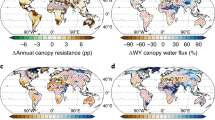
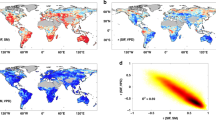
Abstract
Non-vascular vegetation has been shown to capture considerable quantities of rainfall, which may affect the hydrological cycle and climate at continental scales. However, direct measurements of rainfall interception by non-vascular vegetation are confined to the local scale, which makes extrapolation to the global effects difficult. Here we use a process-based numerical simulation model to show that non-vascular vegetation contributes substantially to global rainfall interception. Inferred average global water storage capacity including non-vascular vegetation was 2.7 mm, which is consistent with field observations and markedly exceeds the values used in land surface models, which average around 0.4 mm. Consequently, we find that the total evaporation of free water from the forest canopy and soil surface increases by 61% when non-vascular vegetation is included, resulting in a global rainfall interception flux that is 22% of the terrestrial evaporative flux (compared with only 12% for simulations where interception excludes non-vascular vegetation). We thus conclude that non-vascular vegetation is likely to significantly influence global rainfall interception and evaporation with consequences for regional- to continental-scale hydrologic cycling and climate.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Lange, O. et al. Temperate rainforest lichens in New Zealand: high thallus water content can severely limit photosynthetic CO2 exchange. Oecologia 95, 303–313 (1993).
Veneklaas, E. J. et al. Hydrological properties of the epiphyte mass of a montane tropical rain forest, Colombia. Vegetatio 89, 183–192 (1990).
Proctor, M. C. F. et al. Desiccation-tolerance in bryophytes: a review. Bryologist 110, 595–621 (2007).
Kranner, I., Beckett, R., Hochman, A. & Nash, T. H. III Desiccation-tolerance in lichens: a review. Bryologist 111, 576–593 (2008).
Cornelissen, J. H. C., Lang, S. I., Soudzilovskaia, N. A. & During, H. J. Comparative cryptogam ecology: a review of bryophyte and lichen traits that drive biogeochemistry. Ann. Bot. 99, 987–1001 (2007).
Lakatos, M. in Plant Desiccation Tolerance (eds Lüttge, U., Beck, E. & Bartels, D.) Ch. 5 (Springer, Berlin, 2011)..
Van Stan, J. T. & Pypker, T. G. A review and evaluation of forest canopy epiphyte roles in the partitioning and chemical enrichment of precipitation. Sci. Total Environ. 536, 813–824 (2015).
Savenije, H. H. G. The importance of interception and why we should delete the term evapotranspiration from our vocabulary. Hydrol. Process. 18, 1507–1511 (2004).
Davies-Barnard, T., Valdes, P. J., Jones, C. D. & Singarayer, J. S. Sensitivity of a coupled climate model to canopy interception capacity. Clim. Dyn. 42, 1715–1732 (2014).
Jarvis, A. Measuring and modelling the impact of land-use change in tropical hill-sides: the role of cloud interception to epiphytes. Adv. Environ. Monit. Model. 1, 118–148 (2000).
Wang, B. et al. Effect of succession gaps on the understory water-holding capacity in an over-mature alpine forest at the upper reaches of the Yangtze River. Hydrol. Process. 30, 692–703 (2016).
Porada, P., Weber, B., Elbert, W., Pöschl, U. & Kleidon, A. Estimating global carbon uptake by lichens and bryophytes with a process-based model. Biogeosciences 10, 6989–7033 (2013).
Porada, P., Weber, B., Elbert, W., Pöschl, U. & Kleidon, A. Estimating impacts of lichens and bryophytes on global biogeochemical cycles. Global Biogeochem. Cycles 28, 71–85 (2014).
Teuling, A. J., Uijlenhoet, R., Van den Hurk, B. & Seneviratne, S. I. Parameter sensitivity in LSMs: an analysis using stochastic soil moisture models and ELDAS soil parameters. J. Hydrometeorol. 10, 751–765 (2009).
Murray, S. J. Trends in 20th century global rainfall interception as simulated by a dynamic global vegetation model: implications for global water resources. Ecohydrology 7, 102–114 (2014).
Van Stan, J. T. et al. Tillandsia usneoides (L.) L. (Spanish moss) water storage and leachate characteristics from two maritime oak forest settings. Ecohydrology 8, 988–1004 (2015).
Hölscher, D., Köhler, L., Van Dijk, A. I. J. M. & Bruijnzeel, L. A. The importance of epiphytes to total rainfall interception by a tropical montane rain forest in Costa Rica. J. Hydrol. 292, 308–322 (2004).
Miralles, D. G., Gash, J. H., Holmes, T. R. H., De Jeu, R. A. M. & Dolman, A. J. Global canopy interception from satellite observations. J. Geophys. Res. 115, D16122 (2010).
Wang-Erlandsson, L., van der Ent, R. J., Gordon, L. J. & Savenije, H. H. G. Contrasting roles of interception and transpiration in the hydrological cycle — Part 1: temporal characteristics over land. Earth Syst. Dynam. 5, 441–469 (2014).
Kürschner, H. & Parolly, G. Phytomass and water-storing capacity of epiphytic rain forest bryophyte communities in S Ecuador. Bot. Jahrb. 125, 489–504 (2004).
Shuttleworth, W. J. Macrohydrology: the new challenge for process hydrology. J. Hydrol. 100, 31–56 (1988).
Weedon, G. et al. Creation of the WATCH forcing data and its use to assess global and regional reference crop evaporation over land during the twentieth century. J. Hydrometeorol. 12, 823–848 (2011).
Makkink, G. F. Testing the Penman formula by means of lysimeters. J. Inst. Wat. Engrs. 11, 277–288 (1957).
Porada, P., Pöschl, U., Kleidon, A., Beer, C. & Weber, B. Estimating global nitrous oxide emissions by lichens and bryophytes with a process-based productivity model. Biogeosciences 14, 1593–1602 (2017).
Monteith, J. Evaporation and surface temperature. Q. J. R. Meteorol. Soc. 107, 1–27 (1981).
Warrilow, D. A., Sangster, A. B. & Slingo, J. Modelling of Land Surface Processes and Their Influence on European Climate Technical Note DCTN 38 (Meteorological Office, 1986).
Porada, P., Ekici, A. & Beer, C. Effects of bryophyte and lichen cover on permafrost soil temperature at large scale. Cryosphere 10, 2291–2315 (2016).
Farquhar, G. & Von Caemmerer, S. in Encyclopedia of Plant Physiology Vol. 12 (eds Lange, O., Nobel, P., Osmond, C. & Ziegler, H.) 549–587 (Springer, Heidelberg, 1982).
Porada, P. et al. High potential for weathering and climate effects of non-vascular vegetation in the Late Ordovician. Nat. Commun. 7, 12113 (2016).
Bonan, G. et al. The land surface climatology of the Community Land Model coupled to the NCAR Community Climate Model. J. Clim. 15, 3123–3149 (2002).
Miralles, D. G. Global land-surface evaporation estimated from satellite-based observations. Hydrol. Earth Syst. Sci. 15, 453–469 (2011).
Martens, B. GLEAMv3: satellite-based land evaporation and root-zone soil moisture. Geosci. Model Dev. 10, 1903–1925 (2017).
Dahlman, L. & Palmqvist, K. Growth in two foliose tripartite lichens, Nephroma arcticum and Peltigera aphthosa: empirical modelling of external vs. internal factors. Funct. Ecol. 17, 821–831 (2003).
Gauslaa, Y. & Coxson, D. Interspecific and intraspecific variations in water storage in epiphytic old forest foliose lichens. Botany 89, 787–798 (2011).
Werner, F. A., Homeier, J., Oesker, M. & Boy, J. Epiphytic biomass of a tropical montane forest varies with topography. J. Trop. Ecol. 28, 23–31 (2012).
Fischer, T., Veste, M., Bens, O. & Hütt, R. F. Dew formation on the surface of biological soil crusts in central European sand ecosystems. Biogeosciences 9, 4621–4628 (2012).
Lidén, M., Jonsson-Cabrajic, A. V., Ottosson-Löfvenius, M., Palmqvist, K. & Lundmark, T. Species-specific activation time-lags can explain habitat restrictions in hydrophilic lichens. Plant Cell Environ. 33, 851–862 (2010).
Olson, D. et al. Terrestrial ecoregions of the world: a new map of life on Earth. BioScience 51, 933–938 (2001).
Acknowledgements
The Bolin Centre for Climate Research is thanked for financial support. The Max Planck Institute for Biogeochemistry provided computational resources. P.P. acknowledges funding from the European Union FP7-ENV project PAGE21 under contract number GA282700. J.T.V.S. acknowledges funding from the United States National Science Foundation (EAR-1518726).
Author information
Authors and Affiliations
Contributions
P.P., J.T.V.S. and A.K. designed the study. P.P. did the modelling analyses. P.P. and J.T.V.S. did the literature research for the model validation. P.P. and J.T.V.S. wrote the paper with input from A.K.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1 and 2; Supplementary References
Rights and permissions
About this article
Cite this article
Porada, P., Van Stan, J.T. & Kleidon, A. Significant contribution of non-vascular vegetation to global rainfall interception. Nature Geosci 11, 563–567 (2018). https://doi.org/10.1038/s41561-018-0176-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41561-018-0176-7
This article is cited by
-
Recent global decline in rainfall interception loss due to altered rainfall regimes
Nature Communications (2022)
-
Dew water-uptake pathways in Negev desert plants: a study using stable isotope tracers
Oecologia (2021)