Abstract
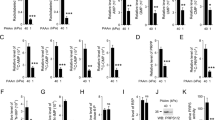
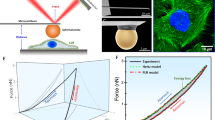
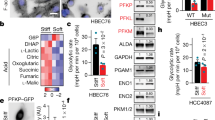
A common feature of cancer cells is the alteration of kinases and biochemical signalling pathways enabling transformed growth on soft matrices, whereas cytoskeletal protein alterations are thought to be a secondary issue. However, we report here that cancer cells from different tissues can be toggled between transformed and rigidity-dependent growth states by the absence or presence of mechanosensory modules, respectively. In various cancer lines from different tissues, cells had over tenfold fewer rigidity-sensing contractions compared with normal cells from the same tissues. Restoring normal levels of cytoskeletal proteins, including tropomyosins, restored rigidity sensing and rigidity-dependent growth. Further depletion of other rigidity sensor proteins, including myosin IIA, restored transformed growth and blocked sensing. In addition, restoration of rigidity sensing to cancer cells inhibited tumour formation and changed expression patterns. Thus, the depletion of rigidity-sensing modules through alterations in cytoskeletal protein levels enables cancer cell growth on soft surfaces, which is an enabling factor for cancer progression.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Data supporting the findings of this study are available within the article and from the corresponding author upon reasonable request;
References
Hamburger, A. W. & Salmon, S. E. Primary bioassay of human tumor stem cells. Science 197, 461–463 (1977).
Frisch, S. M. & Screaton, R. A. Anoikis mechanisms. Curr. Opin. Cell Biol. 13, 555–562 (2001).
Wolfenson, H. et al. Tropomyosin controls sarcomere-like contractions for rigidity sensing and suppressing growth on soft matrices. Nat. Cell Biol. 18, 33–42 (2016).
Weinberg, J. & Drubin, D. G. Clathrin-mediated endocytosis in budding yeast. Trends Cell Biol. 22, 1–13 (2012).
Ghassemi, S. et al. Cells test substrate rigidity by local contractions on submicrometer pillars. Proc. Natl Acad. Sci. USA 109, 5328–5333 (2012).
Yang, B. et al. Mechanosensing controlled directly by tyrosine kinases. Nano Lett. 16, 5951–5961 (2016).
Meacci, G. et al. α-Actinin links ECM rigidity sensing contractile units with periodic cell edge retractions. Mol. Biol. Cell 27, 3471–3479 (2016).
Saxena, M. et al. EGFR and HER2 activate rigidity sensing only on rigid matrices. Nat. Mater. 16, 775–781 (2017).
Fletcher, D. A. & Mullins, R. D. Cell mechanics and the cytoskeleton. Nature 463, 485–492 (2010).
Fife, C. M., McCarroll, J. A. & Kavallaris, M. Movers and shakers: cell cytoskeleton in cancer metastasis. Br. J. Pharmacol. 171, 5507–5523 (2014).
Schramek, D. et al. Direct in vivo RNAi screen unveils myosin IIa as a tumor suppressor of squamous cell carcinomas. Science 343, 309–313 (2014).
Conti, M. A. et al. Conditional deletion of nonmuscle myosin II-A in mouse tongue epithelium results in squamous cell carcinoma. Sci. Rep. 5, 14068 (2015).
Raval, G. N. et al. Loss of expression of tropomyosin-1, a novel class II tumor suppressor that induces anoikis, in primary breast tumors. Oncogene 22, 6194–6203 (2003).
Stehn, J. R. et al. A novel class of anticancer compounds targets the actin cytoskeleton in tumor cells. Cancer Res. 73, 5169–5182 (2013).
Menez, J. et al. Mutant alpha-actinin-4 promotes tumorigenicity and regulates cell motility of a human lung carcinoma. Oncogene 23, 2630–2639 (2004).
Nikolopoulos, S. N. et al. The human non-muscle alpha-actinin protein encoded by the ACTN4 gene suppresses tumorigenicity of human neuroblastoma cells. Oncogene 19, 380–386 (2000).
An, H. T., Yoo, S. & Ko, J. Alpha-actinin-4 induces the epithelial-to-mesenchymal transition and tumorigenesis via regulation of snail expression and beta-catenin stabilization in cervical cancer. Oncogene 35, 5893–5904 (2016).
Bharadwaj, S., Thanawala, R., Bon, G., Falcioni, R. & Prasad, G. L. Resensitization of breast cancer cells to anoikis by tropomyosin-1: role of Rho kinase-dependent cytoskeleton and adhesion. Oncogene 24, 8291–8303 (2005).
Zhu, S. et al. MicroRNA-21 targets tumor suppressor genes in invasion and metastasis. Cell Res. 18, 350–359 (2008).
Rasheed, S., Nelson-Rees, W. A., Toth, E. M., Arnstein, P. & Gardner, M. B. Characterization of a newly derived human sarcoma cell line (HT-1080). Cancer 33, 1027–1033 (1974).
Freedman, V. H. & Shin, S. I. Cellular tumorigenicity in nude mice: correlation with cell growth in semi-solid medium. Cell 3, 355–359 (1974).
Hollestelle, A., Elstrodt, F., Nagel, J. H., Kallemeijn, W. W. & Schutte, M. Phosphatidylinositol-3-OH kinase or RAS pathway mutations in human breast cancer cell lines. Mol. Cancer Res. 5, 195–201 (2007).
Fogh, J., Fogh, J. M. & Orfeo, T. One hundred and twenty-seven cultured human tumor cell lines producing tumors in nude mice. J. Natl Cancer Inst. 59, 221–226 (1977).
Mayo, J. G. Biologic characterization of the subcutaneously implanted lewis lung tumor. Cancer Chemother. Rep. 2 3, 325–330 (1972).
Kozlowski, J. M., Hart, I. R., Fidler, I. J. & Hanna, N. A human melanoma line heterogeneous with respect to metastatic capacity in athymic nude mice. J. Natl Cancer Inst. 72, 913–917 (1984).
Fabricant, R. N., De Larco, J. E. & Todaro, G. J. Nerve growth factor receptors on human melanoma cells in culture. Proc. Natl Acad. Sci. USA 74, 565–569 (1977).
Lieber, M., Mazzetta, J., Nelson-Rees, W., Kaplan, M. & Todaro, G. Establishment of a continuous tumor-cell line (panc-1) from a human carcinoma of the exocrine pancreas. Int. J. Cancer 15, 741–747 (1975).
Bigner, D. D. et al. Heterogeneity of genotypic and phenotypic characteristics of fifteen permanent cell lines derived from human gliomas. J. Neuropathol. Exp. Neurol. 40, 201–229 (1981).
JoveR. & HanafusaH. Cell tTransformation by the vViral src. Oncogene. 3, 31–56 (1987).
Prager-Khoutorsky, M. et al. Fibroblast polarization is a matrix-rigidity-dependent process controlled by focal adhesion mechanosensing. Nat. Cell Biol. 13, 1457–1465 (2011).
Shutova, M. S. et al. Self-sorting of nonmuscle myosins IIA and IIB polarizes the cytoskeleton and modulates cell motility. J. Cell Biol. 216, 2877–2889 (2017).
Gateva, G. et al. Tropomyosin isoforms specify functionally distinct actin filament populations in vitro. Curr. Biol. 27, 705–713 (2017).
Currier, M. A. et al. Identification of cancer-targeted tropomyosin inhibitors and their synergy with microtubule drugs. Mol. Cancer Ther. 16, 1555–1565 (2017).
Helfman, D. M., Flynn, P., Khan, P. & Saeed, A. Tropomyosin as a regulator of cancer cell transformation. Adv. Exp. Med. Biol. 644, 124–131 (2008).
Antony, J. et al. The GAS6-AXL signaling network is a mesenchymal (Mes) molecular subtype–specific therapeutic target for ovarian cancer. Sci. Signal. 9, ra97–ra97 (2016).
Yamamoto, T., Taya, S. & Kaibuchi, K. Ras-induced transformation and signaling pathway. J. Biochem. 126, 799–803 (1999).
Scholl, C. et al. Synthetic lethal interaction between oncogenic KRAS dependency and STK33 suppression in human cancer cells. Cell 137, 821–834 (2009).
Frezzetti, D. et al. Upregulation of miR-21 by ras in vivo and its role in tumor growth. Oncogene 30, 275–286 (2011).
Coombes, J. D. et al. Ras transformation overrides a proliferation defect induced by Tpm3.1 knockout. Cell. Mol. Biol. Lett. 20, 626–646 (2015).
Jiang, G., Huang, A. H., Cai, Y., Tanase, M. & Sheetz, M. P. Rigidity sensing at the leading edge through alphavbeta3 integrins and RPTPalpha. Biophys. J. 90, 1804–1809 (2006).
Sawada, Y. et al. Force sensing by mechanical extension of the Src family kinase substrate p130Cas. Cell 127, 1015–1026 (2006).
Kwon, T. et al. DANGER is involved in high glucose-induced radioresistance through inhibiting DAPK-mediated anoikis in non-small cell lung cancer. Oncotarget 7, 7193–7206 (2016).
Qin, R., Wolfenson, H., Saxena, M. & Sheetz, M. Tumor suppressor DAPK1 catalyzes adhesion assembly on rigid but anoikis on soft matrices. Preprint at https://www.biorxiv.org/content/10.1101/320739v2 (2018).
Yang, Y., Nguyen, E., Mege, R.-M., Ladoux, B. & Sheetz, M. P. Local contractions test rigidity of E-cadherin adhesions. Preprint at https://www.biorxiv.org/content/10.1101/318642v1 (2018).
Mierke, C. T., Frey, B., Fellner, M., Herrmann, M. & Fabry, B. Integrin α5β1 facilitates cancer cell invasion through enhanced contractile forces. J. Cell Sci. 124, 369–383 (2011).
Cui, Y. et al. Cyclic stretching of soft substrates induces spreading and growth. Nat. Commun. 6, 6333 (2015).
Nakazawa, N., Sathe, A. R., Shivashankar, G. V. & Sheetz, M. P. Matrix mechanics controls FHL2 movement to the nucleus to activate p21 expression. Proc. Natl Acad. Sci. USA 113, E6813–e6822 (2016).
Li, C., Huang, Z. & Wang, R. K. Elastic properties of soft tissue-mimicking phantoms assessed by combined use of laser ultrasonics and low coherence interferometry. Opt. Express 19, 10153–10163 (2011).
Mortazavi, A., Williams, B. A., McCue, K., Schaeffer, L. & Wold, B. Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat. Methods 5, 621 (2008).
Rafiq, NishaBteMohd et al. A mechano-signalling network linking microtubules, myosin IIA filaments and integrin-based adhesions. Nat. Mater. 18, 638 (2019).
Acknowledgements
The authors thank J.P. Thiery, P. Gunning, N.C. Gauthier and J. Kadrmas for critical reading of the manuscript. We thank D. Lim Gkeok Stzuan (NUHS) and A. Koh Pei Fern (CSI) for their kind help with H&E staining. We thank the members of Sheetz and Bershadsky laboratories for their kind help. This research was supported by funding to the Mechanobiology Institute, National University of Singapore. B.Y. was supported by the NUS grant ‘Activation of Apotosis by Soft Surfaces’ (no. R-714-000-112-133). H.W. is a David and Inez Myers Career Advancement Chair in Life Sciences fellow. M.P.S. is supported by NIH and NUS grants and the Mechanobiology Institute, National University of Singapore.
Author information
Authors and Affiliations
Contributions
B.Y. and M.P.S. conceived the study and designed the experiments. B.Y. and N.N. performed the experiments. V.Y.C. and R.Y.-J.H performed the CAM experiments and analysed data. S.L. wrote Matlab codes for data analysis. J.H. provided fabrication moulds. B.Y., H.W. and M.P.S wrote and prepared the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–10 and Tables 1–6.
Rights and permissions
About this article
Cite this article
Yang, B., Wolfenson, H., Chung, V.Y. et al. Stopping transformed cancer cell growth by rigidity sensing. Nat. Mater. 19, 239–250 (2020). https://doi.org/10.1038/s41563-019-0507-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-019-0507-0
This article is cited by
-
Biophysical control of plasticity and patterning in regeneration and cancer
Cellular and Molecular Life Sciences (2024)
-
Membrane to cortex attachment determines different mechanical phenotypes in LGR5+ and LGR5- colorectal cancer cells
Nature Communications (2024)
-
Hydrogel-based molecular tension fluorescence microscopy for investigating receptor-mediated rigidity sensing
Nature Methods (2023)
-
Impairment of rigidity sensing caused by mutant TP53 gain of function in osteosarcoma
Bone Research (2023)
-
Biophysics in tumor growth and progression: from single mechano-sensitive molecules to mechanomedicine
Oncogene (2023)