Abstract
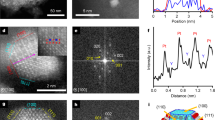
Subnanometric metal species (single atoms and clusters) have been demonstrated to be unique compared with their nanoparticulate counterparts. However, the poor stabilization of subnanometric metal species towards sintering at high temperature (>500 °C) under oxidative or reductive reaction conditions limits their catalytic application. Zeolites can serve as an ideal support to stabilize subnanometric metal catalysts, but it is challenging to localize subnanometric metal species on specific sites and modulate their reactivity. We have achieved a very high preference for localization of highly stable subnanometric Pt and PtSn clusters in the sinusoidal channels of purely siliceous MFI zeolite, as revealed by atomically resolved electron microscopy combining high-angle annular dark-field and integrated differential phase contrast imaging techniques. These catalysts show very high stability, selectivity and activity for the industrially important dehydrogenation of propane to form propylene. This stabilization strategy could be extended to other crystalline porous materials.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All the data reported in this paper are available from the corresponding author on request.
References
Liu, L. & Corma, A. Metal catalysts for heterogeneous catalysis: from single atoms to nanoclusters and nanoparticles. Chem. Rev. 118, 4981–5079 (2018).
Gallego, E. M. et al. “Ab initio” synthesis of zeolites for preestablished catalytic reactions. Science 355, 1051–1054 (2017).
Kosinov, N., Liu, C., Hensen, E. J. M. & Pidko, E. A. Engineering of transition metal catalysts confined in zeolites. Chem. Mater. 30, 3177–3198 (2018).
Ortalan, V., Uzun, A., Gates, B. C. & Browning, N. D. Direct imaging of single metal atoms and clusters in the pores of dealuminated HY zeolite. Nat. Nanotechnol. 5, 506–510 (2010).
Li, C. et al. Selective introduction of acid sites in different confined positions in ZSM-5 and its catalytic implications. ACS Catal. 8, 7688–7697 (2018).
Knott, B. C. et al. Consideration of the aluminum distribution in zeolites in theoretical and experimental catalysis research. ACS Catal. 8, 770–784 (2017).
Yokoi, T., Mochizuki, H., Namba, S., Kondo, J. N. & Tatsumi, T. Control of the Al distribution in the framework of ZSM-5 zeolite and its evaluation by solid-state NMR technique and catalytic properties. J. Phys. Chem. C 119, 15303–15315 (2015).
Goel, S., Zones, S. I. & Iglesia, E. Encapsulation of metal clusters within MFI via interzeolite transformations and direct hydrothermal syntheses and catalytic consequences of their confinement. J. Am. Chem. Soc. 136, 15280–15290 (2014).
Wang, N. et al. In situ confinement of ultrasmall Pd clusters within nanosized silicalite-1 zeolite for highly efficient catalysis of hydrogen generation. J. Am. Chem. Soc. 138, 7484–7487 (2016).
Iida, T., Zanchet, D., Ohara, K., Wakihara, T. & Roman-Leshkov, Y. Concerted bimetallic nanocluster synthesis and encapsulation via induced zeolite framework demetallation for shape and substrate selective heterogeneous catalysis. Angew. Chem. Int. Ed. 57, 6454–6458 (2018).
Zhang, J. et al. Sinter-resistant metal nanoparticle catalysts achieved by immobilization within zeolite crystals via seed-directed growth. Nat. Catal. 1, 540–546 (2018).
Campbell, C. T., Parker, S. C. & Starr, D. E. The effect of size-dependent nanoparticle energetics on catalyst sintering. Science 298, 811–814 (2002).
Flytzani-Stephanopoulos, M. & Gates, B. C. Atomically dispersed supported metal catalysts. Annu. Rev. Chem. Biomol. Eng. 3, 545–574 (2012).
Liu, L. et al. Generation of subnanometric platinum with high stability during transformation of a 2D zeolite into 3D. Nat. Mater. 16, 132–138 (2017).
Liu, L. et al. Evolution and stabilization of subnanometric metal species in confined space by in situ TEM. Nat. Commun. 9, 574 (2018).
Xiong, H. et al. Thermally stable and regenerable platinum-tin clusters for propane dehydrogenation prepared by atom trapping on ceria. Angew. Chem. Int. Ed. 56, 8986–8991 (2017).
Sattler, J. J., Ruiz-Martinez, J., Santillan-Jimenez, E. & Weckhuysen, B. M. Catalytic dehydrogenation of light alkanes on metals and metal oxides. Chem. Rev. 114, 10613–10653 (2014).
Zhu, J. et al. Size-dependent reaction mechanism and kinetics for propane dehydrogenation over Pt catalysts. ACS Catal. 5, 6310–6319 (2015).
Yang, M. et al. A common single-site Pt(II)–O(OH)x– species stabilized by sodium on “active” and “inert” supports catalyzes the water-gas shift reaction. J. Am. Chem. Soc. 137, 3470–3473 (2015).
Yang, M. et al. Catalytically active Au-O(OH)x- species stabilized by alkali ions on zeolites and mesoporous oxides. Science 346, 1498–1501 (2014).
Zhai, Y. et al. Alkali-stabilized Pt-OHx species catalyze low-temperature water-gas shift reactions. Science 329, 1633–1636 (2010).
Lazic, I., Bosch, E. G. T. & Lazar, S. Phase contrast STEM for thin samples: integrated differential phase contrast. Ultramicroscopy 160, 265–280 (2016).
Yucelen, E., Lazic, I. & Bosch, E. G. T. Phase contrast scanning transmission electron microscopy imaging of light and heavy atoms at the limit of contrast and resolution. Sci. Rep. 8, 2676 (2018).
Van Koningsveld, H. On the location and disorder of the tetrapropylammonium (TPA) ion in zeolite ZSM‐5 with improved framework accuracy. Acta Crystallogr. B 43, 127–132 (1987).
Dib, E., Grand, J., Mintova, S. & Fernandez, C. Structure-directing agent governs the location of silanol defects in zeolites. Chem. Mater. 27, 7577–7579 (2015).
Denayer, J. F., De Meyer, K., Martens, J. A. & Baron, G. V. Molecular competition effects in liquid-phase adsorption of long-chain n-alkane mixtures in ZSM-5 zeolite pores. Angew. Chem. Int. Ed. 42, 2774–2777 (2003).
Grand, J. et al. One-pot synthesis of silanol-free nanosized MFI zeolite. Nat. Mater. 16, 1010–1015 (2017).
de Graaf, J., van Dillen, A. J., de Jong, K. P. & Koningsberger, D. C. Preparation of highly dispersed Pt particles in zeolite Y with a narrow particle size distribution: characterization by hydrogen chemisorption, TEM, EXAFS spectroscopy, and particle modeling. J. Catal. 203, 307–321 (2001).
Bare, S. R. et al. Uniform catalytic site in Sn-beta-zeolite determined using X-ray absorption fine structure. J. Am. Chem. Soc. 127, 12924–12932 (2005).
Hammond, C. et al. Identification of active and spectator Sn sites in Sn-beta following solid-state stannation, and consequences for Lewis acid catalysis. ChemCatChem 7, 3322–3331 (2015).
Stakheev, A. Y., Shpiro, E. S., Jaeger, N. I. & Schulz-Ekloff, G. Electronic state and location of Pt metal clusters in KL zeolite: FTIR study of CO chemisorption. Catal. Lett. 32, 147–158 (1995).
Huang, H. et al. Effects of heat treatment atmosphere on the structure and activity of Pt3Sn nanoparticle electrocatalysts: a characterization case study. Faraday Discuss. 208, 555–573 (2018).
Alexeev, O. S. & Gates, B. C. Supported bimetallic cluster catalysts. Ind. Eng. Chem. Res. 42, 1571–1587 (2003).
Sankar, M. et al. Designing bimetallic catalysts for a green and sustainable future. Chem. Soc. Rev. 41, 8099–8139 (2012).
Ferrando, R., Jellinek, J. & Johnston, R. L. Nanoalloys: from theory to applications of alloy clusters and nanoparticles. Chem. Rev. 108, 845–910 (2008).
Zhu, H. et al. Sn surface-enriched Pt–Sn bimetallic nanoparticles as a selective and stable catalyst for propane dehydrogenation. J. Catal. 320, 52–62 (2014).
Wu, J., Peng, Z. & Bell, A. T. Effects of composition and metal particle size on ethane dehydrogenation over PtxSn100-x/Mg(Al)O (70⩽x⩽100). J. Catal. 311, 161–168 (2014).
López-Haro, M. et al. A macroscopically relevant 3D-metrology approach for nanocatalysis research. Part. Part. Syst. Charact. 35, 1700343 (2018).
Kirkland, E. J. Advanced Computing in Electron Microscopy (Springer, 2010).
Bernal, S. et al. The interpretation of HREM images of supported metal catalysts using image simulation: profile view images. Ultramicroscopy 72, 135–164 (1998).
Simonelli, L. et al. CLÆSS: the hard X-ray absorption beamline of the ALBA CELLS synchrotron. Cogent Phys. 3, 1231987 (2016).
Guilera, G., Rey, F., Hernández-Fenollosa, J. & Cortés-Vergaz, J. J. One body, many heads; the Cerberus of catalysis. A new multipurpose in-situ cell for XAS at ALBA. J. Phys. Conf. Ser. 430, 012057 (2013).
Ravel, B. & Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Radiat. 12, 537–541 (2005).
Yin, F., Ji, S., Wu, P., Zhao, F. & Li, C. Deactivation behavior of Pd-based SBA-15 mesoporous silica catalysts for the catalytic combustion of methane. J. Catal. 257, 108–116 (2008).
Allian, A. D. et al. Chemisorption of CO and mechanism of CO oxidation on supported platinum nanoclusters. J. Am. Chem. Soc. 133, 4498–4517 (2011).
Acknowledgements
This work has been supported by the European Union through the European Research Council (grant ERC-AdG-2014-671093, SynCatMatch) and the Spanish government through the Severo Ochoa Programme (SEV-2016-0683). L.L. thanks ITQ for providing a contract. The authors also thank the Microscopy Service of UPV for the TEM and STEM measurements. The XAS measurements were carried out in CLAESS beamline at the ALBA synchrotron. HR STEM measurements were performed at DME-UCA in Cadiz University with financial support from FEDER/MINECO (MAT2017-87579-R and MAT2016-81118-P). A relevant patent application (European patent application No. 19382024.8) has been presented. C.W.L. thanks CAPES (Science without Frontiers-Process no. 13191/13-6) for a predoctoral fellowship.
Author information
Authors and Affiliations
Contributions
A.C. conceived the project, directed the study and wrote the manuscript. L.L. carried out the synthesis, characterizations and catalytic measurements and collaborated in writing the manuscript. M.L.-H. and J.J.C. carried out the HR STEM measurements, image analysis and simulations. C.W.L. carried out the analysis of XAS data. L.L., C.L. and L.S. contributed to the collection of XAS data in the ALBA synchrotron. P.C. carried out the CO infrared adsorption experiments. All the authors discussed the results and contributed to the production of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–5, Supplementary Figs. 1–71 and Supplementary refs. 1–8.
Rights and permissions
About this article
Cite this article
Liu, L., Lopez-Haro, M., Lopes, C.W. et al. Regioselective generation and reactivity control of subnanometric platinum clusters in zeolites for high-temperature catalysis. Nat. Mater. 18, 866–873 (2019). https://doi.org/10.1038/s41563-019-0412-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-019-0412-6
This article is cited by
-
Regioselective hydroformylation of propene catalysed by rhodium-zeolite
Nature (2024)
-
Co-aromatization of methane and hexane over Pt encapsulated in ZSM-5 zeolite and the electronic effect of K promoters
Science China Chemistry (2024)
-
Structure-dependence and metal-dependence on atomically dispersed Ir catalysts for efficient n-butane dehydrogenation
Nature Communications (2023)
-
Trapped by the germanium ring
Nature Catalysis (2023)
-
Germanium-enriched double-four-membered-ring units inducing zeolite-confined subnanometric Pt clusters for efficient propane dehydrogenation
Nature Catalysis (2023)