Abstract
Marine protected areas (MPAs) play a leading role in conserving and restoring marine environments. MPAs can benefit both marine populations within their boundaries and external populations owing to a net export of organisms (spillover). However, little is known about variation in performance within MPAs. For example, edge effects may degrade populations within MPAs close to their boundaries. Here we synthesize empirical estimates of 72 taxa of fish and invertebrates to explore spatial patterns across the borders of 27 no-take MPAs. We show that there is a prominent and consistent edge effect that extends approximately 1 km within the MPA, in which population sizes on the border are 60% smaller than those in the core area. Our analysis of cross-boundary population trends suggests that, globally, the smallest 64% of no-take MPAs (those of less than 10 km2 in area) may hold only about half (45–56%) of the population size that is implied by their area. MPAs with buffer zones did not display edge effects, suggesting that extending no-take areas beyond the target habitats and managing fishing activities around MPA borders are critical for boosting MPA performance.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All sources of data analysed in this meta-analysis are reported in Supplementary Table 1. The dataset generated during the current study is available at https://doi.org/10.6084/m9.figshare.14578344.v1
Code availability
The code generated during the current study and all relevant datasets for analysis are available in the GitHub repository (https://github.com/sarahohayon/A-meta-analysis-reveals-edge-effects-within-marine-protected-areas).
References
Costello, M. J. & Ballantine, B. Biodiversity conservation should focus on no-take marine reserves: 94% of marine protected areas allow fishing. Trends Ecol. Evol. 30, 507–509 (2015).
Edgar, G. J. et al. Global conservation outcomes depend on marine protected areas with five key features. Nature 506, 216–220 (2014).
Giakoumi, S. et al. Ecological effects of full and partial protection in the crowded Mediterranean Sea: a regional meta-analysis. Sci. Rep. 7, 8940 (2017).
Sala, E. & Giakoumi, S. No-take marine reserves are the most effective protected areas in the ocean. ICES J. Mar. Sci. 75, 1166–1168 (2018).
Woodroffe, R. & Ginsberg, J. R. Edge effects and the extinction of populations inside protected areas. Science 280, 2126–2128 (1998).
Hansen, A. J. & Defries, R. Ecological mechanisms linking protected areas to surrounding lands. Ecol. Appl. 17, 974–988 (2016).
Roberts, C. M., Halpern, B., Palumbi, S. R. & Warner, R. R. Designing marine reserve networks. Why small, isolated protected areas are not enough. Conserv. Pract. 2, 10–17 (2001).
Walters, C. Impacts of dispersal, ecological interactions, and fishing effort dynamics on efficacy of marine protected areas: how large should protected areas be? Bull. Mar. Sci. 66, 745–757 (2000).
Kramer, D. L. & Chapman, M. R. Implications of fish home range size and relocation for marine reserve function. Environ. Biol. Fishes 55, 65–79 (1999).
Guidetti, P. et al. Large-scale assessment of Mediterranean marine protected areas effects on fish assemblages. PLoS One 9, e91841 (2014).
Lester, S. E. et al. Biological effects within no-take marine reserves: a global synthesis. Mar. Ecol. Prog. Ser. 384, 33–46 (2009).
Di Lorenzo, M., Claudet, J. & Guidetti, P. Spillover from marine protected areas to adjacent fisheries has an ecological and a fishery component. J. Nat. Conserv. 32, 62–66 (2016).
Harmelin-Vivien, M. et al. Gradients of abundance and biomass across reserve boundaries in six Mediterranean marine protected areas: evidence of fish spillover? Biol. Conserv. 141, 1829–1839 (2008).
Abesamis, R. A. & Russ, G. R. Density-dependent spillover from a marine reserve: long-term evidence. Ecol. Appl. 15, 1798–1812 (2005).
Murawski, S. A., Wigley, S. E., Fogarty, M. J., Rago, P. J. & Mountain, D. G. Effort distribution and catch patterns adjacent to temperate MPAs. ICES J. Mar. Sci. 62, 1150–1167 (2005).
Kellner, J. B., Tetreault, I., Gaines, S. D. & Nisbet, R. M. Fishing the line near marine reserves in single and multispecies fisheries. Ecol. Appl. 17, 1039–1054 (2007).
Stelzenmüller, V. et al. Spatial assessment of fishing effort around European marine reserves: implications for successful fisheries management. Mar. Pollut. Bull. 56, 2018–2026 (2008).
Halpern, B. S., Lester, S. E. & Kellner, J. B. Spillover from marine reserves and the replenishment of fished stocks. Environ. Conserv. 36, 268–276 (2010).
Newmark, W. D. Isolation of African protected areas. Front. Ecol. Environ. 6, 321–328 (2008).
Defries, R., Hansen, A., Newton, A. C. & Hansen, M. C. Increasing isolation of protected areas in tropical forests over the past twenty years. Ecol. Appl. 15, 19–26 (2005).
MPAtlas (Marine Conservation Institute, accessed 4 July 2020); http://www.mpatlas.org
Willis, T. J., Millar, R. B., Babcock, R. C. & Tolimieri, N. Burdens of evidence and the benefits of marine reserves: putting Descartes before des horse? Environ. Conserv. 30, 97–103 (2003).
Roberts, C. M. et al. Application of ecological criteria in selecting marine reserves and developing reserve networks. Ecol. Appl. 13, 215–228 (2003).
Huntington, B. E., Karnauskas, M., Babcock, E. A. & Lirman, D. Untangling natural seascape variation from marine reserve effects using a landscape approach. PLoS One 5, e12327 (2010).
Miller, K. I. & Russ, G. R. Studies of no-take marine reserves: methods for differentiating reserve and habitat effects. Ocean Coast. Manag. 96, 51–60 (2014).
Gill, D. A. et al. Capacity shortfalls hinder the performance of marine protected areas globally. Nature 543, 665–671 (2017).
Brill, G. C. & Raemaekers, S. J. P. N. A decade of illegal fishing in Table Mountain National Park (2000–2009): trends in the illicit harvest of abalone Haliotis midae and West Coast rock lobster Jasus lalandii. African. J. Mar. Sci. 35, 491–500 (2013).
Harasti, D., Davis, T. R., Jordan, A., Erskine, L. & Moltschaniwskyj, N. Illegal recreational fishing causes a decline in a fishery targeted species (snapper: Chrysophrys auratus) within a remote no-take marine protected area. PLoS One 14, e0209926 (2019).
Kleiven, P. J. N. et al. Fishing pressure impacts the abundance gradient of European lobsters across the borders of a newly established marine protected area. Proc. R. Soc. B Biol. Sci. 286, 20182455 (2019).
Simpson, S. D. et al. Anthropogenic noise increases fish mortality by predation. Nat. Commun. 7, 10544 (2016).
Sarà, G. et al. Effect of boat noise on the behavior of bluefin tuna Thunnus thynnus in the Mediterranean Sea. Mar. Ecol. Prog. Ser. 331, 243–253 (2007).
Tran, D. S. C., Langel, K. A., Thomas, M. J. & Blumstein, D. T. Spearfishing-induced behavioral changes of an unharvested species inside and outside a marine protected area. Curr. Zool. 62, 39–44 (2016).
Jiao, J., Pilyugin, S. S., Riotte-Lambert, L. & Osenberg, C. W. Habitat-dependent movement rate can determine the efficacy of marine protected areas. Ecology 99, 2485–2495 (2018).
Potts, J. R., Hillen, T. & Lewis, M. A. The ‘edge effect’ phenomenon: deriving population abundance patterns from individual animal movement decisions. Theor. Ecol. 9, 233–247 (2016).
Gerber, L. R. et al. Population models for marine reserve design: a retrospective and prospective synthesis. Ecol. Appl. 13, 47–64 (2003).
Malvadkar, U. & Hastings, A.Persistence of mobile species in marine protected areas. Fish. Res. 91, 69–78 (2008).
Di Lorenzo, M., Guidetti, P., Calò, A. & Claudet, J. Assessing spillover from marine protected areas and its drivers: a meta-analytical approach. Fish Fish. 21, 906–915 (2020).
Goñi, R., Quetglas, A. & Reñones, O. Spillover of spiny lobsters Palinurus elephas from a marine reserve to an adjoining fishery. Mar. Ecol. Prog. Ser. 308, 207–219 (2006).
Stamoulis, K. A. & Friedlander, A. M. A seascape approach to investigating fish spillover across a marine protected area boundary in Hawai’i. Fish. Res. 144, 2–14 (2013).
Protected Planet: the World Database on Protected Areas (WDPA) (UNEP-WCMC and IUCN, accessed July 2020); http://www.protectedplanet.net
Kulbicki, M. et al. Global biogeography of reef fishes: a hierarchical quantitative delineation of regions. PLoS One 8, e81847 (2013).
Parravicini, V. et al. Global patterns and predictors of tropical reef fish species richness. Ecography 36, 1254–1262 (2013).
Froese, R. & Pauly, D. (eds). FishBase (accessed May 2020); http://www.fishbase.org
Hedges, L. V., Gurevitch, J. & Curtis, P. S. The meta-analysis of response ratios in experimental ecology. Ecology 80, 1150–1156 (1999).
Wood, S. N. Generalized Additive Models: An Introduction with R 2nd edn (CRC Press, 2017).
Harrison, X. A. et al. A brief introduction to mixed effects modelling and multi-model inference in ecology. PeerJ 6, e4794 (2018).
R Core Team. R: a language and environment for statistical computing. v.3.6.1 (2019).
QGIS Geographic Information System. Open Source Geospatial Foundation Project (QGIS Development Team, 2020); http://qgis.osgeo.org
Harmelin-Vivien, M. et al. Species richness, abundance and biomass data for assessing fish spillover from Mediterranean marine protected areas. SEANOE https://doi.org/10.17882/74396 (2020).
Acknowledgements
We are grateful to M. Harmelin-Vivien for assisting with access to the BIOMEX-Spillover project database49, to B. Pike from MPAtlas21 for providing access to their database and to D. Shapiro-Goldberg for revising the manuscript. S.O. was partially supported by the Israeli Nature and Parks Authority.
Author information
Authors and Affiliations
Contributions
S.O. conceived the study, formulated the research question, conducted the literature review, collected the data, performed the meta-analyses and wrote the early draft and final version of the manuscript; I.G. provided guidance on the meta-analyses, performed complementary analyses and contributed to the writing of the manuscript; J.B. conceived the study, participated in the formulation of the research question, revised the manuscript and supervised the work. All authors gave final approval for publication and agree to be held accountable for the work performed herein.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Ecology & Evolution thanks Tessa Mazor, Nils Krueck, Kristian Metcalfe and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 PRISMA flow diagram describing the review process for studies suitable for the meta-analysis.
The diagram provides details on the steps we took to select studies for inclusion in the meta analysis. We initially identified 974 records. The final number of studies included after screening and eligibility assessment was 24.
Extended Data Fig. 2 Spatial pattern of population sizes with distance from MPA borders (with data points superimposed on the plots).
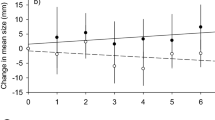
a, Spatial pattern across MPA borders for the entire dataset (22 km inside the MPAs to 27 km in fished areas; n = 1,619, ΔAICc = 257.9, DE = 45.8%), b, spatial pattern across MPA borders zoomed in to distances between −3 km to 6 km, c, spatial pattern with analysis restricted to within the MPAs for the entire available range (n = 594, ΔAICc = 53.9, DE = 44.9%), d, spatial pattern with analysis restricted to within the MPAs, zoomed in to distances between −3 km to 0 (MPA border). The decline in population size starts approximately 1.0–1.5 km inside the MPAs, indicating a strong edge effect. The y axes are effect size (calculated as log of each data point relative to the innermost value reported within the MPA, for each sample) and the x axes are distance (m) from MPA border. The blue line represents the MPAs border, with negative numbers indicating distances within the MPAs and positive numbers distances in fished areas. The red dashed line represents the edge-effect distance, where the estimated effect size drops below zero, that is, biomass/density/CPUE begin to decrease relative to the innermost value). The tics along the x axes represent data point locations. Grey shaded area around the curve denotes 95% confidence interval.
Extended Data Fig. 3 Sensitivity analysis for the spatial pattern of population sizes across MPA borders.
a, GAM model for distance subset data,−3km inside MPAs to 6 km in fished areas (n = 1,484, k = 39, ΔAICc = 378.15, DE = 58.5% when using distance from MPA border compared to a reduced model excluding distance), b, analysis displaying underlying data points, c, GAM model with k = 20 (ΔAICc =366.39, DE = 57.9%), d, GAM model with k = 50 (ΔAICc =379.13, DE = 58.5%), (c) GAM model with different smooth term for distance: bs = ’tp’ (ΔAICc =360.95, DE = 57.8%), e, GAM model with no weighting parameter (ΔAICc =325.49, DE = 57.2%). Under all these analyses a clear edge effect is still noticeable.
Extended Data Fig. 4 Validation tests for the spatial pattern of population sizes across MPA borders.
a, A model with original population size estimates (density/biomass/CPUE, log-transformed) serving as the response variables, b, a model with relative X axis, where distance from the MPA border is standardized to the innermost location for each unique sample. Under both analyses a clear edge effect is still noticeable.
Extended Data Fig. 5 Spatial pattern of population sizes across MPA borders as a function of habitat variables.
The separate effect of (a) habitat type (coral reef: 12 MPAs, 107 samples, n = 1,024; temperate rocky reef: 14 MPAs, 39 samples, n = 439; ΔAICc = 13.45, DE = 59.4%), b, habitat continuity (habitat is continuous: 25 MPAs, 135 samples, n = 1,387; habitat is non-continuous: 2 MPAs, 12 samples, n = 97; ΔAICc = 3.36, DE = 59.4%), c, quantitative control for habitat quality (quantitative control performed: 13 studies, 19 MPAs, 78 samples, n = 727; no quantitative control: 11 studies, 11 MPAs, 69 samples, n = 757; ΔAICc = 29.17, DE = 59.8%).
Extended Data Fig. 6 Spatial pattern of population sizes with distance from MPA borders as a function of MPA and taxa characteristics (with data points superimposed on the plots).
The separate effect of (a) commercial value (commercial taxa: 124 samples, n = 1,248; non-commercial taxa: 23 samples, n = 236), b, taxa (fish: 125 samples, n = 1,190; invertebrates: 22 samples, n = 294) (c) protection level (low: 54 samples, n = 483; high: 93 samples, n = 1,001), and d, buffer zone (without buffer zone: 106 samples, n = 1,220; with buffer zone: 41 samples, n = 264), on the spatial patterns of marine population sizes across MPA borders. The y axes are effect size (calculated as log of each data point relative to the innermost value reported within the MPA, for each sampling set) and the x axes are distance (m) from MPA border. Blue vertical lines represent the MPA borders with negative numbers indicating distances within the MPAs and positive numbers distances within fished areas. Analysis was performed on a reduced dataset, where the majority of data was concentrated (6 km outside the MPA border and 3 km inside the MPA, resulting in n = 1,484) in order to improve model performance. Shaded area around the model curves denotes 95% confidence intervals.
Extended Data Fig. 7 Spatial pattern of population sizes across MPA borders as a function of their mobility level.
a, Sessile taxa (Mollusca; 8 samples, n = 76) display a limited edge effect of ~ 150 m, b, sedentary taxa (lobsters and low-mobility fish such as Serranidae, Pomacentridae, Chaetodontidae; 16 samples, n = 257) display a deeper edge effect of ~ 400 m, c, mobile taxa display a large edge effect reaching ~1,500 m into the MPA (mobile fish; 100 samples, n = 915). The limited sample size for sessile taxa produces high uncertainty in the estimates. The analysis is restricted to the commercial taxa dataset (124 samples, n = 1,248, ΔAICc = 53.83, DE = 63.8%) to remove the masking created by the spatial patterns associated with the non-commercial taxa.
Extended Data Fig. 8 Spatial pattern of commercial fish populations across MPA borders as a function of MPA age or size.
The separate effect of a, MPA age (young: age = < 10 years, 52 samples, n = 399; old: age >10 years, 56 samples, n = 594; ΔAICc = 7.7, DE = 57.7%), b, MPA size (small: size < 3 km2, 65 samples, n = 735; large: size > 3 km2, 43 samples, n = 258; ΔAICc = 5.9, DE = 57%). Young MPAs present shorter edge-effect distance compared to old MPAs, but with similar magnitude of population size reduction. Similarly, small MPAs present shorter edge-effect distance compared to large MPAs with similar magnitude of population size reduction. The analysis is restricted to the commercial fish dataset (containing 108 samples, n = 993) to avoid the confounding effect stemming from the spatial patterns associated with taxa identity and commercial value.
Extended Data Fig. 9 The relationship between the magnitude of the edge effect and MPA size or age.
a, MPA size (estimate: 0.17, t-value: 0.47), and b, MPA age (estimate: 1.7, t-value: 30.19). The response variable (Y axis) is the linear slope of the relationship between the effect size and distance. When the magnitude of the effect size (total reduction in population size) is similar, a larger edge effect will be manifested as a shallower slope. Data was restricted to (1) within the MPA and until -3,000 m, (2) sets that have at least 3 data points, (3) at least one of the data points for each set is located >0.5 km inside the MPA, (4) fish with commercial value. Analyses are based on linear models weighted by the inverse of the estimated variance of the slope. In all cases, MPA name was added as a random effect.
Supplementary information
Rights and permissions
About this article
Cite this article
Ohayon, S., Granot, I. & Belmaker, J. A meta-analysis reveals edge effects within marine protected areas. Nat Ecol Evol 5, 1301–1308 (2021). https://doi.org/10.1038/s41559-021-01502-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-021-01502-3
This article is cited by
-
A decade-long connectivity study of Permit (Trachinotus falcatus) in Florida supports a spatial management approach
Environmental Biology of Fishes (2023)
-
Natural Protected Areas effect on the cover change rate of mangrove forests in the Yucatan Peninsula, Mexico
Wetlands (2023)
-
Sharing goals by timely communication improves fishermen's satisfaction with marine protected areas: A case study in the Mediterranean
Ambio (2022)
-
Marine conservation across protected area boundaries
Nature Ecology & Evolution (2021)