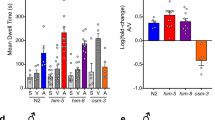
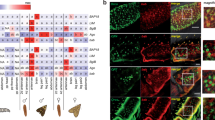
Abstract
Sex differentiation and hormones are essential for the development of sexual signals in animals, and the regulation of sexual signals involves complex gene networks. However, it is unknown whether a core gene is able to connect the upstream regulators for controlling sexual signal outputs and behavioural consequences. Here, we identify a single gene that integrates both sex differentiation and hormone signalling with sexual attractiveness in an insect model. CYP4PC1 in the German cockroach, Blattella germanica, controls the rate-limiting step in producing female-specific contact sex pheromone (CSP) that stimulates male courtship. As revealed by behavioural, biochemical, molecular, genetic and bioinformatic approaches, in sexually mature females, CYP4PC1 expression and CSP production are coordinately induced by sex differentiation genes and juvenile hormone (JH) signalling. In adult males, direct inhibition of CYP4PC1 expression by doublesexM binding in gene promoter and lack of the gonadotropic hormone JH prevent CSP production, thus avoiding male–male attraction. By manipulating the upstream regulators, we show that wild-type males prefer to court cockroaches with higher CYP4PC1 expression and CSP production in a dose-dependent manner, regardless of their sex. These findings shed light on how sex-specific and high sexual attractiveness is conferred in insects.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data and materials generated in this study are available in the main text, supplementary materials and source data. RNA-seq data have been deposited to National Center for Biotechnology Information’s Sequence Read Archive database under accession numbers PRJNA759392 and PRJNA760811. Source data are provided with this paper.
References
Ryan, M. J. Darwin, sexual selection, and the brain. Proc. Natl Acad. Sci. USA 118, e2008194118 (2021).
Le Moëne, O. & Ågmo, A. The neuroendocrinology of sexual attraction. Front. Neuroendocrinol. 51, 46–67 (2018).
Witchel, S. F. Disorders of sex development. Best Pract. Res. Clin. Obstet. Gynaecol. 48, 90–102 (2018).
Mäkelä, J., Koskenniemi, J. J., Virtanen, H. E. & Toppari, J. Testis development. Endocr. Rev. 40, 857–905 (2019).
Bilen, J., Atallah, J., Azanchi, R., Levine, J. D. & Riddiford, L. M. Regulation of onset of female mating and sex pheromone production by juvenile hormone in Drosophila melanogaster. Proc. Natl Acad. Sci. USA 110, 18321–18326 (2013).
Auer, T. O. & Benton, R. Sexual circuitry in Drosophila. Curr. Opin. Neurobiol. 38, 18–26 (2016).
Schal, C., Fan, Y. & Blomquist, G. J. in Insect Pheromone Biochemistry and Molecular Biology (eds Blomquist, G. J. & Vogt, R. G.) 283–322 (Elsevier Academic Press, 2003).
Eliyahu, D., Nojima, S., Mori, K. & Schal, C. New contact sex pheromone components of the German cockroach, Blattella germanica, predicted from the proposed biosynthetic pathway. J. Chem. Ecol. 34, 229–237 (2008).
Nojima, S., Schal, C., Webster, F. X., Santangelo, R. G. & Roelofs, W. L. Identification of the sex pheromone of the German cockroach, Blattella germanica. Science 307, 1104–1106 (2005).
Mori, K. Synthesis of all the six components of the female-produced contact sex pheromone of the German cockroach, Blattella germanica (L.). Tetrahedron 64, 4060–4071 (2008).
Pei, X.-J. et al. Modulation of fatty acid elongation in cockroaches sustains sexually dimorphic hydrocarbons and female attractiveness. PLoS Biol. 19, e3001330 (2021).
Nishida, R., Fukami, H. & Ishii, S. Sex pheromone of the German cockroach (Blattella germanica L.) responsible for male wing-raising: 3,11-dimethyl-2-nonacosanone. Experientia 30, 978–979 (1974).
Chase, J., Touhara, K., Prestwich, G. D., Schal, C. & Blomquist, G. J. Biosynthesis and endocrine control of the production of the German cockroach sex pheromone 3,11-dimethylnonacosan-2-one. Proc. Natl Acad. Sci. USA 89, 6050–6054 (1992).
Harrison, M. C. et al. Hemimetabolous genomes reveal molecular basis of termite eusociality. Nat. Ecol. Evol. 2, 557–566 (2018).
Gu, X., Quilici, D., Juarez, P., Blomquist, G. J. & Schal, C. Biosynthesis of hydrocarbons and contact sex pheromone and their transport by lipophorin in females of the German cockroach (Blattella germanica). J. Insect Physiol. 41, 257–267 (1995).
Chen, N., Pei, X., Li, S., Fan, Y.-L. & Liu, T.-X. Involvement of integument-rich CYP4G19 in hydrocarbon biosynthesis and cuticular penetration resistance in Blattella germanica (L.). Pest Manag. Sci. 76, 215–226 (2020).
Roy, S., Saha, T. T., Zou, Z. & Raikhel, A. S. Regulatory pathways controlling female insect reproduction. Annu. Rev. Entomol. 63, 489–511 (2018).
Li, S. et al. The genomic and functional landscapes of developmental plasticity in the American cockroach. Nat. Commun. 9, 1008 (2018).
Zhu, S. et al. Insulin/IGF signaling and TORC1 promote vitellogenesis via inducing juvenile hormone biosynthesis in the American cockroach. Development. 147, dev188805 (2020).
Luo, W. et al. Juvenile hormone signaling promotes ovulation and maintains egg shape by inducing expression of extracellular matrix genes. Proc. Natl Acad. Sci. USA 118, e2014461118 (2021).
Tillman, J. A., Seybold, S. J., Jurenka, R. A. & Blomquist, G. J. Insect pheromones—an overview of biosynthesis and endocrine regulation. Insect Biochem. Mol. Biol. 29, 481–514 (1999).
Jindra, M., Palli, S. R. & Riddiford, L. M. The juvenile hormone signaling pathway in insect development. Annu. Rev. Entomol. 58, 181–241 (2013).
Piulachs, M. D., Maestro, J. L. & Belles, X. Juvenile hormone production and accessory gland development during sexual maturation of male Blattella germanica (L.) (Dictyoptera: Blattellidae). Comp. Biochem. Physiol. A 102, 477–480 (1992).
Ferveur, J. F. et al. Genetic feminization of pheromones and its behavioral consequences in Drosophila males. Science 276, 1555–1558 (1997).
Shirangi, T., Dufour, H., Williams, T. & Carroll, S. Rapid evolution of sex pheromone-producing enzyme expression in Drosophila. PLoS Biol. 7, e1000168 (2009).
Verhulst, E. C., de Zande, L. & Beukeboom, L. W. Insect sex determination: it all evolves around transformer. Curr. Opin. Genet. Dev. 20, 376–383 (2010).
Yamamoto, D. & Koganezawa, M. Genes and circuits of courtship behaviour in Drosophila males. Nat. Rev. Neurosci. 14, 681–692 (2013).
Wexler, J. et al. Hemimetabolous insects elucidate the origin of sexual development via alternative splicing. eLife 8, e47490 (2019).
Clynen, E., Ciudad, L., Belles, X. & Piulachs, M.-D. Conservation of fruitless’ role as master regulator of male courtship behaviour from cockroaches to flies. Dev. Genes Evol. 221, 43–48 (2011).
Defelipea, L. A. et al. Juvenile hormone synthesis: ‘esterify then epoxidize’ or ‘epoxidize then esterify’? Insights from the structural characterization of juvenile hormone acid methyltransferase. Insect Biochem. Mol. Biol. 41, 228–235 (2011).
Fan, Y., Zurek, L., Dykstra, M. J. & Schal, C. Hydrocarbon synthesis by enzymatically dissociated oenocytes of the abdominal integument of the German cockroach, Blattella germanica. Naturwissenschaften 90, 121–126 (2003).
Beach, F. A. Sexual attractivity, proceptivity, and receptivity in female mammals. Horm. Behav. 7, 105–138 (1976).
Lozano, J. & Belles, X. Conserved repressive function of Krüppel homolog 1 on insect metamorphosis in hemimetabolous and holometabolous species. Sci. Rep. 1, 163 (2011).
Lozano, J. & Belles, X. Role of methoprene-tolerant (Met) in adult morphogenesis and in adult ecdysis of Blattella germanica. PLoS ONE 9, e103614 (2014).
Tian, L. et al. 20-hydroxyecdysone upregulates Atg genes to induce autophagy in the Bombyx fat body. Autophagy 9, 1172–1187 (2013).
Jia, Q. et al. Juvenile hormone and 20-hydroxyecdysone coordinately control the developmental timing of matrix metalloproteinase-induced fat body cell dissociation. J. Biol. Chem. 292, 21504–21516 (2017).
Eliyahu, D., Nojima, S., Mori, K. & Schal, C. Jail baits: how and why nymphs mimic adult females of the German cockroach, Blattella germanica. Anim. Behav. 78, 1097–1105 (2009).
Schal, C., Burns, E. L., Jurenka, R. A. & Blomquist, G. J. A new component of the female sex pheromone of Blattella germanica (L.) (Dictyoptera: Blattellidae) and interaction with other pheromone components. J. Chem. Ecol. 16, 1997–2008 (1990).
Acknowledgements
We thank D. Nelson for naming of CYP4PC1, D. Huang for preparing the cockroach cartoon and Z. Wang and X. Yi for assistance on Python and R software. We are grateful to W. Wang, Q. Feng, X. He, C.X. Zhang and S. Zhan for their comments and suggestions to improve the manuscript. This work was supported by the National Natural Science Foundation of China (grant nos. 31930014 to S.L., 32100378 to N.C. and 31772533 to Y.-L.F.), the Department of Science and Technology in Guangdong Province (grant nos. 2019B090905003 and 2019A0102006 to S.L.), the Laboratory of Lingnan Modern Agriculture Project (grant no. NT2021003 to S.L.), the Shenzhen Science and Technology Program (grant no. KQTD20180411143628272 to S.L.) and the China Postdoctoral Science Foundation (grant no. 2019M652941 to N.C.).
Author information
Authors and Affiliations
Contributions
Conceptualization was done by S.L., N.C., Y.-L.F. and C.S. The methodology was developed by N.C., Y.-J.L., M.-T.L. and N.L. The investigation was carried out by N.C., Y.-J.L., X.-J.P., Y.Y. and J.Z. Visualization was done by N.C. and Y.-J.L. Funding was acquired by S.L., Y.-L.F. and N.C. Project administration was the responsibility of N.L. Supervision was done by S.L., Y.-L.F., G.W., Y.P. and C.S. The original draft was written by N.C. and Y.-L.F. Review and editing of the draft was done by S.L., C.S., Y.P., G.W. and T.-X.L.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Ecology & Evolution thanks Xavier Belles and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Schematic diagram of the CSP biosynthetic pathway in Blattella germanica.
A blend of six structurally related CSP components (in black dashed box) share a common biosynthetic pathway, in which the methyl ketones are formed by age- and sex-specific hydroxylation (in red text, the rate-limiting step possibly mediated by a cytochrome P450 system) of the 3,11-dimethyl alkane precursors and subsequent oxidation13. LCFA, long-chain fatty acid.
Extended Data Fig. 2 Screening of the female-specific CYP4PC1 by RNA-seq and its expression profiling.
a, Distribution of cuticular C29 methyl ketone titre in sexually mature females. The antennae and abdominal integument (in red text and bar) were used for RNA-seq analyses. n = 3 biological replicates. Different letters indicate statistically significant differences between groups using Welch’s ANOVA (Games-Howell multiple comparisons test, P < 0.05). Data are mean ± s.e.m. b, The numbers of total CYP genes identified in the antennae and abdominal integument, and those upregulated in sexually mature females compared to males. The FPKM values of the up-regulated CYPs are available in Supplementary Table 1, 2. c, Genomic organization of CYP4PC1 and characteristic P450 motifs identified from the deduced protein sequence. The numbers nearby indicate the length of each exon or intron (base pair, bp), and conserved motifs typical of P450 are marked with colour-filled boxes. d, Developmental patterns of CYP4PC1 expression in various tissues across sexual maturation. n = 4 biological replicates. Data are mean ± s.e.m. e, Gel electrophoresis verification of a 1,619 bp fragment containing the complete coding sequence of CYP4PC1, as amplified from various tissues by RT-PCR. A 104 bp fragment of actin-5c was used as a loading control. M, DNA marker. -, negative control with no DNA template. f, Western blotting validation of the CYP4PC1 antibody using antennal protein extracts and RNAi experiment. The red frame indicates immune signals of the endogenous CYP4PC1 protein, with a predicted molecular weight of 58 kDa. M, protein marker. The lane of protein markers was spliced from the white light image of the same membrane. g, Dose-dependent CYP4PC1 protein level in various tissues resulted from a gradient of total protein loaded, as determined by western blotting.
Extended Data Fig. 3 CYP4PC1 controls methyl ketone CSP production and female sexual attractiveness.
a, RNAi efficiency in various tissues of adult females after two injections of dsRNA, each on day 1 and day 3, as determined by qPCR on day 5. n = 4 biological replicates. b, C27 and C29 methyl ketone CSP production resulted from CYP4PC1 knockdown in adult females. n = 7 biological replicates. c, Effect of CYP4PC1 knockdown in adult females on the wing-raising percentage and the latency of wing-raising in WT males. The numbers of tested males from eight behavioural replicates are indicated at the bottom of the bars. n = 26 cockroaches per treatment for the right panel. The solid line and dashed lines within each violin plot indicate median and quartiles, respectively. P values were determined by two-tailed unpaired t-test. Data in all bar plots are mean ± s.e.m.
Extended Data Fig. 4 CYP4G19 modulates CSP production by affecting the biosynthesis of hydrocarbon precursors.
a, RNAi knockdown of CYP4G19 from N6 causes a depletion at the protein level (inset) and downregulation in cuticular hydrocarbons in adult females. The bands of tubulin were spliced from those of dsMuslta- and dsCYP4G19-injected groups, each with three lanes, on the same membrane. IS, internal standard of n-hexacosane. Similar results were obtained from six biological replicates. b, Effects of CYP4G19 knockdown on dimethyl alkane and methyl ketone production. n = 6 biological replicates. c, Tissue distribution of two other CYP gene expressions in adult females and the effect of their knockdown on CSP production. n = 4 (gene relative expression) and 6 (methyl ketone production) biological replicates. P values were determined by two-tailed unpaired t-test (b, c). Data in bar plots are mean ± s.e.m.
Extended Data Fig. 5 JH signalling regulates the expression of CYP4PC1 but not CYP4G19.
a, Developmental similarity of Kr-h1 and CYP4PC1 expression across N6 and the first vitellogenic cycle. Inset of the right panel indicates a magnification of CYP4PC1 expression throughout N6. n = 4 biological replicates. b, c, Effect of JH III treatment in adult females on Kr-h1 expression in the antennae and abdominal integument (b), and CYP4G19 expression in the abdominal integument (c). n = 4 biological replicates. d, RNAi knockdown of Met or Kr-h1 in adult females is efficient in the abdominal integument. n = 4 biological replicates. e, Kr-h1 relative expression in the antennae from females with Kr-h1 knockdown and those with both dsKr-h1 and JH III treatments. n = 4 biological replicates. f, JH III application in adult males causes substantial transcription of CYP4PC1 in the antennae. The complete coding sequence of CYP4PC1 was detected in the antennae. P were determined by two-tailed unpaired t-test (b–d). Different letters indicate significant differences using one-way ANOVA (Tukey HSD multiple comparisons test, P < 0.05) (e). Data in a–e are mean ± s.e.m.
Extended Data Fig. 6 The tra-dsx sex differentiation pathway promotes CYP4PC1 expression in females.
a, A diagram showing dsx splicing isoforms in B. germanica males and females. The modified diagram is based on a previous study28. All the primers used for dsRNA synthesis, qPCR analysis, and RT-PCR are summarized in Supplementary Table 3. b, Knockdown of dsxF (dsx2 and 3) results in a depletion of dsx2 but not dsx3. c, Knockdown of tra in adult females affects the alternative splicing between dsxF and dsxM. d, Effect of double knockdown of both tra and dsx in adult females on dsx isoforms. P values were determined by two-tailed unpaired t-test (b–d). Data in bar plots are mean ± s.e.m from four biological replicates.
Extended Data Fig. 7 DsxM represses CYP4PC1 expression in males.
a, Knockdown of dsx in adult males promotes CYP4PC1 transcript. The complete coding sequence of CYP4PC1 was detected in the antennae of dsDsx-treated adult males. b, Effect of double knockdown of both dsxM and CYP4PC1 in adult males on dsx isoforms. c, Knockdown of tra in adult females or dsx in adult males has no effect on CYP4G19 protein level. d, Effect of fru knockdown in adult males on dsxM and CYP4PC1 expressions. n = 4 biological replicates. P values were determined by two-tailed unpaired t-test. Data are mean ± s.e.m.
Extended Data Fig. 8 CYP4PC1 integrates sex differentiation and hormone signalling pathways and controls sexual attractiveness in a dose-dependent manner.
a, Curve fitting between CYP4PC1 expression and CSP production, and CSP production and male courtship index. The 95%CI is shown in grey. The source data are derived from groups 1–5 and 7 in Fig. 6a. Data are mean ± s.d. b, A schematic summary showing the regulatory network of CYP4PC1 expression. Such a mechanism ensures female-specific CYP4PC1 expression (dsx-dependent) and high level CSP production upon sexual maturation (JH-dependent).
Supplementary information
Supplementary Tables
Supplementary Table 1. FPKM values of CYP genes identified from the antennal RNA-seq data. The CYPs showing significant upregulation in sexually mature females compared to males are marked in bold, and the most promising candidate gene is marked in red. Supplementary Table 2. FPKM values of CYP genes identified from the RNA-seq data of abdominal integument. The CYPs showing upregulation, regardless of statistical significance, in sexually mature females compared to males are marked in bold, and the most promising candidate gene is marked in red. Supplementary Table 3. Summary of primers and oligonucleotide probes used in this study.
Supplementary Video 1
Video clip showing a typical courtship behaviour in a WT male elicited by dsMuslta (RNAi control) female. On contact of the male antennae with a sexually mature female (primarily the antennae), the male displays a typical courtship behaviour. This behaviour includes a 180° rotation away from the female followed by a rapid wing-raising and extension of his abdomen, thereby exposing the specialized tergal gland towards the female’s head.
Supplementary Video 2
Video clip shows that knockdown of CYP4PC1 in females results in compromised sexual behaviour in WT males. The male contacts the female’s antennae but does not engage in courtship, showing a negative response to the female.
Supplementary Video 3
Video clip showing no courtship response of a WT male to a dsCYP4PC1 female, whereas a typical courtship is elicited later by a dsMuslta female. The WT male was allowed to contact a dsCYP4PC1 female three times, and after three observations of no courtship wing-raising response, a dsMuslta female was introduced into the dish. Courtship in the WT male was only elicited by the dsMuslta female and not by the dsCYP4PC1 female.
Supplementary Video 4
Video clip showing sexual attraction of JH III-treated male to WT male. Adult males received three applications of JH III, each on days 1, 3 and 5, and a behavioural assay was performed on day 7. Of the JH III-treated males, 27.5% elicited wing-raising displays in WT males.
Supplementary Video 5
Video clip showing homosexual behaviours among dsDsx-treated adult males. Adult males received two injections of dsDsx, each on day 0 and day 3. The injected males began to raise their wings to each other, feed on the tergal secretions and attempted mating on day 5, exhibiting typical sexual behaviours normally displayed in male–female interactions.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 1
Unprocessed western blots.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 2
Unprocessed western blots.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 3
Unprocessed western blots.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 4
Unprocessed western blots and EMSA.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 5
Unprocessed western blots.
Source Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 2
Unprocessed western blots and gels.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 4
Unprocessed western blots
Source Data Extended Data Fig. 5
Statistical source data.
Source Data Extended Data Fig. 5
Unprocessed gels.
Source Data Extended Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 6
Unprocessed gels.
Source Data Extended Data Fig. 7
Statistical source data.
Source Data Extended Data Fig. 7
Unprocessed western blots and gels.
Rights and permissions
About this article
Cite this article
Chen, N., Liu, YJ., Fan, YL. et al. A single gene integrates sex and hormone regulators into sexual attractiveness. Nat Ecol Evol 6, 1180–1190 (2022). https://doi.org/10.1038/s41559-022-01808-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-022-01808-w
This article is cited by
-
Two sex pheromone receptors for sexual communication in the American cockroach
Science China Life Sciences (2024)
-
Egfr signaling promotes juvenile hormone biosynthesis in the German cockroach
BMC Biology (2022)
-
The sex of scents
Nature Ecology & Evolution (2022)