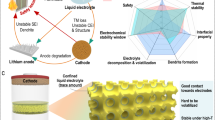
Abstract
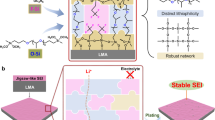
The solid–electrolyte interphase (SEI) in lithium (Li) metal batteries is often heterogeneous, containing a diverse range of species and has poor mechanical stability. The SEI undergoes constant cracking and reconstruction during electrochemical cycling, which is accompanied by the exhaustion of active Li and electrolytes, hindering practical applications of the batteries. Here we propose an in situ structural design of SEI to promote its homogeneity and improve its mechanical stability. A bilayer structure of SEI is tailored through trioxane-modulated electrolytes: the inner layer is dominated by LiF to improve homogeneity while the outer layer contains Li polyoxymethylene to improve mechanical stability, synergistically leading to mitigated reconstruction of SEI and reversible Li plating/stripping. The coin cell consisting of an ultrathin Li metal anode (50 μm) and a high-loading cathode (3.0 mAh cm−2)—with the tailored bilayer SEI—achieves 430 cycles tested at 1.2 mA cm−2, while the cell with an anion-derived SEI undergoes only 200 cycles under same conditions. A prototype 440 Wh kg−1 pouch cell (5.3 Ah), with a low negative/positive capacity ratio of 1.8 and lean electrolytes of 2.1 g Ah−1, achieves 130 cycles.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the article and its supplementary information files.
References
Tarascon, J. M. & Armand, M. Issues and challenges facing rechargeable lithium batteries. Nature 414, 359–367 (2001).
Winter, M., Barnett, B. & Xu, K. Before Li ion batteries. Chem. Rev. 118, 11433–11456 (2018).
Cheng, X.-B. et al. Toward safe lithium metal anode in rechargeable batteries: a review. Chem. Rev. 117, 10403–10473 (2017).
Louli, A. J. et al. Diagnosing and correcting anode-free cell failure via electrolyte and morphological analysis. Nat. Energy 5, 693–702 (2020).
Zhou, M. Y. et al. Quantifying the apparent electron transfer number of electrolyte decomposition reactions in anode-free batteries. Joule 6, 2122–2137 (2022).
Liu, J. et al. Pathways for practical high-energy long-cycling lithium metal batteries. Nat. Energy 4, 180–186 (2019).
Chen, S. et al. Critical parameters for evaluating coin cells and pouch cells of rechargeable Li-metal batteries. Joule 3, 1094–1105 (2019).
Peled, E. The electrochemical behavior of alkali and alkaline earth metals in nonaqueous battery systems—the solid electrolyte interphase model. J. Electrochem. Soc. 126, 2047–2051 (1979).
Xu, K. Nonaqueous liquid electrolytes for lithium-based rechargeable batteries. Chem. Rev. 104, 4303–4418 (2004).
Tikekar, M. D. et al. Design principles for electrolytes and interfaces for stable lithium-metal batteries. Nat. Energy 1, 16114 (2016).
Lin, D., Liu, Y. & Cui, Y. Reviving the lithium metal anode for high-energy batteries. Nat. Nanotechnol. 12, 194–206 (2017).
Yoon, I. et al. Measurement of mechanical and fracture properties of solid electrolyte interphase on lithium metal anodes in lithium ion batteries. Energy Storage Mater. 25, 296–304 (2020).
Gao, Y. et al. Unraveling the mechanical origin of stable solid electrolyte interphase. Joule 5, 1860–1872 (2021).
Li, Y. et al. Correlating structure and function of battery interphases at atomic resolution using cryoelectron microscopy. Joule 2, 2167–2177 (2018).
Wang, W. W. et al. Evaluating solid-electrolyte interphases for lithium and lithium-free anodes from nanoindentation features. Chem 6, 2728–2745 (2020).
Fan, X. et al. Non-flammable electrolyte enables Li-metal batteries with aggressive cathode chemistries. Nat. Nanotechnol. 13, 715–722 (2018).
Zhang, X. Q. et al. Fluoroethylene carbonate additives to render uniform Li deposits in lithium metal batteries. Adv. Funct. Mater. 27, 1605989 (2017).
Aurbach, D. et al. On the surface chemical aspects of very high energy density, rechargeable Li–sulfur batteries. J. Electrochem. Soc. 156, A694–A702 (2009).
Liu, Y. et al. Solubility-mediated sustained release enabling nitrate additive in carbonate electrolytes for stable lithium metal anode. Nat. Commun. 9, 3656 (2018).
Zhang, X. Q. et al. Highly stable lithium metal batteries enabled by regulating the solvation of lithium ions in nonaqueous electrolytes. Angew. Chem. Int. Ed. 57, 5301–5305 (2018).
Ramasubramanian, A. et al. Lithium diffusion mechanism through solid–electrolyte interphase in rechargeable lithium batteries. J. Phys. Chem. C. 123, 10237–10245 (2019).
Chen, S. et al. High-voltage lithium-metal batteries enabled by localized high-concentration electrolytes. Adv. Mater. 30, 1706102 (2018).
Wang, H. et al. Dual-solvent Li-ion solvation enables high-performance Li-metal batteries. Adv. Mater. 33, 2008619 (2021).
Cao, X. et al. Monolithic solid–electrolyte interphases formed in fluorinated orthoformate-based electrolytes minimize Li depletion and pulverization. Nat. Energy 4, 796–805 (2019).
Aurbach, D. Review of selected electrode–solution interactions which determine the performance of Li and Li ion batteries. J. Power Sources 89, 206–218 (2000).
Zhang, Z. et al. Operando electrochemical atomic force microscopy of solid–electrolyte interphase formation on graphite anodes: the evolution of SEI morphology and mechanical properties. ACS Appl. Mater. Interfaces 12, 35132–35141 (2020).
Gao, Y. & Zhang, B. Probing the mechanically stable solid electrolyte interphase and the implications in design strategies. Adv. Mater. 34, e2205421 (2022).
Zhang, D. et al. Atomic-resolution transmission electron microscopy of electron beam-sensitive crystalline materials. Science 359, 675–679 (2018).
Peled, E., Golodnitsky, D. & Ardel, G. Advanced model for solid electrolyte interphase electrodes in liquid and polymer electrolytes. J. Electrochem. Soc. 144, L208–L210 (1997).
Weissermel, K. et al. Polymerization of trioxane. Angew. Chem. Int. Ed. 6, 526–533 (1967).
Xu, K. Electrolytes and interphases in Li-ion batteries and beyond. Chem. Rev. 114, 11503–11618 (2014).
Aurbach, D., Youngman, O. & Dan, P. The electrochemical behavior of 1,3-dioxolane—LiClO4 solutions—II. contaminated solutions. Electrochim. Acta 35, 639–655 (1990).
Greene, J. P. in Automotive Plastics and Composites Ch. 8 (William Andrew Publishing, 2021).
Han, J. et al. Calculated reduction potentials of electrolyte species in lithium–sulfur batteries. J. Phys. Chem. C. 124, 20654–20670 (2020).
Wang, Y. et al. Theoretical studies to understand surface chemistry on carbon anodes for lithium-ion batteries: how does vinylene carbonate play its role as an electrolyte additive? J. Am. Chem. Soc. 124, 4408–4421 (2002).
Zhang, X. Q. et al. Regulating anions in the solvation sheath of lithium ions for stable lithium metal batteries. ACS Energy Lett. 4, 411–416 (2019).
Deng, X. et al. Morphology and modulus evolution of graphite anode in lithium ion battery: an in situ AFM investigation. Sci. China Chem. 57, 178–183 (2014).
Zhang, Z. et al. Characterizing batteries by in situ electrochemical atomic force microscopy: a critical review. Adv. Energy Mater. 11, 2101518 (2021).
Lu, Y. et al. The timescale identification decoupling complicated kinetic processes in lithium batteries. Joule 6, 1172–1198 (2022).
Wan, T. H., Saccoccio, M., Chen, C. & Ciucci, F. Influence of the discretization methods on the distribution of relaxation times deconvolution: implementing radial basis functions with DRTtools. Electrochim. Acta 184, 483–499 (2015).
Zhao, Q. et al. Upgrading carbonate electrolytes for ultra-stable practical lithium metal batteries. Angew. Chem. Int. Ed. 61, e202116214 (2022).
Niu, C. et al. High-energy lithium metal pouch cells with limited anode swelling and long stable cycles. Nat. Energy 4, 551–559 (2019).
Qiao, Y. et al. A high-energy-density and long-life initial-anode-free lithium battery enabled by a Li2O sacrificial agent. Nat. Energy 6, 653–662 (2021).
Gao, Y. et al. Effect of the supergravity on the formation and cycle life of non-aqueous lithium metal batteries. Nat. Commun. 13, 5 (2022).
Niu, C. et al. Balancing interfacial reactions to achieve long cycle life in high-energy lithium metal batteries. Nat. Energy 6, 723–732 (2021).
Zhang, L. et al. Practical 4.4 V Li||NCM811 batteries enabled by a thermal stable and HF free carbonate-based electrolyte. Nano Energy 96, 107122 (2022).
He, B. et al. Scalable fabrication of a large-area lithium/graphene anode towards a long-life 350 W h kg−1 lithium metal pouch cell. J. Mater. Chem. A 9, 25558–25566 (2021).
Tang, Y. et al. Metal carbodiimides-derived organic–inorganic interface protective layer for practical high energy lithium metal batteries. J. Power Sources 536, 231479 (2022).
Becke, A. D. Density-functional thermochemistry. III. the role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993).
Frisch, M. J. et al. Gaussian 09 Revision A.02 (Gaussian Inc., 2009).
Marenich, A. V., Cramer, C. J. & Truhlar, D. G. Universal solvation model based on solute electron density and on a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J. Phys. Chem. B 113, 6378–6396 (2009).
Steve, P. Fast parallel algorithms for short-range molecular-dynamics. J. Comput. Phys. 117, 1–19 (1995).
Jorgensen, W. L., Maxwell, D. S. & Tirado-Rives, J. Development and testing of the OPLS all-atom force field on conformational energetics and properties of organic liquids. J. Am. Chem. Soc. 118, 11225–11236 (1996).
Schauperl, M. et al. Non-bonded force field model with advanced restrained electrostatic potential charges (RESP2). Commun. Chem. 3, 44 (2020).
Lu, T. & Chen, F.-W. Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem. 33, 580–592 (2012).
Jensen, K. P. & Jorgensen, W. L. Halide, ammonium, and alkali metal ion parameters for modeling aqueous solutions. J. Chem. Theory Comput. 2, 1499–1509 (2006).
Canongia Lopes, J. N. et al. Potential energy landscape of bis(fluorosulfonyl)amide. J. Phys. Chem. B 112, 9449–9455 (2008).
Momma, K. & Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 44, 1272–1276 (2011).
Acknowledgements
J.-Q.H. acknowledges support by the National Key Research and Development Program (2021YFB2400300 and 2021YFB2500300) and Beijing Natural Science Foundation (JQ20004). X.-Q.Z. acknowledges support by the National Natural Science Foundation of China (22209010), China Postdoctoral Science Foundation (2021M700404) and Beijing Institute of Technology Research Fund Program for Young Scholars. X.C. acknowledges support by National Natural Science Foundation of China (22109086). We also acknowledge the support from Tsinghua National Laboratory for Information Science and Technology for theoretical simulations. We thank G.-X. Liu, J.-X. Tian, S.-Y. Sun, X.-Q. Ding and Y.-Q. Li for their helpful discussions.
Author information
Authors and Affiliations
Contributions
J.-Q.H. and X.-Q.Z. conceived and designed the experiments. Q.-K.Z. assembled the coin cells and pouch cells. Q.-K.Z., X.-Q.Z., J.X., L.-P.H. and B.-Q.L. carried out material characterizations and electrochemical measurements. J.W. and R.W. performed in situ AFM characterization. T.-L.S carried out the ToF-SIMS tests. N.Y. and X.C. performed the MD simulations. J.-Q.H. supervised this project. All authors engaged in result discussions. X.-Q.Z, M.-Y.Z, H.-J.P., Q.Z. and J.-Q.H. co-wrote the paper with input from all authors.
Corresponding author
Ethics declarations
Competing interests
A patent related to the work has been submitted (application number CN202210942310.X) by Beijing Institute of Technology. The inventors are Jia-Qi Huang, Qian-Kui Zhang and Xue-Qiang Zhang. The patent refers to the methodology in this paper but provides more analogous additives than this work. The other authors declare no competing interests.
Peer review
Peer review information
Nature Energy thanks Chaojiang Niu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–33 and Tables 1–4.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, QK., Zhang, XQ., Wan, J. et al. Homogeneous and mechanically stable solid–electrolyte interphase enabled by trioxane-modulated electrolytes for lithium metal batteries. Nat Energy 8, 725–735 (2023). https://doi.org/10.1038/s41560-023-01275-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-023-01275-y
This article is cited by
-
Hybridizing carbonate and ether at molecular scales for high-energy and high-safety lithium metal batteries
Nature Communications (2024)
-
Molecular anchoring of free solvents for high-voltage and high-safety lithium metal batteries
Nature Communications (2024)
-
Bilayer solid-electrolyte interphase structure enables a 440 Wh kg−1 lithium metal pouch cell to achieve 130 cycles
Science China Chemistry (2024)
-
A review of solid-state lithium metal batteries through in-situ solidification
Science China Chemistry (2024)
-
Roadmap for rechargeable batteries: present and beyond
Science China Chemistry (2024)