Abstract
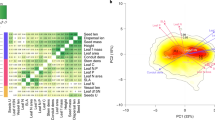
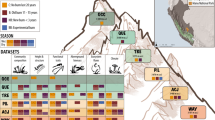
A fundamental assumption in trait-based ecology is that relationships between traits and environmental conditions are globally consistent. We use field-quantified microclimate and soil data to explore if trait–environment relationships are generalizable across plant communities and spatial scales. We collected data from 6,720 plots and 217 species across four distinct tundra regions from both hemispheres. We combined these data with over 76,000 database trait records to relate local plant community trait composition to broad gradients of key environmental drivers: soil moisture, soil temperature, soil pH and potential solar radiation. Results revealed strong, consistent trait–environment relationships across Arctic and Antarctic regions. This indicates that the detected relationships are transferable between tundra plant communities also when fine-scale environmental heterogeneity is accounted for, and that variation in local conditions heavily influences both structural and leaf economic traits. Our results strengthen the biological and mechanistic basis for climate change impact predictions of vulnerable high-latitude ecosystems.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The data are deposited in the Zenodo public repository115 at https://doi.org/10.5281/zenodo.4362216.
Code availability
The code is deposited in the Zenodo public repository115 at https://doi.org/10.5281/zenodo.4362216.
References
Shipley, B. et al. Reinforcing loose foundation stones in trait-based plant ecology. Oecologia 180, 923–931 (2016).
McGill, B. J., Enquist, B. J., Weiher, E. & Westoby, M. Rebuilding community ecology from functional traits. Trends Ecol. Evol. 21, 178–185 (2006).
Vellend, M. Conceptual synthesis in community ecology. Q. Rev. Biol. 85, 183–206 (2010).
Bjorkman, A. D. et al. Plant functional trait change across a warming tundra biome. Nature 562, 57–62 (2018).
Billings, W. D. Arctic and Alpine vegetations: similarities, differences, and susceptibility to disturbance. BioScience 23, 697–704 (1973).
Graae, B. J. et al. Stay or go – how topographic complexity influences alpine plant population and community responses to climate change. Perspect. Plant Ecol. Evol. Syst. 30, 41–50 (2018).
Bruelheide, H. et al. Global trait–environment relationships of plant communities. Nat. Ecol. Evol. 2, 1906–1917 (2018).
Choler, P. Consistent shifts in alpine plant traits along a mesotopographical gradient. Arct. Antarct. Alp. Res. 37, 444–453 (2005).
Wullschleger, S. D. et al. Plant functional types in Earth system models: past experiences and future directions for application of dynamic vegetation models in high-latitude ecosystems. Ann. Bot. 114, 1–16 (2014).
Pearson, R. G. et al. Shifts in Arctic vegetation and associated feedbacks under climate change. Nat. Clim. Change 3, 673–677 (2013).
Myers-Smith, I. H., Thomas, H. J. D. & Bjorkman, A. D. Plant traits inform predictions of tundra responses to global change. New Phytol. 221, 1742–1748 (2019).
Robinson, S. A. et al. Rapid change in East Antarctic terrestrial vegetation in response to regional drying. Nat. Clim. Change 8, 879–884 (2018).
Post, E. et al. Ecological dynamics across the Arctic associated with recent climate change. Science 325, 1355–1358 (2009).
Saros, J. E. et al. Arctic climate shifts drive rapid ecosystem responses across the West Greenland landscape. Environ. Res. Lett. 14, 074027 (2019).
Lavorel, S. & Garnier, E. Predicting changes in community composition and ecosystem functioning from plant traits: revisiting the Holy Grail. Funct. Ecol. 16, 545–556 (2002).
Chapin, F. S. III et al. Consequences of changing biodiversity. Nature 405, 234–242 (2000).
Díaz, S. et al. The global spectrum of plant form and function. Nature 529, 167–171 (2016).
Wright, I. J. et al. The worldwide leaf economics spectrum. Nature 428, 821–827 (2004).
Thomas, H. J. D. et al. Global plant trait relationships extend to the climatic extremes of the tundra biome. Nat. Commun. 11, 1351 (2020).
Billings, W. D. & Bliss, L. C. An alpine snowbank environment and its effects on vegetation, plant development, and productivity. Ecology 40, 388–397 (1959).
Myers-Smith, I. H. & Hik, D. S. Shrub canopies influence soil temperatures but not nutrient dynamics: an experimental test of tundra snow–shrub interactions. Ecol. Evol. 3, 3683–3700 (2013).
Chapin, F. S. III et al. Role of land-surface changes in Arctic summer warming. Science 310, 657–660 (2005).
Cahoon, S. M. P. et al. Interactions among shrub cover and the soil microclimate may determine future Arctic carbon budgets. Ecol. Lett. 15, 1415–1422 (2012).
Reich, P. B. The world-wide ‘fast–slow’ plant economics spectrum: a traits manifesto. J. Ecol. 102, 275–301 (2014).
Diaz, S. et al. The plant traits that drive ecosystems: evidence from three continents. J. Veg. Sci. 15, 295–304 (2004).
Cornelissen, J. H. C. et al. Global negative vegetation feedback to climate warming responses of leaf litter decomposition rates in cold biomes. Ecol. Lett. 10, 619–627 (2007).
Steinbauer, M. J. et al. Accelerated increase in plant species richness on mountain summits is linked to warming. Nature 556, 231–234 (2018).
Myers-Smith, I. H. et al. Climate sensitivity of shrub growth across the tundra biome. Nat. Clim. Change 5, 887–891 (2015).
Post, E. et al. The polar regions in a 2 °C warmer world. Sci. Adv. 5, eaaw9883 (2019).
IPCC Special Report on Global Warming of 1.5 °C (eds Masson-Delmotte, V. et al.) (WMO, 2018).
Bintanja, R. & Andry, O. Towards a rain-dominated Arctic. Nat. Clim. Change 7, 263–267 (2017).
Bromwich, D. H. et al. Central West Antarctica among the most rapidly warming regions on Earth. Nat. Geosci. 6, 139–145 (2013).
Turner, J. et al. Absence of 21st century warming on Antarctic Peninsula consistent with natural variability. Nature 535, 411–415 (2016).
Sonesson, M., Wielgolaski, F. E. & Kallio, P. in Fennoscandian Tundra Ecosystems. Ecological Studies (Analysis and Synthesis) Vol. 16 (ed. Wielgolaski, F. E.) 3–28 (Springer, 1975); https://doi.org/10.1007/978-3-642-80937-8_1
Niittynen, P., Heikkinen, R. K. & Luoto, M. Snow cover is a neglected driver of Arctic biodiversity loss. Nat. Clim. Change 8, 997–1001 (2018).
Klikoff, L. G. Photosynthetic response to temperature and moisture stress of three timberline meadow species. Ecology 46, 516–517 (1965).
Oberbauer, S. F. & Billings, W. D. Drought tolerance and water use by plants along an alpine topographic gradient. Oecologia 50, 325–331 (1981).
Eskelinen, A., Stark, S. & Männistö, M. Links between plant community composition, soil organic matter quality and microbial communities in contrasting tundra habitats. Oecologia 161, 113–123 (2009).
Ernakovich, J. G. et al. Predicted responses of Arctic and alpine ecosystems to altered seasonality under climate change. Glob. Change Biol. 20, 3256–3269 (2014).
Galen, C. & Stanton, M. L. Responses of snowbed plant species to changes in growing-season length. Ecology 76, 1546–1557 (1995).
Starr, G., Oberbauer, S. F. & Ahlquist, L. E. The photosynthetic response of Alaskan tundra plants to increased season length and soil warming. Arct. Antarct. Alp. Res. 40, 181–191 (2008).
Happonen, K. et al. Snow is an important control of plant community functional composition in oroarctic tundra. Oecologia 191, 601–608 (2019).
Niittynen, P. & Luoto, M. The importance of snow in species distribution models of Arctic vegetation. Ecography 41, 1024–1037 (2018).
le Roux, P. C., Aalto, J. & Luoto, M. Soil moisture’s underestimated role in climate change impact modelling in low-energy systems. Glob. Change Biol. 19, 2965–2975 (2013).
Lembrechts, J. J. et al. SoilTemp: a global database of near-surface temperature. Glob. Change Biol. 26, 6616–6629 (2020).
Bjorkman, A. D. et al. Tundra Trait Team: a database of plant traits spanning the tundra biome. Glob. Ecol. Biogeogr. 27, 1402–1411 (2018).
Maitner, B. S. et al. The bien r package: a tool to access the Botanical Information and Ecology Network (BIEN) database. Methods Ecol. Evol. 9, 373–379 (2018).
Kattge, J. et al. TRY - a global database of plant traits. Glob. Change Biol. 17, 2905–2935 (2011).
Pedersen, E. J., Miller, D. L., Simpson, G. L. & Ross, N. Hierarchical generalized additive models in ecology: an introduction with mgcv. PeerJ 7, e6876 (2019).
Niittynen, P. et al. Fine-scale tundra vegetation patterns are strongly related to winter thermal conditions. Nat. Clim. Change 10, 1143–1148 (2020).
Belluau, M. & Shipley, B. Predicting habitat affinities of herbaceous dicots to soil wetness based on physiological traits of drought tolerance. Ann. Bot. 119, 1073–1084 (2017).
Kemppinen, J., Niittynen, P., Riihimäki, H. & Luoto, M. Modelling soil moisture in a high-latitude landscape using LiDAR and soil data. Earth Surf. Proc. Land. 43, 1019–1031 (2018).
Kemppinen, J., Niittynen, P., Aalto, J., le Roux, P. C. & Luoto, M. Water as a resource, stress and disturbance shaping tundra vegetation. Oikos 128, 811–822 (2019).
Giblin, A. E., Nadelhoffer, K. J., Shaver, G. R., Laundre, J. A. & McKerrow, A. J. Biogeochemical diversity along a riverside toposequence in Arctic Alaska. Ecol. Monogr. 61, 415–435 (1991).
le Roux, P. C., Virtanen, R. & Luoto, M. Geomorphological disturbance is necessary for predicting fine-scale species distributions. Ecography 36, 800–808 (2013).
Finger Higgens, R., Hicks Pries, C. & Virginia, R. A. Trade-offs between wood and leaf production in Arctic shrubs along a temperature and moisture gradient in West Greenland. Ecosystems https://doi.org/10.1007/s10021-020-00541-4 (2020).
Porporato, A. & Rodriguez-Iturbe, I. Ecohydrology-a challenging multidisciplinary research perspective / Ecohydrologie: une perspective stimulante de recherche multidisciplinaire. Hydrol. Sci. J. 47, 811–821 (2002).
Legates, D. R. et al. Soil moisture: a central and unifying theme in physical geography. Prog. Phys. Geogr. 35, 65–86 (2011).
McLaughlin, B. C. et al. Hydrologic refugia, plants, and climate change. Glob. Change Biol. 23, 2941–2961 (2017).
Choler, P. Winter soil temperature dependence of alpine plant distribution: implications for anticipating vegetation changes under a warming climate. Perspect. Plant Ecol. Evol. Syst. 30, 6–15 (2018).
Happonen, K. et al. Snow is an important control of plant community functional composition in oroarctic tundra. Oecologia 191, 601–608 (2019).
Doran, P. T. et al. Antarctic climate cooling and terrestrial ecosystem response. Nature 415, 517–520 (2002).
French, D. D. & Smith, V. R. A comparison between Northern and Southern Hemisphere tundras and related ecosystems. Polar Biol. 5, 5–21 (1985).
le Roux, P. C. in The Prince Edward Islands: Land–Sea Interactions in a Changing Ecosystem (eds Chown, S. L. & Froneman, P. W.) 39–64 (African Sun Media, 2008).
Devau, N., Le Cadre, E., Jaillarda, B. & Gérarda, F. Soil pH controls the environmental availability of phosphorus: experimental and mechanistic modelling approaches. Appl. Geochem. 24, 2163–2174 (2009).
Stevens, R. J., Laughlin, R. J. & Malone, J. P. Soil pH affects the processes reducing nitrate to nitrous oxide and di-nitrogen. Soil Biol. Biochem. 30, 1119–1126 (1998).
Freschet, G. T., Cornelissen, J. H. C., Van Logtestijn, R. S. P. & Aerts, R. Evidence of the ‘plant economics spectrum’ in a subarctic flora. J. Ecol. 98, 362–373 (2010).
Bergholz, K. et al. Fertilization affects the establishment ability of species differing in seed mass via direct nutrient addition and indirect competition effects. Oikos 124, 1547–1554 (2015).
Curtin, D., Campbell, C. A. & Jalil, A. Effects of acidity on mineralization: pH-dependence of organic matter mineralization in weakly acidic soils. Soil Biol. Biochem. 30, 57–64 (1998).
Blondeel, H. et al. Light and warming drive forest understorey community development in different environments. Glob. Change Biol. 26, 1681–1696 (2020).
Dahlgren, J. P., Eriksson, O., Bolmgren, K., Strindell, M. & Ehrlén, J. Specific leaf area as a superior predictor of changes in field layer abundance during forest succession. J. Veg. Sci. 17, 577–582 (2006).
Lembrechts, J. J. et al. Comparing temperature data sources for use in species distribution models: from in‐situ logging to remote sensing. Glob. Ecol. Biogeogr. 28, 1578–1596 (2019).
Körner, C. & Hiltbrunner, E. The 90 ways to describe plant temperature. Perspect. Plant Ecol. Evol. Syst. 30, 16–21 (2018).
Maclean, I. M. D. Predicting future climate at high spatial and temporal resolution. Glob. Change Biol. 26, 1003–1011 (2019).
Aalto, J., Scherrer, D., Lenoir, J., Guisan, A. & Luoto, M. Biogeophysical controls on soil–atmosphere thermal differences: implications on warming Arctic ecosystems. Environ. Res. Lett. 13, 074003 (2018).
Aalto, J., le Roux, P. C. & Luoto, M. Vegetation mediates soil temperature and moisture in Arctic-alpine environments. Arct. Antarct. Alp. Res. 45, 429–439 (2013).
Moles, A. T. et al. Which is a better predictor of plant traits: temperature or precipitation? J. Veg. Sci. 25, 1167–1180 (2014).
Taylor, R. V. & Seastedt, T. R. Short- and long-term patterns of soil moisture in alpine tundra. Arct. Alp. Res. 26, 14–20 (1994).
Lembrechts, J. J. & Lenoir, J. Microclimatic conditions anywhere at any time! Glob. Change Biol. https://doi.org/10.1111/gcb.14942 (2019).
Zellweger, F., De Frenne, P., Lenoir, J., Rocchini, D. & Coomes, D. Advances in microclimate ecology arising from remote sensing. Trends Ecol. Evol. 34, 327–341 (2019).
Bramer, I. et al. Advances in monitoring and modelling climate at ecologically relevant scales. Adv. Ecol. Res. 58, 101–161 (2018).
Halbritter, A. H. et al. The handbook for standardized field and laboratory measurements in terrestrial climate change experiments and observational studies (ClimEx). Methods Ecol. Evol. 2, 16147 (2019).
Wild, J. et al. Climate at ecologically relevant scales: a new temperature and soil moisture logger for long-term microclimate measurement. Agr. Forest Meteorol. 268, 40–47 (2019).
Aalto, J., Riihimäki, H., Meineri, E., Hylander, K. & Luoto, M. Revealing topoclimatic heterogeneity using meteorological station data. Int. J. Climatol. 37, 544–556 (2017).
Kearney, M. R., Gillingham, P. K., Bramer, I., Duffy, J. P. & Maclean, I. M. D. A method for computing hourly, historical, terrain‐corrected microclimate anywhere on Earth. Methods Ecol. Evol. https://doi.org/10.1111/2041-210x.13330 (2019).
Bjorkman, A. D. et al. Status and trends in Arctic vegetation: evidence from experimental warming and long-term monitoring. Ambio 49, 678–692 (2020).
Vandvik, V., Halbritter, A. H. & Telford, R. J. Greening up the mountain. Proc. Natl Acad. Sci. USA 115, 833–835 (2018).
Bonfils, C. J. W. et al. On the influence of shrub height and expansion on northern high latitude climate. Environ. Res. Lett. 7, 015503 (2012).
Zwieback, S., Chang, Q., Marsh, P. & Berg, A. Shrub tundra ecohydrology: rainfall interception is a major component of the water balance. Environ. Res. Lett. 14, 055005 (2019).
Robinson, D. A. et al. Global environmental changes impact soil hydraulic functions through biophysical feedbacks. Glob. Change Biol. 25, 1895–1904 (2019).
Loranty, M. M. et al. Reviews and syntheses: changing ecosystem influences on soil thermal regimes in northern high-latitude permafrost regions. Biogeosciences 15, 5287–5313 (2018).
Parker, T. C., Subke, J.-A. & Wookey, P. A. Rapid carbon turnover beneath shrub and tree vegetation is associated with low soil carbon stocks at a subarctic treeline. Glob. Change Biol. 21, 2070–2081 (2015).
DeMarco, J., Mack, M. C. & Bret-Harte, M. S. Effects of Arctic shrub expansion on biophysical vs. biogeochemical drivers of litter decomposition. Ecology 95, 1861–1875 (2014).
Qian, H., Joseph, R. & Zeng, N. Enhanced terrestrial carbon uptake in the northern high latitudes in the 21st century from the Coupled Carbon Cycle Climate Model Intercomparison Project model projections. Glob. Change Biol. 16, 641–656 (2010).
Sistla, S. A. et al. Long-term warming restructures Arctic tundra without changing net soil carbon storage. Nature 497, 615–618 (2013).
Climate in Svalbard 2100 – A Knowledge Base for Climate Adaptation (Norwegian Centre for Climate Services, 2019); https://go.nature.com/3tFTKAr
Weather Observations from Greenland 1958–2018 - Observation Data with Description DMI Report 19-08 (Danish Meteorological Institute, 2019); https://go.nature.com/36RkdBk
Enontekiö Kilpisjärvi Saana. Daily Climate Observations (Finnish Meteorological Institute, 2019); https://en.ilmatieteenlaitos.fi/download-observations
Enontekiö Kilpisjärvi Kyläkeskus. Daily Climate Observations (Finnish Meteorological Institute, 2019); https://en.ilmatieteenlaitos.fi/download-observations
Smith, V. R. & Steenkamp, M. Classification of the terrestrial habitats on Marion Island based on vegetation and soil chemistry. J. Veg. Sci. 12, 181–198 (2001).
Beck, H. E. et al. Present and future Köppen–Geiger climate classification maps at 1-km resolution. Sci. Data 5, 180214 (2018).
Canadell, J. et al. Maximum rooting depth of vegetation types at the global scale. Oecologia 108, 583–595 (1996).
Iversen, C. M. et al. The unseen iceberg: plant roots in Arctic tundra. New Phytol. 205, 34–58 (2015).
Kern, R. et al. Comparative vegetation survey with focus on cryptogamic covers in the high Arctic along two differing catenas. Polar Biol. 42, 2131–2145 (2019).
Miller, R. O. & Kissel, D. E. Comparison of soil pH methods on soils of North America. Soil Sci. Soc. Am. J. 74, 310–316 (2010).
McCune, B. & Keon, D. Equations for potential annual direct incident radiation and heat load. J. Veg. Sci. 13, 603 (2002).
McCune, B. Improved estimates of incident radiation and heat load using non- parametric regression against topographic variables. J. Veg. Sci. 18, 751 (2007).
Karger, D. N. et al. Climatologies at high resolution for the earth’s land surface areas. Sci. Data 4, 170122 (2017).
NASA/METI/AIST/Japan Spacesystems, and U.S./Japan ASTER Science Team ASTER Global Digital Elevation Model (GDEM) V003 (NASA EOSDIS Land Processes DAAC, 2018); https://doi.org/10.5067/ASTER/ASTGTM.003
Hengl, T. et al. SoilGrids250m: global gridded soil information based on machine learning. PLoS ONE 12, e0169748 (2017).
Wood, S. N. Generalized Additive Models: An Introduction with R 2nd edn (Chapman and Hall/CRC, 2017).
R Core Team R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2018).
Husson, F., Le, S. & Pagès, J. Exploratory Multivariate Analysis by Example Using R (CRC, 2017).
Lê, S., Josse, J. & Husson, F. FactoMineR: an R package for multivariate analysis. J. Stat. Softw. 25, 31844 (2008).
Kemppinen, J. et al. Data from: Consistent trait–environment relationships within and across tundra plant communities. Zenodo https://doi.org/10.5281/zenodo.4362216 (2020).
Acknowledgements
We thank the past and present members of the BioGeoClimate Modelling Lab and the le Roux Lab for their hard work collecting the field data. We also thank the laboratory personnel at the University of Helsinki and University of Pretoria, as well as the staff at The University Centre in Svalbard, Kangerlussuaq International Support Services, Kilpisjärvi Biological research station and Marion Island field assistants (specifically E. Mostert, N. Mhlongo, J. van Berkel and J. Schoombie). We are also grateful to P. Eidesen for helping with the temperature loggers and E. Pedersen for his help regarding HGAMs. We thank H. Riihimäki for providing the drone image composition, on which the study grid vector is based in Fig. 1. J.K. was funded by the Doctoral Programme in Geosciences at the University of Helsinki, P.N. by the Kone Foundation, M.M. by the National Research Foundation via the SANAP programme and K.H. by the Doctoral Programme in Wildlife Biology Research at the University of Helsinki. The field campaigns were funded by the Academy of Finland (project numbers 307761 and 286950) and the National Research Foundation’s South African National Antarctic Program (unique grant numbers 93077 and 110726). We acknowledge the funding by the Finnish Ministry of Education and Culture (The FinCEAL Plus BRIDGES coordinated by the Finnish University Partnership for International Development). Permission to carry out fieldwork was granted by the Governor of Svalbard for the high-Arctic site, the Government of Greenland for the low-Arctic site, Metsähallitus for the sub-Arctic site and the Prince Edward Islands Management Committee (permit PEIMC1/2013) for the sub-Antarctic site.
Author information
Authors and Affiliations
Contributions
J.K. conceived the research together with M.L. J.K., P.N., P.C.L.R., J.A. and M.L. designed the study setting. J.K., P.N., P.C.L.R., M.M., J.A. and M.L. performed the field research and H.R. laboratory analyses. J.K., with support from K.H. and B.J.E., analysed the data. J.K. wrote the first version of the paper with support, comments and input from all other authors. All authors revised the paper based on peer review comments.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Ecology & Evolution thanks Sylvia Haider and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–4, Figs. 1–3 and Data 1–2.
Supplementary Table
A comprehensive summary and basic statistics of the trait data.
Rights and permissions
About this article
Cite this article
Kemppinen, J., Niittynen, P., le Roux, P.C. et al. Consistent trait–environment relationships within and across tundra plant communities. Nat Ecol Evol 5, 458–467 (2021). https://doi.org/10.1038/s41559-021-01396-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-021-01396-1
This article is cited by
-
Climate-induced tree-mortality pulses are obscured by broad-scale and long-term greening
Nature Ecology & Evolution (2024)
-
Relationships of stomatal morphology to the environment across plant communities
Nature Communications (2023)
-
Climate-trait relationships exhibit strong habitat specificity in plant communities across Europe
Nature Communications (2023)
-
Plant traits and associated data from a warming experiment, a seabird colony, and along elevation in Svalbard
Scientific Data (2023)