Abstract
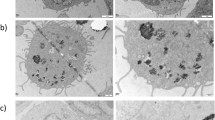
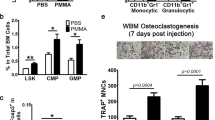
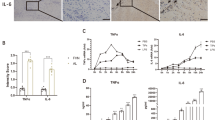
Initiation of the innate sterile inflammatory response that can develop in response to microparticle exposure is little understood. Here, we report that a potent type 2 immune response associated with the accumulation of neutrophils, eosinophils and alternatively activated (M2) macrophages was observed in response to sterile microparticles similar in size to wear debris associated with prosthetic implants. Although elevations in interleukin-33 (IL-33) and type 2 cytokines occurred independently of caspase-1 inflammasome signalling, the response was dependent on Bruton’s tyrosine kinase (BTK). IL-33 was produced by macrophages and BTK-dependent expression of IL-33 by macrophages was sufficient to initiate the type 2 response. Analysis of inflammation in patient periprosthetic tissue also revealed type 2 responses under aseptic conditions in patients undergoing revision surgery. These findings indicate that microparticle-induced sterile inflammation is initiated by macrophages activated to produce IL-33. They further suggest that both BTK and IL-33 may provide therapeutic targets for wear debris-induced periprosthetic inflammation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Kurtz, S., Ong, K., Lau, E., Mowat, F. & Halpern, M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J. Bone Joint Surg. Am. 89, 780–785 (2007).
Cobelli, N., Scharf, B., Crisi, G. M., Hardin, J. & Santambrogio, L. Mediators of the inflammatory response to joint replacement devices. Nat. Rev. Rheumatol. 7, 600–608 (2011).
Goodman, S. B. et al. Novel biological strategies for treatment of wear particle-induced periprosthetic osteolysis of orthopaedic implants for joint replacement. J. R. Soc. Interface 11, 20130962 (2014).
Veiseh, O. et al. Size- and shape-dependent foreign body immune response to materials implanted in rodents and non-human primates. Nat. Mater. 14, 643–651 (2015).
Antonios, J. K., Yao, Z., Li, C., Rao, A. J. & Goodman, S. B. Macrophage polarization in response to wear particles in vitro. Cell. Mol. Immunol. 10, 471–482 (2013).
Nakashima, Y. et al. Signaling pathways for tumor necrosis factor-alpha and interleukin-6 expression in human macrophages exposed to titanium-alloy particulate debris in vitro. J. Bone Joint Surg. Am. 81, 603–615 (1999).
Potnis, P. A., Dutta, D. K. & Wood, S. C. Toll-like receptor 4 signaling pathway mediates proinflammatory immune response to cobalt-alloy particles. Cell Immunol. 282, 53–65 (2013).
Bi, Y., Collier, T. O., Goldberg, V. M., Anderson, J. M. & Greenfield, E. M. Adherent endotoxin mediates biological responses of titanium particles without stimulating their phagocytosis. J. Orthop. Res. 20, 696–703 (2002).
Greenfield, E. M., Beidelschies, M. A., Tatro, J. M., Goldberg, V. M. & Hise, A. G. Bacterial pathogen-associated molecular patterns stimulate biological activity of orthopaedic wear particles by activating cognate Toll-like receptors. J. Biol. Chem. 285, 32378–32384 (2010).
Mishra, P. K. et al. Micrometer-sized titanium particles can induce potent Th2-type responses through TLR4-independent pathways. J. Immunol. 187, 6491–6498 (2011).
Kool, M. et al. An unexpected role for uric acid as an inducer of T helper 2 cell immunity to inhaled antigens and inflammatory mediator of allergic asthma. Immunity 34, 527–540 (2011).
Kuroda, E. et al. Silica crystals and aluminum salts regulate the production of prostaglandin in macrophages via NALP3 inflammasome-independent mechanisms. Immunity 34, 514–526 (2011).
Rathinam, V. A., Vanaja, S. K. & Fitzgerald, K. A. Regulation of inflammasome signaling. Nat. Immunol. 13, 333–342 (2012).
Mayer-Barber, K. D. et al. Caspase-1 independent IL-1β production is critical for host resistance to Mycobacterium tuberculosis and does not require TLR signaling in vivo. J. Immunol. 184, 3326–3330 (2010).
Ng, G. et al. Receptor-independent, direct membrane binding leads to cell-surface lipid sorting and Syk kinase activation in dendritic cells. Immunity 29, 807–818 (2008).
Flach, T. L. et al. Alum interaction with dendritic cell membrane lipids is essential for its adjuvanticity. Nat. Med. 17, 479–487 (2011).
Yamamoto, N. et al. The orally available spleen tyrosine kinase inhibitor 2-[7-(3,4-dimethoxyphenyl)-imidazo[1,2-c]pyrimidin-5-ylamino]nicotinamide dihydrochloride (BAY 61-3606) blocks antigen-induced airway inflammation in rodents. J. Pharmacol. Exp. Ther. 306, 1174–1181 (2003).
Schmitz, J. et al. IL-33, an interleukin-1-like cytokine that signals via the IL-1 receptor-related protein ST2 and induces T helper type 2-associated cytokines. Immunity 23, 479–490 (2005).
Whang, J. A. & Chang, B. Y. Bruton’s tyrosine kinase inhibitors for the treatment of rheumatoid arthritis. Drug Discov. Today 19, 1200–1204 (2014).
Baba, Y. et al. BLNK mediates Syk-dependent Btk activation. Proc. Natl Acad. Sci. USA 98, 2582–2586 (2001).
Shinohara, M. et al. The orally available Btk inhibitor ibrutinib (PCI-32765) protects against osteoclast-mediated bone loss. Bone 60, 8–15 (2014).
Pan, Z. et al. Discovery of selective irreversible inhibitors for Bruton’s tyrosine kinase. ChemMedChem 2, 58–61 (2007).
Honigberg, L. A. et al. The Bruton tyrosine kinase inhibitor PCI-32765 blocks B-cell activation and is efficacious in models of autoimmune disease and B-cell malignancy. Proc. Natl Acad. Sci. USA 107, 13075–13080 (2010).
Dubovsky, J. A. et al. Ibrutinib is an irreversible molecular inhibitor of ITK driving a Th1-selective pressure in T lymphocytes. Blood 122, 2539–2549 (2013).
Anthony, R. M. et al. Memory T(H)2 cells induce alternatively activated macrophages to mediate protection against nematode parasites. Nat. Med. 12, 955–960 (2006).
Thomas, J. D. et al. Colocalization of X-linked agammaglobulinemia and X-linked immunodeficiency genes. Science 261, 355–358 (1993).
Rawlings, D. J. et al. Mutation of unique region of Bruton’s tyrosine kinase in immunodeficient XID mice. Science 261, 358–361 (1993).
Khan, W. N. et al. Defective B cell development and function in Btk-deficient mice. Immunity 3, 283–299 (1995).
Sonnenberg, G. F. & Artis, D. Innate lymphoid cells in the initiation, regulation and resolution of inflammation. Nat. Med. 21, 698–708 (2015).
Carriere, V. et al. IL-33, the IL-1-like cytokine ligand for ST2 receptor, is a chromatin-associated nuclear factor in vivo. Proc. Natl Acad. Sci. USA 104, 282–287 (2007).
Liu, T. et al. Aspirin-exacerbated respiratory disease involves a cysteinyl leukotriene-driven IL-33-mediated mast cell activation pathway. J. Immunol. 195, 3537–3545 (2015).
Mishra, P. K., Patel, N., Wu, W., Bleich, D. & Gause, W. C. Prevention of type 1 diabetes through infection with an intestinal nematode parasite requires IL-10 in the absence of a Th2-type response. Mucosal Immunol. 6, 297–308 (2013).
Vannella, K. M. et al. Combinatorial targeting of TSLP, IL-25, and IL-33 in type 2 cytokine-driven inflammation and fibrosis. Sci. Transl. Med. 8, 337ra365 (2016).
Gause, W. C., Wynn, T. A. & Allen, J. E. Type 2 immunity and wound healing: evolutionary refinement of adaptive immunity by helminths. Nat. Rev. Immunol. 13, 607–614 (2013).
Qi, F. et al. Macrophages produce IL-33 by activating MAPK signaling pathway during RSV infection. Mol. Immunol. 87, 284–292 (2017).
Fock, V. et al. Macrophage-derived IL-33 is a critical factor for placental growth. J. Immunol. 191, 3734–3743 (2013).
Chang, Y. J. et al. Innate lymphoid cells mediate influenza-induced airway hyper-reactivity independently of adaptive immunity. Nat. Immunol. 12, 631–638 (2011).
Casella, J. F., Flanagan, M. D. & Lin, S. Cytochalasin D inhibits actin polymerization and induces depolymerization of actin filaments formed during platelet shape change. Nature 293, 302–305 (1981).
Kim, S. M. et al. TREM2 promotes Aβ phagocytosis by upregulating C/EBPα-dependent CD36 expression in microglia. Sci. Rep. 7, 11118 (2017).
Degterev, A. et al. Chemical inhibitor of nonapoptotic cell death with therapeutic potential for ischemic brain injury. Nat. Chem. Biol. 1, 112–119 (2005).
Cui, H. et al. Necrostatin-1 treatment inhibits osteocyte necroptosis and trabecular deterioration in ovariectomized rats. Sci. Rep. 6, 33803 (2016).
Gastman, B. R., Johnson, D. E., Whiteside, T. L. & Rabinowich, H. Caspase-mediated degradation of T-cell receptor zeta-chain. Cancer Res. 59, 1422–1427 (1999).
Amaral, F. A. et al. NLRP3 inflammasome-mediated neutrophil recruitment and hypernociception depend on leukotriene B(4) in a murine model of gout. Arthritis Rheum. 64, 474–484 (2012).
van Beuningen, H. M., Glansbeek, H. L., van der Kraan, P. M. & van den Berg, W. B. Osteoarthritis-like changes in the murine knee joint resulting from intra-articular transforming growth factor-β injections. Osteoarthritis Cartilage 8, 25–33 (2000).
Zhang, T. et al. The effect of osteoprotegerin gene modification on wear debris-induced osteolysis in a murine model of knee prosthesis failure. Biomaterials 30, 6102–6108 (2009).
Hashimoto, D. et al. Tissue-resident macrophages self-maintain locally throughout adult life with minimal contribution from circulating monocytes. Immunity 38, 792–804 (2013).
Jenkins, S. J. et al. IL-4 directly signals tissue-resident macrophages to proliferate beyond homeostatic levels controlled by CSF-1. J. Exp. Med. 210, 2477–2491 (2013).
Martinez, F. O., Gordon, S., Locati, M. & Mantovani, A. Transcriptional profiling of the human monocyte-to-macrophage differentiation and polarization: new molecules and patterns of gene expression. J. Immunol. 177, 7303–7311 (2006).
Koulouvaris, P. et al. Expression profiling reveals alternative macrophage activation and impaired osteogenesis in periprosthetic osteolysis. J. Orthop. Res. 26, 106–116 (2008).
Acknowledgements
This work was supported by National Institutes of Health grants: R01DK11379002, R01AI13163401A1, R01AI121250, R01AI101935, RO1AI124346, RO1AR060782 and R01AI114647-01A1. We would like to acknowledge Sandvik Osprey Powders, UK for generously providing the micrometre-sized CoCr material used in this paper. In addition we would like to thank E. M. Adler, M. C. Riley, F. R. Patterson and J. S. Hwang (Department of Orthopaedic Surgery, Rutgers – New Jersey Medical School) and S. R. Peters (Department of Pathology and Laboratory Medicine, Rutgers – New Jersey Medical School) for providing clinical samples and assistance in tissue preparation.
Author information
Authors and Affiliations
Contributions
W.C.G., P.K.M., M.P. and K.S.B. designed the experiments. P.K.M. and M.P. conducted all the experiments. B.B. and J.M. helped in collecting and processing of tissue biopsies from human clinical samples and A.M. provided technical assistance with immune staining. N.J.H. determined the sizes of wear debris particles and helped in particle separation. T.-D.K. provided caspase1-/- mice and technical suggestions for the experiments. K.S.B. and J.B. coordinated the clinical studies with human subjects. V.D. and R.B.B. provided technical assistance and advice in experiments assessing macrophage death and phagocytosis. N.C. and E.M.B. provided ST2-/- mice, laboratory assistance and suggestions for the experiments. A.R. provided the CCR2 reporter mice and associated technical assistance. W.C.G., P.K.M. and M.P. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–12
Rights and permissions
About this article
Cite this article
Mishra, P.K., Palma, M., Buechel, B. et al. Sterile particle-induced inflammation is mediated by macrophages releasing IL-33 through a Bruton’s tyrosine kinase-dependent pathway. Nature Mater 18, 289–297 (2019). https://doi.org/10.1038/s41563-018-0271-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-018-0271-6
This article is cited by
-
Macrophage Polarization and the Osteoimmunology of Periprosthetic Osteolysis
Current Osteoporosis Reports (2022)
-
Activating an adaptive immune response from a hydrogel scaffold imparts regenerative wound healing
Nature Materials (2021)
-
Heterogeneity in the initiation, development and function of type 2 immunity
Nature Reviews Immunology (2020)
-
Induction of sterile type 2 inflammation
Nature Materials (2019)