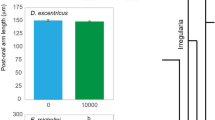
Abstract
Plasticity-first evolution (PFE) posits that novel features arise when selection refines pre-existing phenotypic plasticity into an adaptive phenotype. However, PFE is controversial because few tests have been conducted in natural populations. Here we present evidence that PFE fostered the origin of an evolutionary novelty that allowed certain amphibians to invade a new niche—a distinctive carnivore morph. We compared morphology, gene expression and growth of three species of spadefoot toad tadpoles when reared on alternative diets: Scaphiopus holbrookii, which (like most frogs) never produce carnivores; Spea multiplicata, which sometimes produce carnivores, but only through diet-induced plasticity; and Spea bombifrons, which often produce carnivores regardless of diet. Consistent with PFE, we found diet-induced plasticity—in morphology and gene expression—in Sc. holbrookii, adaptive refinement of this plasticity in Sp. multiplicata, and further refinement of the carnivore phenotype in Sp. bombifrons. Generally, phenotypic plasticity might play a significant, if underappreciated, role in evolutionary innovation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Mayr, E. in Evolution after Darwin Vol. 1 (ed. Tax, S.) 349–380 (Univ. Chicago Press, Chicago, 1959).
Wagner, G. P. & Lynch, V. J. Evolutionary novelties. Curr. Biol. 20, R48–R52 (2010).
Schwander, T. & Leimar, O. Genes as leaders and followers in evolution. Trends Ecol. Evol. 26, 143–151 (2011).
Santos, M. E., Le Bouquin, A., Crumière, A. J. J. & Khila, A. Taxon-restricted genes at the origin of a novel trait allowing access to a new environment. Science 358, 386–390 (2017).
West-Eberhard, M. J. Developmental Plasticity and Evolution (Oxford Univ. Press, New York, 2003).
Moczek, A. P. et al. The role of developmental plasticity in evolutionary innovation. Proc. R. Soc. B 278, 2705–2713 (2011).
Levis, N. A. & Pfennig, D. W. Evaluating ‘plasticity-first’ evolution in nature: key criteria and empirical approaches 31, 563–574. Trends Ecol. Evol. 31, 563–574 (2016).
Wray, G. A. et al. Does evolutionary theory need a rethink? No, all is well. Nature 514, 161–164 (2014).
Gilbert, S. F., Bosch, T. C. G. & Ledón-Rettig, C. Eco-Evo-Devo: developmental symbiosis and developmental plasticity as evolutionary agents. Nat. Rev. Genet. 16, 611–622 (2015).
Ghalambor, C. K., McKay, J. K., Carroll, S. P. & Reznick, D. N. Adaptive versus non-adaptive phenotypic plasticity and the potential for contemporary adaptation in new environments. Funct. Ecol. 21, 394–407 (2007).
Standen, E. M., Du, T. Y. & Larsson, H. C. Developmental plasticity and the origin of tetrapods. Nature 513, 54–58 (2014).
Ledón-Rettig, C. C., Pfennig, D. W. & Nascone-Yoder, N. Ancestral variation and the potential for genetic accommodation in larval amphibians: implications for the evolution of novel feeding strategies. Evol. Dev. 10, 316–325 (2008).
Kulkarni, S. S., Denver, R. J., Gomez-Mestre, I. & Buchholz, D. R. Genetic accommodation via modified endocrine signalling explains phenotypic divergence among spadefoot toad species. Nat. Commun. 8, 993 (2017).
Badyaev, A. V. Stress-induced variation in evolution: from behavioural plasticity to genetic assimilation. Proc. R. Soc. B 272, 877–886 (2005).
Rutherford, S. L. & Lindquist, S. Hsp90 as a capacitor for morphological evolution. Nature 396, 336–342 (1998).
Ghalambor, C. K. et al. Non-adaptive plasticity potentiates rapid adaptive evolution of gene expression in nature. Nature 525, 372–375 (2015).
Scheiner, S. M. Genetics and evolution of phenotypic plasticity. Annu. Rev. Ecol. Syst. 24, 35–68 (1993).
Scheiner, S. M. Selection experiments and the study of phenotypic plasticity. J. Evol. Biol. 15, 889–898 (2002).
Pfennig, D. W. Polyphenism in spadefoot toad tadpoles as a locally adjusted evolutionarily stable strategy. Evolution 46, 1408–1420 (1992).
Wells, K. D. The Ecology and Behavior of Amphibians (Univ. Chicago Press, Chicago, 2007).
Pfennig, D. W. The adaptive significance of an environmentally-cued developmental switch in an anuran tadpole. Oecologia 85, 101–107 (1990).
Levis, N. A., de la Serna Buzon, S. & Pfennig, D. W. An inducible offense: carnivore morph tadpoles induced by tadpole carnivory. Ecol. Evol. 5, 1405–1411 (2015).
Pfennig, D. W. & Murphy, P. J. How fluctuating competition and phenotypic plasticity mediate species divergence. Evolution 56, 1217–1228 (2002).
Pfennig, D. W. & Murphy, P. J. A test of alternative hypotheses for character divergence between coexisting species. Ecology 84, 1288–1297 (2003).
Pfennig, D. W. & Martin, R. A. Evolution of character displacement in spadefoot toads: different proximate mechanisms in different species. Evolution 64, 2331–2341 (2010).
Ledón-Rettig, C. C., Pfennig, D. W. & Crespi, E. J. Diet and hormonal manipulation reveal cryptic genetic variation: implications for the evolution of novel feeding strategies. Proc. R. Soc. B 277, 3569–3578 (2010).
Zeng, C., Gomez-Mestre, I. & Wiens, J. J. Evolution of rapid development in spadefoot toads is unrelated to arid environments. PLoS ONE 9, e96637 (2014).
Gomez-Mestre, I. & Buchholz, D. R. Developmental plasticity mirrors differences among taxa in spadefoot toads linking plasticity and diversity. Proc. Natl Acad. Sci. USA 103, 19021–19026 (2006).
McDiarmid, R. W. & Altig, R. Tadpoles: The Biology of Anuran Larvae (Univ. Chicago Press, Chicago, 1999).
Leichty, A. R., Pfennig, D. W., Jones, C. D. & Pfennig, K. S. Relaxed genetic constraint is ancestral to the evolution of phenotypic plasticity. Integr. Comp. Biol. 52, 16–30 (2012).
Levis, N. A., Serrato-Capuchina, A. & Pfennig, D. W. Genetic accommodation in the wild: evolution of gene expression plasticity during character displacement. J. Evol. Biol. 30, 1712–1723 (2017).
Huang, Y. & Agrawal, A. F. Experimental evolution of gene expression and plasticity in alternative selective regimes. PLoS Genet. 12, e1006336 (2016).
van Gestel, J. & Weissing, F. J. Is plasticity caused by single genes? Nature 555, E19–E20 (2018).
Paull, J. S., Martin, R. A. & Pfennig, D. W. Increased competition as a cost of specialization during the evolution of resource polymorphism. Biol. J. Linn. Soc. 107, 845–853 (2012).
Levis, N. A., Martin, R. A., O’Donnell, K. A. & Pfennig, D. W. Intraspecific adaptive radiation: competition, ecological opportunity, and phenotypic diversification within species. Evolution 71, 2496–2509 (2017).
Ledón-Rettig, C. C., Pfennig, D. W. & Crespi, E. J. Stress hormones and the fitness consequences associated with the transition to a novel diet in larval amphibians. J. Exp. Biol. 212, 3743–3750 (2009).
Ho, W. C. & Zhang, J. Evolutionary adaptations to new environments generally reverse plastic phenotypic changes. Nat. Commun. 9, 350 (2018).
Casasa, S. & Moczek, A. P. The role of ancestral phenotypic plasticity in evolutionary diversification: population density effects in horned beetles. Anim. Behav. 137, 53–61 (2018).
Pfennig, D. W., Rice, A. M. & Martin, R. A. Ecological opportunity and phenotypic plasticity interact to promote character displacement and species coexistence. Ecology 87, 769–779 (2006).
Gosner, K. L. A simplified table for staging anuran embryos with notes on identification. Herpetologica 16, 183–190 (1960).
lme4 R Package (R Foundation for Statistical Computing, Vienna, 2014).
Masek, T., Vopalensky, V., Suchomelova, P. & Pospisek, M. Denaturing RNA electrophoresis in TAE agarose gels. Anal. Biochem. 336, 46–50 (2005).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the Method. Methods 25, 402–408 (2001).
Pfennig, D. W. & Murphy, P. J. Character displacement in polyphenic tadpoles. Evolution 54, 1738–1749 (2000).
Pfennig, D. W., Rice, A. M. & Martin, R. A. Field and experimental evidence for competition’s role in phenotypic divergence. Evolution 61, 257–271 (2007).
Martin, R. A. & Pfennig, D. W. Widespread disruptive selection in the wild is associated with intense resource competition. BMC Evol. Biol. 12, 136 (2012).
Martin, R. A. & Pfennig, D. W. Disruptive selection in natural populations: the roles of ecological specialization and resource competition. Am. Nat. 174, 268–281 (2009).
Martin, R. A. & Pfennig, D. W. Field and experimental evidence that competition and ecological opportunity promote resource polymorphism. Biol. J. Linn. Soc. 100, 73–88 (2010).
Pfennig, D. W. Proximate and functional causes of polyphenism in an anuran tadpole. Funct. Ecol. 6, 167–174 (1992).
Collyer, M. L. & Adams, D. C. Analysis of two-state multivariate phenotypic change in ecological studies. Ecology 88, 683–692 (2007).
Adams, D. C., Collyer, M. L. & Kaliontzopoulou, A. geomorph: Software for Geometric Morphometric Analyses R Package Version 3.0.6 (2018); https://cran.r-project.org/package=geomorph
Collyer, M. L., Sekora, D. J. & Adams, D. C. A method for analysis of phenotypic change for phenotypes described by high-dimensional data. Heredity 115, 357–365 (2015).
Roff, D. A. Evolutionary Quantitative Genetics (Chapman and Hall, New York, 1997).
Levis, N. A. & Pfennig, D. W. Phenotypic plasticity, canalization, and the origins of novelty: evidence and mechanisms from amphibians. Semin. Cell Dev. Biol. https://doi.org/10.1016/j.semcdb.2018.01.012 (2018).
Acknowledgements
We thank W. Zhang and K. O’Donnell for assistance with animal care, A. Serrato-Capuchina for help collecting tadpoles, and K. Pfennig, I. Ehrenreich, C. Ledón-Rettig, D. Matute, C. Martin, R. Martin and A. Levis for comments on the manuscript. Funding was provided by the National Science Foundation grants DEB-1643239 and DEB-1753865.
Author information
Authors and Affiliations
Contributions
N.A.L. and D.W.P. conceived and designed the study; N.A.L. and A.J.I. collected data; N.A.L. analysed the data; N.A.L. and D.W.P. wrote the manuscript. Photos in Figs. 2,3 were taken by D.W.P. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables and Supplementary Figures
Rights and permissions
About this article
Cite this article
Levis, N.A., Isdaner, A.J. & Pfennig, D.W. Morphological novelty emerges from pre-existing phenotypic plasticity. Nat Ecol Evol 2, 1289–1297 (2018). https://doi.org/10.1038/s41559-018-0601-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-018-0601-8
This article is cited by
-
Bacteria evolve macroscopic multicellularity by the genetic assimilation of phenotypically plastic cell clustering
Nature Communications (2023)
-
Widespread learned predator recognition to an alien predator across populations in an amphibian species
Scientific Reports (2023)
-
Genetic assimilation of ancestral plasticity during parallel adaptation to zinc contamination in Silene uniflora
Nature Ecology & Evolution (2023)
-
Condition- and context-dependent variation of sexual dimorphism across lizard populations at different spatial scales
Scientific Reports (2022)
-
Phenotypic variation in Xenopus laevis tadpoles from contrasting climatic regimes is the result of adaptation and plasticity
Oecologia (2022)