Abstract
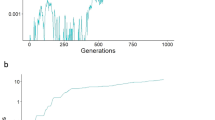
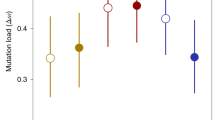
It is unclear why sexually reproducing isogamous species frequently contain just two self-incompatible mating types. Deterministic theory suggests that since rare novel mating types experience a selective advantage (by virtue of their many potential partners), the number of mating types should consistently grow. However, in nature, species with thousands of mating types are exceedingly rare. Several competing theories for the predominance of species with two mating types exist, yet they lack an explanation for how many are possible and in which species to expect high numbers. Here, we present a theoretical null model that explains the distribution of mating type numbers using just three biological parameters: mutation rate, population size and the rate of sex. If the number of mating types results from a mutation–extinction balance, the rate of sexual reproduction plays a crucial role. If sex is facultative and rare (a very common combination in isogamous species), mating type diversity will remain low. In this rare sex regime, small fitness differences between the mating types lead to more frequent extinctions, further lowering mating type diversity. We also show that the empirical literature supports the role of drift and facultativeness of sex as a determinant of mating type dynamics.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Billiard, S. et al. Having sex, yes, but with whom? Inferences from fungi on the evolution of anisogamy and mating types. Biol. Rev. 86, 421–442 (2011).
Iwasa, Y. & Sasaki, A. Evolution of the number of sexes. Evolution 41, 49–65 (1987).
Nagylaki, T. The deterministic behavior of self-incompatibility alleles. Genetics 79, 545–550 (1975).
Kothe, E. Tetrapolar fungal mating types: sexes by the thousands. FEMS Microbiol. Rev. 18, 65–87 (1996).
Lehtonen, J., Kokko, H. & Parker, G. A. What do isogamous organisms teach us about sex and the two sexes? Phil. Trans. R. Soc. B 371, 20150532 (2016).
Beukeboom, L. & Perrin, N. The Evolution of Sex Determination (Oxford Univ. Press, Oxford, 2014).
Geng, S., De Hoff, P. & Umen, J. G. Evolution of sexes from an ancestral mating-type specification pathway. PLoS Biol. 12, e1001904 (2014).
Hamaji, T. et al. Sequence of the gonium pectorale mating locus reveals a complex and dynamic history of changes in volvocine algal mating haplotypes. G3 6, 1179–1189 (2016).
Togashi, T. & Cox, P. A. The Evolution of Anisogamy (Cambridge Univ. Press, Cambridge, 2011).
Lehtonen, J. & Kokko, H. Two roads to two sexes: unifying gamete competition and gamete limitation in a single model of anisogamy evolution. Behav. Ecol. Sociobiol. 65, 445–459 (2011).
Parker, G. A., Baker, R. R. & Smith, V. G. The origin and evolution of gamete dimorphism and the male–female phenomenon. J. Theor. Biol. 36, 529–553 (1972).
Togashi, T., Bartelt, J. L., Yoshimura, J., Tainaka, K. & Cox, P. A. Evolutionary trajectories explain the diversified evolution of isogamy and anisogamy in marine green algae. Proc. Natl Acad. Sci. USA 109, 13692–13697 (2012).
Hoekstra, R. F. in The Evolution of Sex and its Consequences (ed. Stearns, S. C.) 59–91 (Birkhaeuser, Basel, 1987).
Nozaki, H., Mori, T., Misumi, O., Matsunaga, S. & Kuroiwa, T. Males evolved from the dominant isogametic mating type. Curr. Biol. 16, R1018–R1020 (2006).
Hadjivasiliou, Z., Iwasa, Y. & Pomiankowski, A. Cell–cell signalling in sexual chemotaxis: a basis for gametic differentiation, mating types and sexes. J. R. Soc. Interface 12, 20150342 (2015).
James, T. Y. Why mushrooms have evolved to be so promiscuous: insights from evolutionary and ecological patterns. Fungal Biol. Rev. 29, 167–178 (2015).
Hoekstra, R. F. On the asymmetry of sex: evolution of mating types in isogamous populations. J. Theor. Biol. 98, 427–451 (1982).
Hadjivasiliou, Z. & Pomiankowski, A. Gamete signalling underlies the evolution of mating types and their number. Phil. Trans. R. Soc. B 371, 20150531 (2016).
Phadke, S. S. & Zufall, R. A. Rapid diversification of mating systems in ciliates. Biol. J. Linn. Soc. 98, 187–197 (2009).
Hurst, L. D. Why are there only two sexes? Proc. R. Soc. B 263, 415–422 (1996).
Hurst, L. D. & Hamilton, W. D. Cytoplasmic fusion and the nature of sexes. Proc. R. Soc. B 247, 189–194 (1992).
Hutson, V. & Law, R. Four steps to two sexes. Proc. R. Soc. Lond. B 253, 43–51 (1993).
Hadjivasiliou, Z., Lane, N., Seymour, R. M. & Pomiankowski, A. Dynamics of mitochondrial inheritance in the evolution of binary mating types and two sexes. Proc. R. Soc. B 280, 20131920 (2013).
Nieuwenhuis, B. P. S. et al. Evolution of uni- and bifactorial sexual compatibility systems in fungi. Heredity 111, 445–455 (2013).
Vuilleumier, S., Alcala, N. & Niculita-Hirzel, H. Transitions from reproductive systems governed by two self-incompatible loci to one in fungi. Evolution 67, 501–516 (2013).
Umen, J. G. Evolution of sex and mating loci: an expanded view from volvocine algae. Curr. Opin. Microbiol. 14, 634–641 (2011).
Nedelcu, A. M. & Michod, R. E. Sex as a response to oxidative stress: the effect of antioxidants on sexual induction in a facultatively sexual lineage. Proc. R. Soc. B 270, S136–S139 (2003).
Harris, E. H. The Chlamydomonas Sourcebook: A Comprehensive Guide to Biology and Laboratory Use (Academic Press, Cambridge, 1989).
Van den Hoek, C., Mann, D. & Jahns, H. M. Algae: An Introduction to Phycology (Cambridge Univ. Press, Cambridge, 1996).
Goodenough, U., Lin, H. & Lee, J. H. Sex determination in Chlamydomonas. Semin. Cell. Dev. Biol. 18, 350–361 (2007).
Hartl, D. L. & Clark, A. G. Principles of Population Genetics 4th edn (Sinauer Associates, Sunderland, 2007).
Hartfield, M. & Keightley, P. D. Current hypotheses for the evolution of sex and recombination. Integr. Zool. 7, 192–209 (2012).
Power, H. W. On forces of selection in the evolution of mating types. Am. Nat. 110, 937–944 (1976).
Paixao, T., Phadke, S. S., Azevedo, R. B. & Zufall, R. A. Sex ratio evolution under probabilistic sex determination. Evolution 65, 2050–2060 (2011).
Abramowitz, M. & Stegun, I. A. Handbook of Mathematical Functions (Dover Publications, New York, 1965).
Wright, S. The distribution of self-sterility alleles in populations. Genetics 24, 538–552 (1939).
Nunney, L. The effect of neighborhood size on effective population size in theory and in practice. Heredity 117, 224–232 (2016).
Charlesworth, B. Effective population size. Curr. Biol. 12, 716–717 (2002).
Baranova, M. A. et al. Extraordinary genetic diversity in a wood decay mushroom. Mol. Biol. Evol. 32, 2775–2783 (2015).
Gossmann, T. I., Keightley, P. D. & Eyre-Walker, A. The effect of variation in the effective population size on the rate of adaptive molecular evolution in eukaryotes. Genome Biol. Evol. 4, 658–667 (2012).
Tsai, I. J., Bensasson, D., Burt, A. & Koufopanou, V. Population genomics of the wild yeast Saccharomyces paradoxus: quantifying the life cycle. Proc. Natl Acad. Sci. USA 105, 4957–4962 (2008).
Nieuwenhuis, B. P. S. & James, T. Y. The frequency of sex in fungi. Phil. Trans. R. Soc. B 371, 20150540 (2016).
Doerder, F. P., Gates, M. A., Eberhardt, F. P. & Arslanyolu, M. High frequency of sex and equal frequencies of mating types in natural populations of the ciliate Tetrahymena thermophila. Proc. Natl Acad. Sci. USA 92, 8715–8718 (1995).
Lucchesi, P. & Santangelo, G. How often does conjugation in ciliates occur? Clues from a seven-year study on marine sandy shores. Aquat. Microb. Ecol. 36, 195–200 (2004).
Lee, S. C., Ni, M., Li, W., Shertz, C. & Heitman, J. The evolution of sex: a perspective from the fungal kingdom. Microbiol. Mol. Biol. Rev. 74, 298–340 (2010).
Dunthorn, M. & Katz, L. A. Secretive ciliates and putative asexuality in microbial eukaryotes. Trends Microbiol. 18, 183–188 (2010).
Ruderfer, D. M., Pratt, S. C., Seidel, H. S. & Kruglyak, L.Population genomic analysis of outcrossing and recombination in yeast. Nat. Genet. 38, 1077–1081 (2006).
Jang, H. & Ehrenreich, I. M. Genome-wide characterization of genetic variation in the unicellular, green alga Chlamydomonas reinhardtii. PLoS ONE 7, e41307 (2012).
Hadjivasiliou, Z. Theoretical Studies on the Role and Evolution of Mating Types and Two Sexes. PhD thesis, Univ. College London (2014).
Nieuwenhuis, B. P. S. & Aanen, D. K. Sexual selection in fungi. J. Evol. Biol. 25, 2397–2411 (2012).
Bull, J. J. & Pease, C. M. Combinatorics and variety of mating-type systems. Evolution 43, 667–671 (1989).
Wilson, A. M. et al. Homothallism: an umbrella term for describing diverse sexual behaviours. IMA Fungus 6, 207–214 (2015).
Bell, G. Experimental sexual selection in Chlamydomonas. J. Evol. Biol. 18, 722–734 (2005).
Milgroom, M. G. Recombination and the multilocus structure of fungal populations.Annu. Rev. Phytopathol. 34, 457–477 (1996).
Teixeira, M. et al. Asexual propagation of a virulent clone complex in a human and feline outbreak of sporotrichosis. Eukaryot. Cell 14, 158–169 (2015).
Brisse, S. et al. Uneven distribution of mating types among genotypes of Candida glabrata isolates from clinical samples. Eukaryot. Cell 8, 287–295 (2009).
Kwon-Chung, K. J., Edman, J. C. & Wickes, B. L. Genetic association of mating types and virulence in Cryptococcus neoformans. Infect. Immun. 60, 602–605 (1992).
Narmani, A., Arzanlou, M. & Babai-Ahari, A. Uneven distribution of mating-type alleles among Togninia minima isolates, one of the causal agents of leaf stripe disease on grapevines in northwest iran. J. Phytopathol. 164, 441–447 (2016).
Du, X.-H. et al. Mixed-reproductive strategies, competitive mating-type distribution and life cycle of fourteen black morel species. Sci. Rep. 7, 1493 (2017).
West, S. Sex Allocation (Princeton Univ. Press, Princeton, 2009).
Mandel, M. A., Barker, S., Kroken, B. M., Rounsley, S. D. & Orbach, M. J. Genomic and population analyses of the mating type loci in Coccidioides species reveal evidence for sexual reproduction and gene acquisition. Eukaryot. Cell 6, 1189–1199 (2007).
Douglas, T. E., Strassmann, J. E. & Queller, D. C. Sex ratio and gamete size across eastern North America in Dictyostelium discoideum, a social amoeba with three sexes. J. Evol. Biol. 29, 1298–1306 (2016).
Gervais, C. E., Castric, V., Ressayre, S. & Billiard, A.Origin and diversification dynamics of self-incompatibility haplotypes. Genetics 188, 625–636 (2011).
Wright, S. On the number of self-incompatibility alleles maintained in equilibrium by a given mutation rate in a population of a given size: a re-examination. Biometrics 16, 61–85 (1960).
Wright, S. The distribution of self-incompatibility alleles in populations. Evolution 18, 609–619 (1964).
Hadjivasiliou, Z., Pomiankowski, A. & Kuijper, B. The evolution of mating type switching. Evolution 70, 1569–1581 (2016).
Nieuwenhuis, B. P. S. & Immler, S. The evolution of mating-type switching for reproductive assurance. Bioessays 38, 1141–1149 (2016).
Becks, L. & Agrawal, A. F. Higher rates of sex evolve under K-selection. J. Evol. Biol. 26, 900–905 (2013).
Bengtsson, B. O. & Ceplitis, A. The balance between sexual and asexual reproduction in plants living in variable environments. J. Evol. Biol. 13, 415–422 (2000).
Crow, J. F. & Kimura, M. An Introduction to Population Genetics Theory (Blackburn Press, Caldwell, 1970).
Yokoyama, S. & Hetherington, L. E. The expected number of self-incompatibility alleles in finite plant populations. Heredity 48, 299–303 (1982).
McKane, A. J., Biancalani, T. & Rogers, T. Stochastic pattern formation and spontaneous polarisation: the linear noise approximation and beyond. Bull. Math. Biol. 76, 895–921 (2014).
Muirhead, C. A. & Wakeley, J. Modeling multiallelic selection using a Moran model. Genetics 182, 1141–1157 (2009).
Gillespie, D. T. A general method for numerically simulating the stochastic time evolution of coupled chemical reactions. J. Comput. Phys. 22, 403–434 (1976).
Acknowledgements
We thank J. Christie, L. Turner and the audience of the seminar series at the Milner Centre for Evolution for useful discussions and input. G.W.A.C. thanks the Finnish Center for Excellence in Biological Interactions and Leverhulme Early Career Fellowship provided by the Leverhulme Trust for funding. H.K. thanks the Swiss National Science Foundation and Academy of Finland for funding.
Author information
Authors and Affiliations
Contributions
G.W.A.C. designed the project and conducted the mathematical analysis. G.W.A.C. and H.K. developed the model and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Mathematical theory, model analysis and simulations
Rights and permissions
About this article
Cite this article
Constable, G.W.A., Kokko, H. The rate of facultative sex governs the number of expected mating types in isogamous species. Nat Ecol Evol 2, 1168–1175 (2018). https://doi.org/10.1038/s41559-018-0580-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-018-0580-9
This article is cited by
-
Oxygen, life forms, and the evolution of sexes in multicellular eukaryotes
Heredity (2020)
-
Estimating the number of sexual events per generation in a facultatively sexual haploid population
Heredity (2019)
-
Sex begets sexes
Nature Ecology & Evolution (2018)