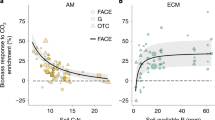
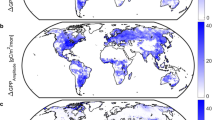
Abstract
Clarifying how increased atmospheric CO2 concentration (eCO2) contributes to accelerated land carbon sequestration remains important since this process is the largest negative feedback in the coupled carbon–climate system. Here, we constrain the sensitivity of the terrestrial carbon sink to eCO2 over the temperate Northern Hemisphere for the past five decades, using 12 terrestrial ecosystem models and data from seven CO2 enrichment experiments. This constraint uses the heuristic finding that the northern temperate carbon sink sensitivity to eCO2 is linearly related to the site-scale sensitivity across the models. The emerging data-constrained eCO2 sensitivity is 0.64 ± 0.28 PgC yr−1 per hundred ppm of eCO2. Extrapolating worldwide, this northern temperate sensitivity projects the global terrestrial carbon sink to increase by 3.5 ± 1.9 PgC yr−1 for an increase in CO2 of 100 ppm. This value suggests that CO2 fertilization alone explains most of the observed increase in global land carbon sink since the 1960s. More CO2 enrichment experiments, particularly in boreal, arctic and tropical ecosystems, are required to explain further the responsible processes.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
Driver data of MsTMIP models are available from https://doi.org/10.3334/ORNLDAAC/1220. The outputs of MsTMIP models are available from https://doi.org/10.3334/ORNLDAAC/1225. Global Carbon Budget 2017 data (Global_Carbon_Budget_2017v1.3.xlsx) are available from https://doi.org/10.18160/GCP-2017. CRU climatology data (CRU CL v. 2.0) are available from https://crudata.uea.ac.uk/cru/data/hrg/.
Code availability
The code for fitting the two-box model, as given in equation (3), is shown in Supplementary Information.
References
Field, C. B. & Raupach, M. R. The Global Carbon Cycle: Integrating Humans, Climate, and the Natural World (Island Press, 2004).
Le Quéré, C. et al. Global Carbon Budget 2017. Earth Syst. Sci. Data 10, 405–448 (2018).
Friedlingstein, P. et al. On the contribution of CO2 fertilization to the missing biospheric sink. Glob. Biogeochem. Cycles 9, 541–556 (1995).
Shevliakova, E. et al. Historical warming reduced due to enhanced land carbon uptake. Proc. Natl. Acad. Sci. USA 110, 16730–16735 (2013).
Schimel, D., Stephens, B. B. & Fisher, J. B. Effect of increasing CO2 on the terrestrial carbon cycle. Proc. Natl. Acad. Sci. USA 112, 436–441 (2015).
Huntzinger, D. N. et al. Uncertainty in the response of terrestrial carbon sink to environmental drivers undermines carbon–climate feedback predictions. Sci. Rep. 7, 4765 (2017).
Keenan, T. F. et al. Recent pause in the growth rate of atmospheric CO2 due to enhanced terrestrial carbon uptake. Nat. Commun. 7, 13428 (2016).
Sitch, S. et al. Recent trends and drivers of regional sources and sinks of carbon dioxide. Biogeosciences 12, 653–679 (2015).
Ilyina, T. & Friedlingstein, P. WCRP Grand Challenge: Carbon feedbacks in the climate system. White paper (37th Session of the WCRP Joint Scientific Committee, 2016); https://www.wcrp-climate.org/component/content/article/872-carbon-feedbacks-overview?catid=139&Itemid=539
Norby, R. J. & Zak, D. R. Ecological lessons from free-air CO2 enrichment (FACE) experiments. Annu. Rev. Ecol. Evol. Syst. 42, 181–203 (2011).
Zaehle, S. et al. Evaluation of 11 terrestrial carbon–nitrogen cycle models against observations from two temperate free-air CO2 enrichment studies. New Phytol. 202, 803–822 (2014).
Jastrow, J. D. et al. Elevated atmospheric carbon dioxide increases soil carbon. Glob. Change Biol. 11, 2057–2064 (2005).
De Graaff, M. A., van Groenigen, K. J., Six, J., Hungate, B. & van Kessel, C. Interactions between plant growth and soil nutrient cycling under elevated CO2: a meta-analysis. Glob. Change Biol. 12, 2077–2091 (2006).
Hickler, T. et al. CO2 fertilization in temperate FACE experiments not representative of boreal and tropical forests. Glob. Change Biol. 14, 1531–1542 (2008).
Finzi, A. C. et al. Increases in nitrogen uptake rather than nitrogen-use efficiency support higher rates of temperate forest productivity under elevated CO2. Proc. Natl. Acad. Sci. USA 104, 14014–14019 (2007).
Drake, J. E. et al. Increases in the flux of carbon belowground stimulate nitrogen uptake and sustain the long-term enhancement of forest productivity under elevated CO2. Ecol. Lett. 14, 349–357 (2011).
Terrer, C., Vicca, S., Hungate, B. A., Phillips, R. P. & Prentice, I. C. Mycorrhizal association as a primary control of the CO2 fertilization effect. Science 353, 72–74 (2016).
Cox, P. M. et al. Sensitivity of tropical carbon to climate change constrained by carbon dioxide variability. Nature 494, 341–344 (2013).
Wenzel, S., Cox, P. M., Eyring, V. & Friedlingstein, P. Projected land photosynthesis constrained by changes in the seasonal cycle of atmospheric CO2. Nature 538, 499–501 (2016).
Mystakidis, S., Davin, E. L., Gruber, N. & Seneviratne, S. I. Constraining future terrestrial carbon cycle projections using observation-based water and carbon flux estimates. Glob. Change Biol. 22, 2198–2215 (2016).
Zhao, C. et al. Plausible rice yield losses under future climate warming. Nat. Plants 3, 16202 (2016).
Lian, X. et al. Partitioning global land evapotranspiration using CMIP5 models constrained by observations. Nat. Clim. Change 8, 640–646 (2018).
Huntzinger, D. N. et al. The North American Carbon Program Multi-scale Synthesis and Terrestrial Model Intercomparison Project - Part 1: overview and experimental design. Geosci. Model Dev. 6, 2121–2133 (2013).
Norby, R. J., Warren, J. M., Iversen, C. M., Medlyn, B. E. & McMurtrie, R. E. CO2 enhancement of forest productivity constrained by limited nitrogen availability. Proc. Natl. Acad. Sci. USA 107, 19368–19373 (2010).
De Kauwe, M. G. et al. Where does the carbon go? A model-data intercomparison of vegetation carbon allocation and turnover processes at two temperate forest free-air CO2 enrichment sites. New Phytol. 203, 883–899 (2014).
Franks, P. J. et al. Sensitivity of plants to changing atmospheric CO2 concentration: from the geological past to the next century. New Phytol. 197, 1077–1094 (2013).
Wang, H. et al. Towards a universal model for carbon dioxide uptake by plants. Nat. Plants 3, 734–741 (2017).
Bacastow, R. & Keeling, C. D. in Carbon and the Biosphere (eds Woodwell, G. M. & Pecan, E. V.) 86–135 (US Dep. of Comm., 1973).
Peñuelas, J. et al. Human-induced nitrogen–phosphorus imbalances alter natural and managed ecosystems across the globe. Nat. Commun. 4, 2934 (2013).
McCarthy, H. R. et al. Re-assessment of plant carbon dynamics at the Duke free-air CO2 enrichment site: interactions of atmospheric [CO2] with nitrogen and water availability over stand development. New Phytol. 185, 514–528 (2010).
Reich, P. B. & Hobbie, S. E. Decade-long soil nitrogen constraint on the CO2 fertilization of plant biomass. Nat. Clim. Change 3, 278–282 (2013).
Norby, R. J. et al. Model-data synthesis for the next generation of forest free-air CO2 enrichment (FACE) experiments. New Phytol. 209, 17–28 (2016).
Drake, J. E. et al. Short-term carbon cycling responses of a mature eucalypt woodland to gradual stepwise enrichment of atmospheric CO2 concentration. Glob. Change Biol. 22, 380–390 (2016).
Medlyn, B. E. et al. Using ecosystem experiments to improve vegetation models. Nat. Clim. Change 5, 528–534 (2015).
Huntzinger, D. N. et al. NACP MsTMIP: Global 0.5-deg Terrestrial Biosphere Model Outputs in Standard Format. v.1.0 (ORNL DAAC, 2016); https://doi.org/10.3334/ORNLDAAC/1225
Wei, Y. et al. NACP MsTMIP: Global and North American Driver Data for Multi-model Intercomparison (ORNL DAAC, 2014); https://doi.org/10.3334/ORNLDAAC/1220
Supplemental data of Global Carbon Budget 2017 v.1.0 (Global Carbon Project, 2017); https://doi.org/10.18160/gcp-2017
Jung, M., Henkel, K., Herold, M. & Churkina, G. Exploiting synergies of global land cover products for carbon cycle modeling. Remote Sens. Environ. 101, 534–553 (2006).
Wei, Y. et al. The North American Carbon Program Multi-scale Synthesis and Terrestrial Model Intercomparison Project: part 2: environmental driver data. Geosci. Model Dev. 7, 2875–2893 (2014).
Farquhar, G. D., Caemmerer, S. V. & Berry, J. A. A biochemical model of photosynthetic CO2 assimilation in leaves of C3 species. Planta 149, 78–90 (1980).
Maire, V. et al. The coordination of leaf photosynthesis links C and N fluxes in C3 plant species. PLoS ONE 7, e38345 (2012).
Togashi, H. F. et al. Thermal acclimation of leaf photosynthetic traits in an evergreen woodland, consistent with the coordination hypothesis. Biogeosciences 15, 3461–3474 (2018).
Mark, N., David, L., Mike, H. & Ian, M. A high-resolution data set of surface climate over global land areas. Clim. Res. 21, 1–25 (2002).
Ito, A. et al. Decadal trends in the seasonal-cycle amplitude of terrestrial CO2 exchange resulting from the ensemble of terrestrial biosphere models. Tellus B Chem. Phys. Meteor. 68, 28968 (2016).
Acknowledgements
This study was supported by the Strategic Priority Research Program (A) of the Chinese Academy of Sciences (Grant XDA20050101), the National Key R&D Program of China (2017YFA0604702), and the National Natural Science Foundation of China (41861134036, 41701089). The full list of acknowledgements is provided in the Supplementary Information.
Author information
Authors and Affiliations
Contributions
S.P., Y.L., T.G. and P.C. designed the study. Y.L. performed the analysis. Y.L. and S.P. drafted the paper. All authors contributed to the interpretation of the results and to the text.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Description, Supplementary Figs. 1–25 and Tables 1–4, Supplementary References and full list of acknowledgements.
Rights and permissions
About this article
Cite this article
Liu, Y., Piao, S., Gasser, T. et al. Field-experiment constraints on the enhancement of the terrestrial carbon sink by CO2 fertilization. Nat. Geosci. 12, 809–814 (2019). https://doi.org/10.1038/s41561-019-0436-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41561-019-0436-1
This article is cited by
-
Aiming at a moving target: economic evaluation of adaptation strategies under the uncertainty of climate change and CO2 fertilization of European beech (Fagus sylvatica L.) and Silver fir (Abies alba Mill.)
Annals of Forest Science (2024)
-
Elevated CO2 levels promote both carbon and nitrogen cycling in global forests
Nature Climate Change (2024)
-
Transition from positive to negative indirect CO2 effects on the vegetation carbon uptake
Nature Communications (2024)
-
Past and future adverse response of terrestrial water storages to increased vegetation growth in drylands
npj Climate and Atmospheric Science (2023)
-
Evidence and attribution of the enhanced land carbon sink
Nature Reviews Earth & Environment (2023)