Abstract
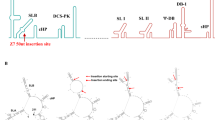
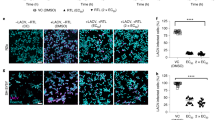
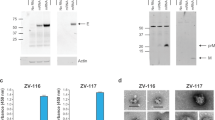
Zika virus is a mosquito-borne virus that is associated with neurodegenerative diseases, including Guillain–Barré syndrome1 and congenital Zika syndrome2. As Zika virus targets the nervous system, there is an urgent need to develop therapeutic strategies that inhibit Zika virus infection in the brain. Here, we have engineered a brain-penetrating peptide that works against Zika virus and other mosquito-borne viruses. We evaluated the therapeutic efficacy of the peptide in a lethal Zika virus mouse model exhibiting systemic and brain infection. Therapeutic treatment protected against mortality and markedly reduced clinical symptoms, viral loads and neuroinflammation, as well as mitigated microgliosis, neurodegeneration and brain damage. In addition to controlling systemic infection, the peptide crossed the blood–brain barrier to reduce viral loads in the brain and protected against Zika-virus-induced blood–brain barrier injury. Our findings demonstrate how engineering strategies can be applied to develop peptide therapeutics and support the potential of a brain-penetrating peptide to treat neurotropic viral infections.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Data supporting the findings of this study are available within the Article and its Supplementary Information files and from the corresponding author upon reasonable request. The data sets generated during and/or analysed during the current study are available from the corresponding author upon reasonable request.
References
Cao-Lormeau, V.-M. et al. Guillain–Barré syndrome outbreak associated with Zika virus infection in French Polynesia: a case–control study. Lancet 387, 1531–1539 (2016).
Rasmussen, S. A., Jamieson, D. J., Honein, M. A. & Petersen, L. R. Zika virus and birth defects—reviewing the evidence for causality. N. Engl. J. Med. 2016, 1981–1987 (2016).
Cugola, F. R. et al. The Brazilian Zika virus strain causes birth defects in experimental models. Nature 534, 267–271 (2016).
Garcez, P. P. et al. Zika virus impairs growth in human neurospheres and brain organoids. Science 352, 816–818 (2016).
Broutet, N. et al. Zika virus as a cause of neurologic disorders. N. Engl. J. Med. 374, 1506–1509 (2016).
Araujo, A. Q., Silva, M. T. T. & Araujo, A. P. Zika virus-associated neurological disorders: a review. Brain 139, 2122–2130 (2016).
Abrams, R. P., Solis, J. & Nath, A. Therapeutic approaches for Zika virus infection of the nervous system. Neurotherapeutics 14, 1027–1048 (2017).
Costa, V. V. et al. N-Methyl-d-aspartate (NMDA) receptor blockade prevents neuronal death induced by Zika virus infection. mBio 8, e00350-17 (2017).
Dallmeier, K. & Neyts, J. Zika and other emerging viruses: aiming at the right target. Cell Host Microbe 20, 420–422 (2016).
Saiz, J.-C. & Martín-Acebes, M. A. The race to find antivirals for Zika virus. Antimicrob. Agents Chemother. 61, e00411–e00417 (2017).
Boldescu, V., Behnam, M. A., Vasilakis, N. & Klein, C. D. Broad-spectrum agents for flaviviral infections: dengue, Zika and beyond. Nat. Rev. Drug Discov. 16, 565–586 (2017).
Li, F. et al. Viral infection of the central nervous system and neuroinflammation precede blood–brain barrier disruption during Japanese encephalitis virus infection. J. Virol. 89, 5602–5614 (2015).
Burt, F. J., Rolph, M. S., Rulli, N. E., Mahalingam, S. & Heise, M. T. Chikungunya: a re-emerging virus. Lancet 379, 662–671 (2012).
Carod-Artal, F. J., Wichmann, O., Farrar, J. & Gascón, J. Neurological complications of dengue virus infection. Lancet Neurol. 12, 906–919 (2013).
Badani, H., Garry, R. F. & Wimley, W. C. Peptide entry inhibitors of enveloped viruses: the importance of interfacial hydrophobicity. Biochim. Biophys. Acta Biomembr. 1838, 2180–2197 (2014).
Kostyuchenko, V. A. et al. Structure of the thermally stable Zika virus. Nature 533, 425–428 (2016).
Cho, N.-J. et al. Mechanism of an amphipathic α-helical peptide’s antiviral activity involves size-dependent virus particle lysis. ACS Chem. Biol. 4, 1061–1067 (2009).
Jackman, J. A., Saravanan, R., Zhang, Y., Tabaei, S. R. & Cho, N. J. Correlation between membrane partitioning and functional activity in a single lipid vesicle assay establishes design guidelines for antiviral peptides. Small. 11, 2372–2379 (2015).
Stalmans, S. et al. Cell-penetrating peptides selectively cross the blood–brain barrier in vivo. PLoS One 10, e0139652 (2015).
Garton, M. et al. Method to generate highly stable d-amino acid analogs of bioactive helical peptides using a mirror image of the entire PDB. Proc. Natl Acad. Sci. USA 115, 1505–1510 (2018).
Jackman, J. A., Goh, H. Z., Zhdanov, V. P., Knoll, W. & Cho, N.-J. Deciphering how pore formation causes strain-induced membrane lysis of lipid vesicles. J. Am. Chem. Soc. 138, 1406–1413 (2016).
Hatzakis, N. S. et al. How curved membranes recruit amphipathic helices and protein anchoring motifs. Nat. Chem. Biol. 5, 835–841 (2009).
Sapparapu, G. et al. Neutralizing human antibodies prevent Zika virus replication and fetal disease in mice. Nature 540, 443–447 (2016).
Fernandez, E. et al. Human antibodies to the dengue virus E-dimer epitope have therapeutic activity against Zika virus infection. Nat. Immunol. 18, 1261–1269 (2017).
Yu, Y. et al. A peptide-based viral inactivator inhibits Zika virus infection in pregnant mice and fetuses. Nat. Commun. 8, 15672 (2017).
Iwasaki, A. Immune regulation of antibody access to neuronal tissues. Trends Mol. Med. 18, 227–245 (2017).
Jurado, K. A. et al. Antiviral CD8 T cells induce Zika-virus-associated paralysis in mice. Nat. Microbiol. 3, 141–147 (2018).
Olmo, I. G. et al. Zika virus promotes neuronal cell death in a non-cell autonomous manner by triggering the release of neurotoxic factors. Front. Immunol. 8, 1016 (2017).
Adibi, J. J., Marques, E. T. Jr, Cartus, A. & Beigi, R. H. Teratogenic effects of the Zika virus and the role of the placenta. Lancet 387, 1587–1590 (2016).
Anderson, M. R., Kashanchi, F. & Jacobson, S. Exosomes in viral disease. Neurotherapeutics 13, 535–546 (2016).
Cho, N.-J., Cho, S.-J., Cheong, K. H., Glenn, J. S. & Frank, C. W. Employing an amphipathic viral peptide to create a lipid bilayer on Au and TiO2. J. Am. Chem. Soc. 129, 10050–10051 (2007).
Jackman, J. A., Zhao, Z., Zhdanov, V. P., Frank, C. W. & Cho, N.-J. Vesicle adhesion and rupture on silicon oxide: influence of freeze–thaw pretreatment. Langmuir 30, 2152–2160 (2014).
Krauson, A. J. et al. Conformational fine-tuning of pore-forming peptide potency and selectivity. J. Am. Chem. Soc. 137, 16144–16152 (2015).
Cho, N.-J., Frank, C. W., Kasemo, B. & Höök, F. Quartz crystal microbalance with dissipation monitoring of supported lipid bilayers on various substrates. Nat. Protoc. 5, 1096–1106 (2010).
Kunding, A. H., Mortensen, M. W., Christensen, S. M. & Stamou, D. A fluorescence-based technique to construct size distributions from single-object measurements: application to the extrusion of lipid vesicles. Biophys. J. 95, 1176–1188 (2008).
Haddow, A. D. et al. Genetic characterization of Zika virus strains: geographic expansion of the Asian lineage. PLoS Negl. Trop. Dis. 6, e1477 (2012).
Foureaux, G. et al. Antiglaucomatous effects of the activation of intrinsic angiotensin-converting enzyme 2. Invest. Ophthalmol. Visual Sci. 54, 4296–4306 (2013).
Costa, V. V. et al. Subversion of early innate antiviral responses during antibody-dependent enhancement of Dengue virus infection induces severe disease in immunocompetent mice. Med. Microbiol. Immunol. 203, 231–250 (2014).
Amaral, D. C. et al. Intracerebral infection with dengue-3 virus induces meningoencephalitis and behavioral changes that precede lethality in mice. J. Neuroinflammation 8, 23 (2011).
Schmued, L. C., Stowers, C. C., Scallet, A. C. & Xu, L. Fluoro-Jade C results in ultra high resolution and contrast labeling of degenerating neurons. Brain Res. 1035, 24–31 (2005).
St John, A. L., Rathore, A. P. S., Raghavan, B., Ng, M.-L. & Abraham, S. N. Contributions of mast cells and vasoactive products, leukotrienes and chymase, to dengue virus-induced vascular leakage. eLife 2, e00481 (2013).
DiResta, G. et al. in Brain Edema VIII (eds Reulen, H. J., Baethmann, A., Fenstermacher, J., Marmarou, A. & Spatz, M.) 34–36 (Springer, Vienna, 1990).
Fenyk-Melody, J. E. et al. Comparison of the effects of perfusion in determining brain penetration (brain-to-plasma ratios) of small molecules in rats. Comp. Med. 54, 378–381 (2004).
Acknowledgements
This work was supported by the National Research Foundation of Singapore through an NRF Fellowship grant (NRF-NRFF2011-01), a Competitive Research Programme grant (NRF-CRP10-2012-07) and a Proof-of-Concept grant (NRF2015NRF-POC0001-19), the National Medical Research Council of Singapore (NMRC/CBRG/0005/2012) and the Centre for Precision Biology at Nanyang Technological University. This work also received support from the National Institute of Science and Technology in Dengue and Host-microorganism Interaction (INCT dengue), which is a programme sponsored by the Brazilian National Science Council (CNPq, Brazil) and the Minas Gerais Foundation for Science (FAPEMIG, Brazil). This work also received support from Financiadora de Estudos ePesquisa (FINEP 01.16.0050.00, Brazil), PP SUS: APQ-03744-17 and Comissao de Apoio a Pessoal de Ensino Superior (CAPES, Brazil). I. Marcal, T. Colina, G. dos Santos and F. Assis are acknowledged for technical assistance with experiments. The authors also acknowledge G. Batista Menezes and M. Mota Antunes for help with the acquisition of confocal microscopy images.
Author information
Authors and Affiliations
Contributions
J.A.J., V.V.C., M.M.T. and N.-J.C. planned the studies. J.A.J., V.V.C., S.P., A.L.C.V.R., P.L.C., J.H.P, I.O.G., T.P.M., J.L.B., V.F.Q., C.M.Q-J., G.F., D.G.S., F.M.R., A.R.F., B.K.Y. and E.W. conducted experiments. J.A.J., V.V.C., S.P., D.G.S., F.M.R., B.D.S., M.M.T. and N.-J.C. interpreted the results. J.A.J. and N.-J.C. wrote the first draft of the paper. M.M.T., F.M.R. and N.-J.C. obtained funding. All authors reviewed, edited and approved the paper.
Corresponding author
Ethics declarations
Competing interests
N.-J.C. is a co-inventor on US patent no. 8,728,793. The other authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–15, Supplementary Tables 1–6
Rights and permissions
About this article
Cite this article
Jackman, J.A., Costa, V.V., Park, S. et al. Therapeutic treatment of Zika virus infection using a brain-penetrating antiviral peptide. Nature Mater 17, 971–977 (2018). https://doi.org/10.1038/s41563-018-0194-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-018-0194-2
This article is cited by
-
Exosome-disrupting peptides for cancer immunotherapy
Nature Materials (2023)
-
Curvature-sensing peptide inhibits tumour-derived exosomes for enhanced cancer immunotherapy
Nature Materials (2023)
-
Nanotechnology-based strategies against SARS-CoV-2 variants
Nature Nanotechnology (2022)
-
A review on structural genomics approach applied for drug discovery against three vector-borne viral diseases: Dengue, Chikungunya and Zika
Virus Genes (2022)
-
Antiviral surfaces and coatings and their mechanisms of action
Communications Materials (2021)