion and participated in manuscript input.
Corresponding author
Ethics declarations
Competing interests
A US provisional application (no. 62/770934) related to this work was filed by S.I. and D.O.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Real-time response of the OECT channel current to varying concentrations of glucose in the measurement solution.
The concentration of glucose that is introduced to the solution increases incrementally from 10 nM to 100 μM, followed by a stepwise decrease back to 10 nM. In the end, glucose is washed away by a rinsing step. When the P-90 film is incubated with GOx, the enzyme adsorbs on P-90 without prior surface treatment because of the glycol-rich regions of the surface.
Extended Data Fig. 2 Real-time response of the OECT to successive amounts of H2O2.
H2O2 was added as molar equivalent of the glucose solutions used in the sensing experiments. Arrows indicate the addition of the H2O2 aliquots with concentrations of 10 nM, 50 nM, 100 nM, 500 nM, 1 µM, 5 µM, 10 µM, 25 µM, 50 µM, 100 µM, 250 µM, 500 µM, 750 µM, 1 mM, 2.5 mM, 5 mM, 7.5 mM, 10 mM, 12 mM, 15 mM and 20 mM. An increase in drain current is observed only with 20 mM of H2O2. Once the current stabilizes, the biosensor is rinsed with fresh buffer solution (‘Rinse’). The OECT shows a negligible response to H2O2 (only 1.3 nA increase) and requires long stabilization time, compared to changes induced by the enzymatic reaction. See Supplementary Fig. 5 for possible reactions that take place on P-90/GOx surface.
Extended Data Fig. 3 Spectroscopic investigations on P-90/GOx system.
(a) UV-VIS spectrum of a P-90/GOx film in PBS before and after the addition of glucose (1 mM). The film is subject to a constant doping potential at 0.5 V vs Ag/AgCl during the course of the experiments. (b) UV-VIS spectrum of a P-90/GOx film measured in PBS with and without the addition of 1 mM of H2O2. (c) Raman spectrum of a P-90 film subject to increasing doping potentials, from 0 to +1 V vs Ag/AgCl. After 1 V, the film was de-doped by applying 0 V vs Ag/AgCl. (d) Raman spectra of a P-90 film and P-90/GOx film. Inset shows the magnified spectral region between 1390 and 1730 cm−1 and the main peak attributions. In the spectrum, the region between 1100 and 1800 cm−1 refers to the resonant region of the backbone, while the low energy region ( < 1100 cm−1) is associated with the side-chains. See Supporting Information for further discussion.
Extended Data Fig. 4 Oxygen reduction reaction (ORR) activity of p(EDOT-co-EDOTOH).
(a) Chronoamperometry of the p(EDOT-co-EDOTOH) cathode under various atmospheres. The gases were introduced by bubbling the solution for 30 minutes. Afterwards, the gas tubing was held above the solution to shield it from the outside atmosphere. The measurement was performed at 0 V vs. VOC. (b) Cyclic voltammogram of p(EDOT-co-EDOTOH) cathode recorded in air (blue) and in O2 saturated buffer (red) under a rotating disk electrode. Scan rate is 10 mV.s−1 and rotation rate is 10 Hz (600 rpm). Arrow indicates the scan direction. (c) Reciprocal of current density of p(EDOT-co-EDOTOH) as a function of the rotation speed. The number of electrons (n) involved in the ORR is extracted from the slope of each curve and at a specific potential of the voltammogram. At low negative potentials (≤−0.375 V) the system follows a 4 e− direct reduction of oxygen to water. See Supporting Information for further discussion.
Extended Data Fig. 5 Electrochemical characterization of the biofuel cell.
(a) Extended cyclic voltammogram of the EFC comprising a P-90/GOx anode and a p(EDOT-co-EDOTOH) cathode, fuelled by 1 mM of glucose in PBS. Scan rate is 5 mV.s−1 and the arrow indicates the scan direction. The dotted lines depict the onset potential for the reduction and oxidation reactions. The difference between the onset potentials gives the theoretical open circuit voltage of the cell12. Inset shows the magnified region of the voltammogram until 0.6 V. (b) Half-cell open circuit potentials of the P-90/GOx anode and p(EDOT-co-EDOTOH) cathode in PBS. Glucose concentration is 1 mM. Triplicate experiments were performed for each sample while the open circuit stabilization time was ~2 hours.
Extended Data Fig. 6 Power output of the fuel cell when the anode is P-90 (no GOx) or P-90 functionalized with GOx (P-90/GOx).
All measurements were performed in PBS (pH 7.2) and in the absence of glucose. The EFC comprised a Nafion membrane.
Extended Data Fig. 7 The figure of merit for the membrane-free, all-polymer biofuel cell.
(a) Dependence of cell voltage (red circles) and power (green squares) on current density of the EFC fed with 10 mM of glucose solution. The measurements were acquired with an external resistor (1 kΩ - 10 MΩ). (b) OCV and MPD dependence on glucose concentration. (c) Comparison of the MPD of EFCs prepared with and without a Nafion membrane as a function of glucose concentration. The error bars show the standard deviation of the mean value of OCV and MPD values for three independent measurements.
Extended Data Fig. 8 The performance of a membrane-free EFC comprising an electrochemically doped P-90.
The P-90 film is functionalized with GOx and placed in PBS as the working electrode of a three-electrode system where a Pt coil is the counter electrode and Ag/AgCl is the reference electrode. The P-90 electrode is doped at –0.6 V vs Ag/AgCl and is also connected to the p(EDOT-co-EDOTOH) cathode. The power output of this EFC is markedly higher than the pristine EFC in the presence of 1 mM glucose, determined using an external resistor (1 kΩ - 10 MΩ).
Extended Data Fig. 9 Enzymatic biofuel cells power an accumulation mode OECT.
The VG and VD are supplied by the fuel cells with an output voltage of −0.15 V and −0.07 V, respectively. The OECT was switched ON and OFF by reversing the polarity of the gate voltage (by swapping the anode/cathode of the fuel cell connected to the gate electrode). The EFCs are fed with a constant concentration of aqueous glucose as fuel. The equivalent electrical circuit is presented on the right-hand side. The figure demonstrates the real-time changes in the source-drain current of a fully EFC powered accumulation mode OECT (based on a p-type organic semiconducting channel)42 as we reverse the polarity of the EFC biasing the gate electrode.
Supplementary information
Supplementary Information
Supplementary Tables 1–3, Figs. 1–15, Discussion and References.
Supplementary Video 1
Biofuel cell with a membrane supplies power to light up an LED.
Supplementary Video 2
Membrane-free biofuel cell supplies power to light up an LED.
Rights and permissions
About this article
Cite this article
Ohayon, D., Nikiforidis, G., Savva, A. et al. Biofuel powered glucose detection in bodily fluids with an n-type conjugated polymer. Nat. Mater. 19, 456–463 (2020). https://doi.org/10.1038/s41563-019-0556-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-019-0556-4
This article is cited by
-
Creation of a point-of-care therapeutics sensor using protein engineering, electrochemical sensing and electronic integration
Nature Communications (2024)
-
FeNi Prussian blue analogues on highly graphitized carbon nanosheets as efficient glucose sensors
Rare Metals (2024)
-
Metal oxide ion gated transistors based sensors
Science China Technological Sciences (2024)
-
Peculiar transient behaviors of organic electrochemical transistors governed by ion injection directionality
Nature Communications (2023)
-
A single n-type semiconducting polymer-based photo-electrochemical transistor
Nature Communications (2023)