Abstract
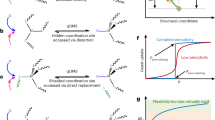
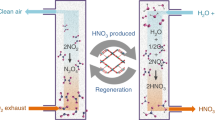
Emissions of SO2 from flue gas and marine transport have detrimental impacts on the environment and human health, but SO2 is also an important industrial feedstock if it can be recovered, stored and transported efficiently. Here we report the exceptional adsorption and separation of SO2 in a porous material, [Cu2(L)] (H4L = 4′,4‴-(pyridine-3,5-diyl)bis([1,1′-biphenyl]-3,5-dicarboxylic acid)), MFM-170. MFM-170 exhibits fully reversible SO2 uptake of 17.5 mmol g−1 at 298 K and 1.0 bar, and the SO2 binding domains for trapped molecules within MFM-170 have been determined. We report the reversible coordination of SO2 to open Cu(ii) sites, which contributes to excellent adsorption thermodynamics and selectivities for SO2 binding and facile regeneration of MFM-170 after desorption. MFM-170 is stable to water, acid and base and shows great promise for the dynamic separation of SO2 from simulated flue gas mixtures, as confirmed by breakthrough experiments.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Results of the refinements of the solvated, evacuated and SO2-loaded crystal structures of MFM-170 have been deposited as CIF files with CCDC numbers 1538125–1538126, 1538129 and 1853512–1853514. These data can be obtained free of charge from https://www.ccdc.cam.ac.uk/structures/.
References
Rezaei, F. et al. SOx/NOx removal from flue gas streams by solid adsorbents: a review of current challenges and future directions. Energy Fuels 29, 5467–5486 (2015).
Gao, J. et al. Pilot-scale experimental study on the CO2 capture process with existing of SO2: degradation, reaction rate, and mass transfer. Energy Fuels 25, 5802–5809 (2011).
Ding, S. et al. Significant promotion effect of Mo additive on a novel Ce–Zr mixed oxide catalyst for the selective catalytic reduction of NOx with NH3. ACS Appl. Mater. Interf. 7, 9497–9506 (2015).
Kinnunen, N. M. et al. Case study of a modern lean-burn methane combustion catalyst for automotive applications: what are the deactivation and regeneration mechanisms? Appl. Catal. B Environ. 207, 114–119 (2017).
Han, X., Yang, S. & Schröder, M. Porous metal–organic frameworks as emerging sorbents for clean air. Nat. Rev. Chem. 3, 108–118 (2019).
Raymundo-Piñero, E. et al. Factors controling the SO2 removal by porous carbons: relevance of the SO2 oxidation step. Carbon 38, 335–344 (2000).
Mathieu, Y. et al. Adsorption of SOx by oxide materials: a review. Fuel Process. Technol. 114, 81–100 (2013).
Kohl, A. L. & Nielsen, R. Gas Purification (Gulf Professional, 1997).
Nabais, A. R. et al. CO2/N2 gas separation using Fe(BTC)-based mixed matrix membranes: a view on the adsorptive and filler properties of metal-organic frameworks. Sep. Purif. Technol. 202, 174–184 (2018).
Peng, J. et al. Efficient kinetic separation of propene and propane using two microporous metal organic frameworks. Chem. Commun. 53, 9332–9335 (2017).
Chen, D.-M. et al. Tunable robust pacs-MOFs: a platform for systematic enhancement of the C2H2 uptake and C2H2/C2H4 separation performance. Inorg. Chem. 57, 2883–2889 (2018).
Zhong, R. et al. A solvent ‘squeezing’ strategy to graft ethylenediamine on Cu3(BTC)2 for highly efficient CO2/CO separation. Chem. Eng. Sci. 184, 85–92 (2018).
Zhang, Z. et al. MOFs for CO2 capture and separation from flue gas mixtures: the effect of multifunctional sites on their adsorption capacity and selectivity. Chem. Commun. 49, 653–661 (2013).
Peralta, D. et al. Comparison of the behavior of metal–organic frameworks and zeolites for hydrocarbon separations. J. Am. Chem. Soc. 134, 8115–8126 (2012).
Carter, J. H. et al. Exceptional adsorption and binding of sulfur dioxide in a robust zirconium-based metal–organic framework. J. Am. Chem. Soc. 140, 15564–15567 (2018).
Cui, X. et al. Ultrahigh and selective SO2 uptake in inorganic anion-pillared hybrid porous materials. Adv. Mater. 29, 1606929 (2017).
Glomb, S. et al. Metal–organic frameworks with internal urea-functionalized dicarboxylate linkers for SO2 and NH3 adsorption. ACS Appl. Mater. Inter. 9, 37419–37434 (2017).
Yang, S. et al. Irreversible network transformation in a dynamic porous host catalyzed by sulfur dioxide. J. Am. Chem. Soc. 135, 4954–4957 (2013).
Tan, K. et al. Mechanism of preferential adsorption of SO2 into two microporous paddle wheel frameworks M(bdc)(ted)0.5. Chem. Mater. 25, 4653–4662 (2013).
Savage, M. et al. Selective adsorption of sulfur dioxide in a robust metal-organic framework material. Adv. Mater. 28, 8705–8711 (2016).
Li, L. et al. Post-synthetic modulation of the charge distribution in a metal–organic framework for optimal binding of carbon dioxide and sulfur dioxide. Chem. Sci. 10, 1472–1482 (2019).
Lee, G.-Y. et al. Amine-functionalized covalent organic framework for efficient SO2 capture with high reversibility. Sci. Rep. 7, 557 (2017).
Thallapally, P. K. et al. Prussian blue analogues for CO2 and SO2 capture and separation applications. Inorg. Chem. 49, 4909–4915 (2010).
Fernandez, C. et al. Gas-induced expansion and contraction of a fluorinated metal−organic framework. Cryst. Growth Des. 10, 1037–1039 (2010).
Tchalala, M. R. et al. Fluorinated MOF platform for selective removal and sensing of SO2 from flue gas and air. Nat. Commun. 10, 1328 (2019).
Riboldi, L. & Bolland, O. Overview on pressure swing adsorption (PSA) as CO2 capture technology: state-of-the-art, limits and potentials. Energy Proc. 114, 2390–2400 (2017).
Riboldi, L. & Bolland, O. Evaluating pressure swing adsorption as a CO2 separation technique in coal-fired power plants. Int. J. Greenh. Gas. Control 39, 1–16 (2015).
Britt, D. et al. Highly efficient separation of carbon dioxide by a metal-organic framework replete with open metal sites. Proc. Natl. Acad. Sci. USA 106, 20637–20640 (2009).
Wong-Foy, A. G., Matzger, A. J. & Yaghi, O. M. Exceptional H2 saturation uptake in microporous metal-organic frameworks. J. Am. Chem. Soc. 128, 3494–3495 (2006).
Caskey, S. R., Wong-Foy, A. G. & Matzger, A. J. Dramatic tuning of carbon dioxide uptake via metal substitution in a coordination polymer with cylindrical pores. J. Am. Chem. Soc. 130, 10870–10871 (2008).
Britt, D., Tranchemontagne, D. & Yaghi, O. M. Metal-organic frameworks with high capacity and selectivity for harmful gases. Proc. Natl. Acad. Sci. USA 105, 11623–11627 (2008).
Guillerm, V. et al. A supermolecular building approach for the design and construction of metal–organic frameworks. Chem. Soc. Rev. 43, 6141–6172 (2014).
Park, J. et al. A versatile metal–organic framework for carbon dioxide capture and cooperative catalysis. Chem. Commun. 48, 9995–9997 (2012).
Lu, Z. et al. The utilization of amide groups to expand and functionalize metal-organic frameworks simultaneously. Chem. A Eur. J. 22, 6277–6285 (2016).
Branton, P. J., Hall, P. G., Treguer, M. & Sing, K. S. W. Adsorption of carbon dioxide, sulfur dioxide and water vapour by MCM-41, a model mesoporous adsorbent. J. Chem. Soc. Faraday Trans. 91, 2041–2043 (1995).
Tan, K. et al. Interaction of acid gases SO2 and NO2 with coordinatively unsaturated metal organic frameworks: MOF-74 (M = Zn, Mg, Ni, Co). Chem. Mater. 29, 4227–4235 (2017).
Goodman, A. L., Li, P., Usher, C. R. & Grassian, V. H. Heterogeneous uptake of sulfur dioxide on aluminum and magnesium oxide particles. J. Phys. Chem. A 105, 6109–6120 (2001).
Schneider, W. F., Li, J. & Hass, K. C. Combined computational and experimental investigation of SOx adsorption on MgO. J. Phys. Chem. B 105, 6972–6979 (2001).
Marinho, M. V. et al. Synthesis, crystal structure, and spectroscopic characterization of trans-bis[(μ-1,3-bis(4-pyridyl)propane)(μ-(3-thiopheneacetate-O))(3-thiopheneacetate-O)]dicopper(II), {[Cu2(O2CCH2C4H3S)4 μ-(BPP)2]}n: from a dinuclear paddle-wheel copper(II) unit to a 2-D coordination polymer involving monatomic carboxylate bridges. Inorg. Chem. 43, 1539–1544 (2004).
Acknowledgements
We thank EPSRC (EP/I011870), ERC (AdG 742041), the Royal Society and University of Manchester for funding. We are especially grateful to Diamond Light Source, Advanced Light Source, Oak Ridge National Laboratory and STFC/ISIS Neutron Facility for access to the beamlines B22/I11, 11.3.1, VISION and TOSCA, respectively. We thank M. Kibble for help at TOSCA beamline. The computing resources were made available through the VirtuES and the ICE-MAN projects, funded by the Laboratory Directed Research and Development programme at ORNL. This research used resources of the Advanced Light Source, which is a US Department of Energy Office of Science User Facility under contract no. DE-AC02-05CH11231. J.L. and X.Z. thank the China Scholarship Council (CSC) for funding.
Author information
Authors and Affiliations
Contributions
G.L.S. and J.E.E. performed the synthesis and characterization of MOF samples and measurements of adsorption isotherms. G.L.S. and X.H. performed measurements and the analysis of the breakthrough data. G.L.S., X.Z., S.P.A., L.J.M., S.J.T. and S.Y. collected and analysed the synchrotron single-crystal X-ray diffraction data. G.L.S., H.G.W.G., Y.C., S.R. and A.J.R.-C. collected and analysed the neutron scattering data. G.L.S., S.J.D. and C.C.T. collected and analysed the long-duration synchrotron X-ray diffraction data. G.L.S., J.L., N.M.J., M.D.F., G.C. and T.L.E. collected and analysed the synchrotron IR data. T.L.E. supervised the laboratory work of J.E.E. S.Y. and M.S. led the overall design and direction of the project. G.L.S., S.Y. and M.S. prepared the manuscript with help from all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–29, Figs. 1–26 and refs. 1–12
Supplementary Data
Compressed archive file that contains six crystallographic information files corresponding to the crystal structures of MFM-170·H2O·solv (CCDC no. 1538125), MFM-170·H2O (CCDC no. 1538126), MFM-170·H2O·3.27SO2 (CCDC no. 1538129), MFM-170 (CCDC no. 1853512), MFM-170·5.46SO2 (CCDC no. 1853513) and MFM-170·0.09SO2 (CCDC no. 1853514)
Rights and permissions
About this article
Cite this article
Smith, G.L., Eyley, J.E., Han, X. et al. Reversible coordinative binding and separation of sulfur dioxide in a robust metal–organic framework with open copper sites. Nat. Mater. 18, 1358–1365 (2019). https://doi.org/10.1038/s41563-019-0495-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-019-0495-0
This article is cited by
-
Quasi-open Cu(i) sites for efficient CO separation with high O2/H2O tolerance
Nature Materials (2024)
-
Preparation of UiO-66-type adsorbents for the separation of SO2 from flue gas
Adsorption (2024)
-
Temperature-dependent rearrangement of gas molecules in ultramicroporous materials for tunable adsorption of CO2 and C2H2
Nature Communications (2023)
-
Silver nanoparticle enhanced metal-organic matrix with interface-engineering for efficient photocatalytic hydrogen evolution
Nature Communications (2023)
-
Chelating Metal Ions in a Metal-Organic Framework for Constructing a Biomimetic Catalyst Through Post-modification
Chemical Research in Chinese Universities (2022)