Abstract
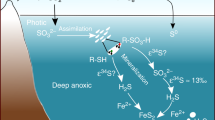
Metabolisms that evolved in the Archaean era (4.0–2.5 billion years ago) preferentially selected iron, manganese and molybdenum to form metalloproteins, whereas the majority of zinc-, copper- and vanadium-binding proteins emerged much later. The initial preference for these elements is commonly interpreted to reflect their availability in anoxic seawater, with free sulfide proposed as a key influence. While sulfidic waters reduce the availability of zinc and copper, they also remove molybdenum and leave behind vanadium. Furthermore, current geochemical data reflect predominantly ferruginous (Fe2+-rich), rather than sulfidic, conditions. Consistent with this, recent sedimentological work has uncovered abundant iron silicate minerals in Archaean rocks. Here we quantify metal partitioning during the formation and subsequent diagenesis of an Fe(ii) silicate mineral, a precursor to crystalline greenalite, in both seawater and hot hydrothermal fluids. Our data show that Fe(ii) silicates could have precipitated rapidly in Archaean hydrothermal plumes, severely attenuating hydrothermal delivery of key nutrients, in particular copper, zinc and vanadium. These results provide a mechanistic explanation for metal availability patterns in Archaean oceans that is consistent with temporal patterns of metal utilization predicted from protein structures and comparative genomics. Further, our data suggest natural greenalite may provide an archive of metal availability in deep time.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
A supplementary file containing experimental data is available online in the BGS data repository (https://doi.org/10.5285/c40cdd54-583a-43b2-b5fe-1784cbabfd71). Correspondence and requests for data or materials should be directed to the corresponding author.
References
Saito, M. A., Sigman, D. M. & Morel, F. M. M. The bioinorganic chemistry of the ancient ocean: the co-evolution of cyanobacterial metal requirements and biogeochemical cycles at the Archean–Proterozoic boundary? Inorg. Chim. Acta 356, 308–318 (2003).
Anbar, A. D. & Knoll, A. H. Proterozoic ocean chemistry and evolution: a bioinorganic bridge? Science 297, 1137–1143 (2002).
Dupont, C. L., Butcher, A., Valas, R. E., Bourne, P. E. & Caetano-Anollés, G. History of biological metal utilization inferred through phylogenomic analysis of protein structures. Proc. Natl Acad. Sci. USA 107, 10567–10572 (2010).
Dupont, C. L., Yang, S., Palenik, B. & Bourne, P. E. Modern proteomes contain putative imprints of ancient shifts in trace metal geochemistry. Proc. Natl Acad. Sci. USA 103, 17822–17827 (2006).
Guilbaud, R., Poulton, S. W., Butterfield, N. J., Zhu, M. & Shields-Zhou, G. A. A global transition to ferruginous conditions in the early Neoproterozoic oceans. Nat. Geosci. 8, 466–470 (2015).
Poulton, S. W. & Canfield, D. E. Ferruginous conditions: a dominant feature of the ocean through Earth’s history. Elements 7, 107–112 (2011).
Zegeye, A. et al. Green rust formation controls nutrient availability in a ferruginous water column. Geology 40, 599–602 (2012).
Crowe, S. A. et al. Deep-water anoxygenic photosythesis in a ferruginous chemocline. Geobiology 12, 322–339 (2014).
Maliva, R. G., Knoll, A. H. & Simonson, B. M. Secular change in the Precambrian silica cycle: insights from chert petrology. Geol. Soc. Am. Bull. 117, 835–845 (2005).
Tosca, N. J., Guggenheim, S. & Pufahl, P. K. An authigenic origin for Precambrian greenalite: implications for iron formation and the chemistry of ancient seawater. Geol. Soc. Am. Bull. https://doi.org/10.1130/B31339.1 (2016).
Hinz, I. L., Nims, C., Theuer, S., Templeton, A. S. & Johnson, J. E. Ferric iron triggers greenalite formation in simulated Archean seawater. Geology 49, 905–910 (2021).
Jiang, C. Z. & Tosca, N. J. Fe(ii)-carbonate precipitation kinetics and the chemistry of anoxic ferruginous seawater. Earth Planet. Sci. Lett. 506, 231–242 (2019).
Rasmussen, B., Krapež, B., Muhling, J. R. & Suvorova, A. Precipitation of iron silicate nanoparticles in early Precambrian oceans marks Earth’s first iron age. Geology 43, 303–306 (2015).
Rasmussen, B., Muhling, J. R., Suvorova, A. & Krapež, B. Greenalite precipitation linked to the deposition of banded iron formations downslope from a late Archean carbonate platform. Precambrian Res. 290, 49–62 (2017).
Rasmussen, B., Krapež, B. & Muhling, J. R. Hematite replacement of iron-bearing precursor sediments in the 3.46-b.y.-old Marble Bar Chert, Pilbara craton, Australia. Geol. Soc. Am. Bull. 126, 1245–1258 (2014).
Pufahl, P. K., Anderson, S. L. & Hiatt, E. E. Dynamic sedimentation of Paleoproterozoic continental margin iron formation, Labrador Trough, Canada: paleoenvironments and sequence stratigraphy. Sediment. Geol. 309, 48–65 (2014).
Edwards, C. T., Pufahl, P. K., Hiatt, E. E. & Kyser, T. K. Paleoenvironmental and taphonomic controls on the occurrence of Paleoproterozoic microbial communities in the 1.88 Ga Ferriman Group, Labrador Trough, Canada. Precambrian Res. 212–213, 91–106 (2012).
Rasmussen, B., Muhling, J. R. & Krapež, B. Greenalite and its role in the genesis of early Precambrian iron formations—a review. Earth Sci. Rev. 217, 103613 (2021).
Rasmussen, B., Muhling, J. R., Tosca, N. J. & Tsikos, H. Evidence for anoxic shallow oceans at 2.45 Ga: implications for the rise of oxygenic photosynthesis. Geology 47, 622–626 (2019).
Johnson, J. E., Muhling, J. R., Cosmidis, J., Rasmussen, B. & Templeton, A. S. Low‐Fe(iii) dreenalite was a primary mineral from Neoarchean oceans. Geophys. Res. Lett. https://doi.org/10.1002/2017GL076311 (2018).
Halevy, I. & Bachan, A. The geologic history of seawater pH. Science 355, 1069–1071 (2017).
Krissansen-Totton, J., Arney, G. N. & Catling, D. C. Constraining the climate and ocean pH of the early Earth with a geological carbon cycle model. Proc. Natl Acad. Sci. USA 115, 4105–4110 (2018).
Halevy, I., Alesker, M., Schuster, E. M., Popovitz-Biro, R. & Feldman, Y. A key role for green rust in the Precambrian oceans and the genesis of iron formations. Nat. Geosci. 10, 135–139 (2017).
Tosca, N. J., Jiang, C. Z., Rasmussen, B. & Muhling, J. Products of the iron cycle on the early Earth. Free Radic. Biol. Med. 140, 138–153 (2019).
Tosca, N. J. & Tutolo, B. M. Hydrothermal vent fluid–seawater mixing and the origins of Archean iron formation. Geochim. Cosmochim. Acta 352, 51–68 (2023).
Urabe, T. et al. The effect of magmatic activity on hydrothermal venting along the superfast-spreading East Pacific Rise. Science 269, 1092–1095 (1995).
Krapež, B., Barley, M. E. & Pickard, A. L. Hydrothermal and resedimented origins of the precursor sediments to banded iron formation: sedimentological evidence from the Early Palaeoproterozoic Brockman Supersequence of Western Australia. Sedimentology 50, 979–1011 (2003).
Dodd, M. S. et al. Widespread occurrences of variably crystalline 13C-depleted graphitic carbon in banded iron formations. Earth Planet. Sci. Lett. 512, 163–174 (2019).
Jacobsen, S. B. & Pimentel-Klose, M. R. Nd isotopic variations in Precambrian banded iron formations. Geophys. Res. Lett. 15, 393–396 (1988).
Derry, L. A. & Jacobsen, S. B. The chemical evolution of Precambrian seawater: evidence from REEs in banded iron formations. Geochim. Cosmochim. Acta 54, 2965–2977 (1990).
Beukes, N. J., Klein, C., Kaufman, A. J. & Hayes, J. M. Carbonate petrography, kerogen distribution, and carbon and oxygen isotope variations in an early Proterozoic transition from limestone to iron-formation deposition, Transvaal Supergroup, South Africa. Econ. Geol. 85, 663–690 (1990).
Kump, L. R. & Seyfried, W. E. Jr. Hydrothermal Fe fluxes during the Precambrian: effect of low oceanic sulfate concentrations and low hydrostatic pressure on the composition of black smokers. Earth Planet. Sci. Lett. 235, 654–662 (2005).
Tivey, M. K. Generation of seafloor hydrothermal vent fluids and associated mineral deposits. Oceanography 20, 50–65 (2007).
Rasmussen, B., Muhling, J. R. & Fischer, W. W. Greenalite nanoparticles in alkaline vent plumes as templates for the origin of life. Astrobiology 21, 246–259 (2021).
Klein, C. Greenalite, stilpnomelane, minnesotaite, crocidolite and carbonates in a very low-grade metamorphic Precambrian iron formation. Can. Mineral. 12, 475–498 (1974).
Nutman, A. P., Bennett, V. C. & Friend, C. R. L. Seeing through the magnetite: reassessing Eoarchean atmosphere composition from Isua (Greenland) ≥3.7 Ga banded iron formations. Geosci. Front. https://doi.org/10.1016/j.gsf.2017.02.008 (2017).
Rasmussen, B., Muhling, J. R., Suvorova, A. & Krapež, B. Dust to dust: evidence for the formation of ‘primary’ hematite dust in banded iron formations via oxidation of iron silicate nanoparticles. Precambrian Res. 284, 49–63 (2016).
Johnson, B. R. et al. Phosphorus burial in ferruginous SiO2-rich Mesoproterozoic sediments. Geology 48, 92–96 (2019).
Roshan, S., Wu, J. & Jenkins, W. J. Long-range transport of hydrothermal dissolved Zn in the tropical South Pacific. Mar. Chem. 183, 25–32 (2016).
Findlay, A. J., Gartman, A., Shaw, T. J. & Luther, G. W. Trace metal concentration and partitioning in the first 1.5 m of hydrothermal vent plumes along the Mid-Atlantic Ridge: TAG, Snakepit, and Rainbow. Chem. Geol. 412, 117–131 (2015).
Swanner, E. D. et al. Cobalt and marine redox evolution. Earth Planet. Sci. Lett. 390, 253–263 (2014).
Scott, C. et al. Bioavailability of zinc in marine systems through time. Nat. Geosci. 6, 125–128 (2013).
Robbins, L. J. et al. Authigenic iron oxide proxies for marine zinc over geological time and implications for eukaryotic metallome evolution. Geobiology 11, 295–306 (2013).
Konhauser, K. O. et al. Oceanic nickel depletion and a methanogen famine before the Great Oxidation Event. Nature 458, 750–753 (2009).
Scott, C. et al. Tracing the stepwise oxygenation of the Proterozoic ocean. Nature 452, 456–459 (2008).
Robbins, L. J. et al. Limited Zn and Ni mobility during simulated iron formation diagenesis. Chem. Geol. 402, 30–39 (2015).
Sumoondur, A., Shaw, S., Ahmed, I. & Benning, L. G. Green rust as a precursor for magnetite: an in situ synchrotron based study. Mineral. Mag. 72, 201–204 (2016).
Moore, E. K., Jelen, B. I., Giovannelli, D., Raanan, H. & Falkowski, P. G. Metal availability and the expanding network of microbial metabolisms in the Archaean eon. Nat. Geosci. 10, 629–636 (2017).
David, L. A. & Alm, E. J. Rapid evolutionary innovation during an Archaean genetic expansion. Nature 469, 93–96 (2011).
Zerkle, A. L., House, C. H., Cox, R. P. & Canfield, D. E. Metal limitation of cyanobacterial N2 fixation and implications for the Precambrian nitrogen cycle. Geobiology 4, 285–297 (2006).
Stüeken, E. E., Buick, R., Guy, B. M. & Koehler, M. C. Isotopic evidence for biological nitrogen fixation by molybdenum-nitrogenase from 3.2 Gyr. Nature 520, 666–669 (2015).
Tostevin, R. Solubility, kinetics and metal drawdown during precipitation of Fe(II)-silicate over a range of temperatures. NERC EDS National Geoscience Data Centre https://doi.org/10.5285/c40cdd54-583a-43b2-b5fe-1784cbabfd71 (2023).
Kandegedara, A. & Rorabacher, D. B. Noncomplexing Tertiary amines as ‘better’ buffers covering the range of pH 3−11. Temperature dependence of their acid dissociation constants. Anal. Chem. 71, 3140–3144 (1999).
Dietzel, M. Dissolution of silicates and the stability of polysilicic acid. Geochim. Cosmochim. Acta 64, 3275–3281 (2000).
Siever, R. The silica cycle in the Precambrian. Geochim. Cosmochim. Acta 56, 3265–3272 (1992).
Mortlock, R. A. et al. Silica and germanium in Pacific Ocean hydrothermal vents and plumes. Earth Planet. Sci. Lett. 119, 365–378 (1993).
Crowe, S. A. et al. Sulfate was a trace constituent of Archean seawater. Science 346, 735–739 (2014).
Dent, A. J. et al. B18: a core XAS spectroscopy beamline for Diamond. J. Phys. Conf. Ser. 190, 012039 (2009).
Acknowledgements
R.T. acknowledges financial support from NERC grant NE/M013014/1, NRF-DSIs CIMERA and GENUS and the BIOGRIP platform. We are grateful to N. Tosca for access to laboratory facilities and comments on an earlier version of this paper and to E. Tipper, M. Greaves and R. Hindshaw for assistance with ICP-OES analyses at the Department of Earth Science, University of Cambridge.
Author information
Authors and Affiliations
Contributions
R.T. designed the study, performed the experiments and analysed the results. I.A.M.A. and R.T. acquired synchrotron data, and I.A.M.A. modelled the results. R.T. wrote the paper with input from I.A.M.A.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Geoscience thanks Romain Guilbaud, Elizabeth Swanner and Jena Johnson for their contribution to the peer review of this work. Primary Handling Editor: James Super, in collaboration with the Nature Geoscience team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–5, Tables 1 and 2 and Discussion.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tostevin, R., Ahmed, I.A.M. Micronutrient availability in Precambrian oceans controlled by greenalite formation. Nat. Geosci. 16, 1188–1193 (2023). https://doi.org/10.1038/s41561-023-01294-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41561-023-01294-0
This article is cited by
-
Metals for microbes in the ancient sea
Nature Geoscience (2023)