Abstract
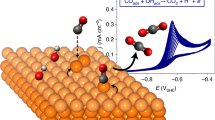
High-temperature CO2 electrolysers offer exceptionally efficient storage of renewable electricity in the form of CO and other chemical fuels, but conventional electrodes catalyse destructive carbon deposition. Ceria catalysts are known carbon inhibitors for fuel cell (oxidation) reactions; however, for more severe electrolysis (reduction) conditions, catalyst design strategies remain unclear. Here we establish the inhibition mechanism on ceria and show selective CO2 to CO conversion well beyond the thermodynamic carbon deposition threshold. Operando X-ray photoelectron spectroscopy during CO2 electrolysis—using thin-film model electrodes consisting of samarium-doped ceria, nickel and/or yttria-stabilized zirconia—together with density functional theory modelling, reveal the crucial role of oxidized carbon intermediates in preventing carbon build-up. Using these insights, we demonstrate stable electrochemical CO2 reduction with a scaled-up 16 cm2 ceria-based solid-oxide cell under conditions that rapidly destroy a nickel-based cell, leading to substantially improved device lifetime.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Data underlying the study can be found at Figshare57 (APXPS and cell testing) and https://www.catalysis-hub.org/publications/SkafteSelective2019 (DFT)58.
References
Jensen, S. H., Larsen, P. H. & Mogensen, M. Hydrogen and synthetic fuel production from renewable energy sources. Int. J. Hydrogen Energy 32, 3253–3257 (2007).
Graves, C., Ebbesen, S. D., Mogensen, M. & Lackner, K. S. Sustainable hydrocarbon fuels by recycling CO2 and H2O with renewable or nuclear energy. Renew. Sustain. Energy Rev. 15, 1–23 (2011).
Seh, Z. W. et al. Combining theory and experiment in electrocatalysis: insights into materials design. Science 355, eaad4998 (2017).
Davis, S. J. et al. Net-zero emissions energy systems. Science 360, eaas9793 (2018).
Bidrawn, F. et al. Efficient reduction of CO2 in a solid oxide electrolyzer. Electrochem. Solid State Lett. 11, B167 (2008).
Jensen, S. H. et al. Large-scale electricity storage utilizing reversible solid oxide cells combined with underground storage of CO2 and CH4. Energy Environ. Sci. 8, 2471–2479 (2015).
Graves, C., Ebbesen, S. D., Jensen, S. H., Simonsen, S. B. & Mogensen, M. B. Eliminating degradation in solid oxide electrochemical cells by reversible operation. Nat. Mater. 14, 239–244 (2015).
Hartvigsen, J., Elangovan, S., Elwell, J. & Larse, D. Oxygen production from Mars atmosphere carbon dioxide using solid oxide electrolysis. ECS Trans. 78, 2953–2963 (2017).
Skafte, T. L., Blennow, P., Hjelm, J. & Graves, C. Carbon deposition and sulfur poisoning during CO2 electrolysis in nickel-based solid oxide cell electrodes. J. Power Sources 373, 54–60 (2018).
Duboviks, V. et al. A Raman spectroscopic study of the carbon deposition mechanism on Ni/CGO electrodes during CO/CO2 electrolysis. Phys. Chem. Chem. Phys. 16, 13063 (2014).
Helveg, S. et al. Atomic-scale imaging of carbon nanofibre growth. Nature 427, 426–429 (2004).
Tao, Y., Ebbesen, S. D. & Mogensen, M. B. Carbon deposition in solid oxide cells during co-electrolysis of H2O and CO2. J. Electrochem. Soc. 161, F337–F343 (2014).
Navasa, M., Frandsen, H. L., Skafte, T. L., Sundén, B. & Graves, C. Localized carbon deposition in solid oxide electrolysis cells studied by multiphysics modeling. J. Power Sources 394, 102–113 (2018).
Homel, M., Gür, T. M., Koh, J. H. & Virkar, A. V. Carbon monoxide-fueled solid oxide fuel cell. J. Power Sources 195, 6367–6372 (2010).
Boldrin, P. et al. Strategies for carbon and sulfur tolerant solid oxide fuel cell materials, incorporating lessons from heterogeneous catalysis. Chem. Rev. 116, 13633–13684 (2016).
Murray, E. P., Tsai, T. & Barnett, S. A. A direct-methane fuel cell with a ceria-based anode. Nature 400, 649–651 (1999).
Park, S., Vohs, J. M. & Gorte, R. J. Direct oxidation of hydrocarbons in a solid-oxide fuel cell. Nature 404, 265–267 (2000).
Yang, L. et al. Enhanced sulfur and coking tolerance of a mixed ion conductor for SOFCs: BaZr0.1Ce0.7Y0.2-xYbxO3-δ. Science 326, 126–129 (2009).
Li, X. et al. In situ probing of the mechanisms of coking resistance on catalyst-modified anodes for solid oxide fuel cells. Chem. Mater. 27, 822–828 (2015).
Choi, Y., Brown, E. C., Haile, S. M. & Jung, W. Electrochemically modified, robust solid oxide fuel cell anode for direct-hydrocarbon utilization. Nano Energy 23, 161–171 (2016).
Skafte, T. L., Sudireddy, B. R., Blennow, P. & Graves, C. Carbon and redox tolerant infiltrated oxide fuel-electrodes for solid oxide cells. ECS Trans. 72, 201–214 (2016).
Irvine, J. T. S. et al. Evolution of the electrochemical interface in high-temperature fuel cells and electrolysers. Nat. Energy 1, 15014 (2016).
Rostrup-Nielsen, J. R. & Alstrup, I. Innovation and science in the process industry: steam reforming and hydrogenolysis. Catal. Today 53, 311–316 (1999).
Mortensen, P. M. & Dybkjær, I. Industrial scale experience on steam reforming of CO2-rich gas. Appl. Catal. A 495, 141–151 (2015).
Guo, J. et al. Improving the carbon resistance of Ni-based steam reforming catalyst by alloying with Rh: a computational study coupled with reforming experiments and EXAFS characterization. ACS Catal. 1, 574–582 (2011).
Nikolla, E., Schwank, J. & Linic, S. Hydrocarbon steam reforming on Ni alloys at solid oxide fuel cell operating conditions. Catal. Today 136, 243–248 (2008).
Feng, Za, Machala, M. L. & Chueh, W. C. Surface electrochemistry of CO2 reduction and CO oxidation on Sm-doped CeO2-x: coupling between Ce3+ and carbonate adsorbates. Phys. Chem. Chem. Phys. 17, 12273–12281 (2015).
Yu, Y. et al. CO2 activation and carbonate intermediates: an operando AP-XPS study of CO2 electrolysis reactions on solid oxide electrochemical cells. Phys. Chem. Chem. Phys. 16, 11633–11639 (2014).
Cheng, Z., Sherman, B. J. & Lo, C. S. Carbon dioxide activation and dissociation on ceria (110): a density functional theory study. J. Chem. Phys. 138, 014702 (2013).
Zhang, C. et al. Measuring fundamental properties in operating solid oxide electrochemical cells by using in situ X-ray photoelectron spectroscopy. Nat. Mater. 9, 944–949 (2010).
Opitz, A. K. et al. Surface chemistry of perovskite-type electrodes during high temperature CO2 electrolysis investigated by operando photoelectron spectroscopy. ACS Appl. Mater. Interfaces 9, 35847–35860 (2017).
Chueh, W. C., Hao, Y., Jung, W. & Haile, S. M. High electrochemical activity of the oxide phase in model ceria–Pt and ceria–Ni composite anodes. Nat. Mater. 11, 155–161 (2012).
Graves, C., Martinez, L. & Sudireddy, B. R. High performance nano-ceria electrodes for solid oxide cells. ECS Trans. 72, 183–192 (2016).
Yu, Y. et al. Carbon deposits and Pt/YSZ overpotentials in CO/CO2 solid oxide electrochemical cells. ECS Trans. 57, 3119–3126 (2013).
Wang, J. et al. Threshold catalytic onset of carbon formation on CeO2during CO2 electrolysis: mechanism and inhibition. J. Mater. Chem. A 7, 15233–15243 (2019).
Morar, J. F. et al. C 1s excitation studies of diamond (111). I. Surface core levels. Phys. Rev. B 33, 1346–1349 (1986).
Haerle, R., Riedo, E., Pasquarello, A. & Baldereschi, A. sp2/sp3 hybridization ratio in amorphous carbon from C 1s core-level shifts: X-ray photoelectron spectroscopy and first-principles calculation. Phys. Rev. B 65, 045101 (2001).
Ermolieff, A. et al. XPS, Raman spectroscopy, X-ray diffraction, specular X-ray reflectivity, transmission electron microscopy and elastic recoil detection analysis of emissive carbon film characterization. Surf. Interface Anal. 31, 185–190 (2001).
Alzate-Restrepo, V. & Hill, J. M. Carbon deposition on Ni/YSZ anodes exposed to CO/H2 feeds. J. Power Sources 195, 1344–1351 (2010).
El Gabaly, F., McCarty, K. F., Bluhm, H. & McDaniel, A. H. Oxidation stages of Ni electrodes in solid oxide fuel cell environments. Phys. Chem. Chem. Phys. 15, 8334 (2013).
Mudiyanselage, K. et al. Importance of the metal-oxide interface in catalysis: in situ studies of the water-gas shift reaction by ambient-pressure X-ray photoelectron spectroscopy. Angew. Chem. Int. Ed. 52, 5101–5105 (2013).
Staudt, T. et al. Electronic structure of magnesia-ceria model catalysts, CO2 adsorption, and CO2 activation: a synchrotron radiation photoelectron spectroscopy study. J. Phys. Chem. C 115, 8716–8724 (2011).
Czekaj, I. et al. Characterization of surface processes at the Ni-based catalyst during the methanation of biomass-derived synthesis gas: X-ray photoelectron spectroscopy (XPS). Appl. Catal. A 329, 68–78 (2007).
Ming, H. et al. Large scale electrochemical synthesis of high quality carbon nanodots and their photocatalytic property. Dalton Trans. 41, 9526 (2012).
Paier, J., Penschke, C. & Sauer, J. Oxygen defects and surface chemistry of ceria: quantum chemical studies compared to experiment. Chem. Rev. 113, 3949–3985 (2013).
Vayssilov, G. N., Mihaylov, M., Petkov, P. S., Hadjiivanov, K. I. & Neyman, K. M. Reassignment of the vibrational spectra of carbonates, formates, and related surface species on ceria: a combined density functional and infrared spectroscopy investigation. J. Phys. Chem. C 115, 23435–23454 (2011).
Bengaard, H. S. et al. Steam reforming and graphite formation on Ni catalysts. J. Catal. 209, 365–384 (2002).
Grass, M. E. et al. New ambient pressure photoemission endstation at Advanced Light Source beamline 9.3.2. Rev. Sci. Instrum. 81, 053106 (2010).
Frank Ogletree, D., Bluhm, H., Hebenstreit, E. D. & Salmeron, M. Photoelectron spectroscopy under ambient pressure and temperature conditions. Nucl. Instrum. Methods Phys. Res. A 601, 151–160 (2009).
Whaley, Ja et al. Note: Fixture for characterizing electrochemical devices in-operando in traditional vacuum systems. Rev. Sci. Instrum. 81, 1–3 (2010).
Hwang, W., Kim, Y.-K. & Rudd, M. E. New model for electron-impact ionization cross sections of molecules. J. Chem. Phys. 104, 2956 (1996).
Balaji Gopal, C. et al. Equilibrium oxygen storage capacity of ultrathin CeO2-δ depends non-monotonically on large biaxial strain. Nat. Commun. 8, 15360 (2017).
Fabris, S., de Gironcoli, S., Baroni, S., Vicario, G. & Balducci, G. Reply to “Comment on ‘Taming multiple valency with density functionals: a case study of defective ceria’”. Phys. Rev. B 72, 237102 (2005).
Farra, R. et al. Promoted ceria: a structural, catalytic, and computational study. ACS Catal. 3, 2256–2268 (2013).
Nørskov, J. K., Studt, F., Abild-Pedersen, F. & Bligaard, T. Fundamental Concepts in Heterogeneous Catalysis (Wiley, 2014).
Jensen, S. H., Hauch, A., Hendriksen, P. V. & Mogensen, M. Advanced test method of solid oxide cells in a plug-flow setup. J. Electrochem. Soc. 156, B757 (2009).
Skafte, T. L. et al. Data for the publication “Selective high-temperature CO2 electrolysis enabled by oxidized carbon intermediates”. Figshare https://doi.org/10.11583/DTU.4479050 (2019).
Winther, K. T. et al. Catalysis-Hub.Org, an open electronic structure database for surface reactions. Sci. Data 6, 75 (2019).
Acknowledgements
We thank L. Zhang for SEM assistance, A. Lyck Smitshuysen for assistance with preparing the large-format cells, R. M. Ortiz de la Morena for 3D modelling and rendering the wind turbine in Fig. 5, and H. Bluhm for assistance at beamline 11.0.2. This research used resources of the Advanced Light Source, which is a US Department of Energy Office of Science User Facility, under contract no. DE-AC02-05CH11231. The authors gratefully acknowledge financial support from Haldor Topsoe A/S, Innovation Fund Denmark, the Danish Agency for Science, Technology and Innovation (grant no. 5176-00001B and 5176-00003B) and Energinet.dk under the project ForskEL 2014-1-12231. The work at Stanford was supported by the National Science Foundation CAREER Award (1455369). M.B. acknowledges support from the US Department of Energy, Chemical Sciences, Geosciences, and Biosciences (CSGB) Division of the Office of Basic Energy Sciences, via grant DE-AC02-76SF00515 to the SUNCAT Center for Interface Science and Catalysis. We thank J. Nørskov and T. Bligaard at the SUNCAT Center for Interface Science and Catalysis for hosting T.L.S. and C.G. All calculations in this work were performed with the use of the computer time allocation (m2997) at the National Energy Research Scientific Computing Center, a DOE Office of Science User Facility supported by the Office of Science of the US Department of Energy under contract no. DE-AC02-05CH11231.
Author information
Authors and Affiliations
Contributions
T.L.S., C.G. and W.C.C. designed the experiments. T.L.S. carried out the spectroscopic and electrochemical analysis. M.L.M., L.M., E.S., S.S. and T.L.S. manufactured samples. T.L.S., Z.G. and C.G. carried out preliminary experiments and sample characterization. Z.G., T.L.S., C.B.G., M.M., C.G., M.L.M. and E.J.C. carried out the XPS experiments. M.B. and M.G.-M. designed and conducted the DFT calculations. T.L.S. and C.G. carried out the large-format cell experiments. T.L.S., C.G., W.C.C., M.B., M.G.-M., J.A.G.T., Z.G. and M.M. contributed to writing the article. C.G. initiated the collaborative project. W.C.C. and C.G. supervised and guided the work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Notes 1–3, Figs. 1–21, Tables 1–5 and refs.
Rights and permissions
About this article
Cite this article
Skafte, T.L., Guan, Z., Machala, M.L. et al. Selective high-temperature CO2 electrolysis enabled by oxidized carbon intermediates. Nat Energy 4, 846–855 (2019). https://doi.org/10.1038/s41560-019-0457-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-019-0457-4
This article is cited by
-
Efficient multicarbon formation in acidic CO2 reduction via tandem electrocatalysis
Nature Nanotechnology (2024)
-
Carbon-efficient carbon dioxide electrolysers
Nature Sustainability (2022)
-
Efficient electrosynthesis of n-propanol from carbon monoxide using a Ag–Ru–Cu catalyst
Nature Energy (2022)
-
Structural transformations of solid electrocatalysts and photocatalysts
Nature Reviews Chemistry (2021)
-
Solid Oxide Electrolysis of H2O and CO2 to Produce Hydrogen and Low-Carbon Fuels
Electrochemical Energy Reviews (2021)