Abstract
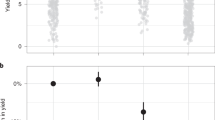
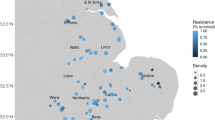
Repeated use of xenobiotic chemicals has selected for the rapid evolution of resistance, threatening health and food security at a global scale. Strategies for preventing the evolution of resistance include cycling and mixtures of chemicals and diversification of management. We currently lack large-scale studies that evaluate the efficacy of these different strategies for minimizing the evolution of resistance. Here we use a national-scale data set of occurrence of the weed Alopecurus myosuroides (black-grass) in the United Kingdom to address this. Weed densities are correlated with assays of evolved resistance, supporting the hypothesis that resistance is driving weed abundance at a national scale. Resistance was correlated with the frequency of historical herbicide applications, suggesting that evolution of resistance is primarily driven by intensity of exposure to herbicides, but was unrelated directly to other cultural techniques. We find that populations resistant to one herbicide are likely to show resistance to multiple herbicide classes. Finally, we show that the economic costs of evolved resistance are considerable: loss of control through resistance can double the economic costs of weeds. This research highlights the importance of managing threats to food production and healthcare systems using an evolutionarily informed approach in a proactive not reactive manner.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Tilman, D., Cassman, K. G., Matson, P. A., Naylor, R. & Polasky, S. Agricultural sustainability and intensive production practices. Nature 418, 671–677 (2002).
Palumbi, S. R. Humans as the world’s greatest evolutionary force. Science 293, 1786–1790 (2001).
Laxminarayan, R. et al. Access to effective antimicrobials: a worldwide challenge. Lancet 387, 168–175 (2016).
Hairston, N. G., Ellner, S. P., Geber, M. A., Yoshida, T. & Fox, J. A. Rapid evolution and the convergence of ecological and evolutionary time. Ecol. Lett. 8, 1114–1127 (2005).
Duke, S. O. Why have no new herbicide modes of action appeared in recent years? Pest Manag. Sci. 68, 505–512 (2012).
Gluckman, P. D., Hanson, M. A. & Mitchell, M. D. Developmental origins of health and disease: reducing the burden of chronic disease in the next generation. Genome Med. 2, 14 (2010).
Powles, S. B. & Yu, Q. Evolution in action: plants resistant to herbicides. Annu. Rev. Plant Biol. 61, 317–347 (2010).
Roush, R. & Tabashnik, B. E. (eds) Pesticide Resistance in Arthropods (Springer, Boston, 2012).
REX Consortium Heterogeneity of selection and the evolution of resistance. Trends Ecol. Evol. 28, 110–118 (2013).
Antiretroviral Therapy for HIV Infection in Adults and Adolescents: Recommendations for a Public Health Approach 2010 Revision (World Health Organization, 2010).
Treatment of Tuberculosis: Guidelines (World Health Organization, 2010).
Guidelines for the Treatment of Malaria 3rd edn (World Health Organization, 2015).
Beckie, H. J. & Reboud, X. Selecting for weed resistance: herbicide rotation and mixture. Weed Tech. 23, 363–370 (2009).
Diggle, A. J., Neve, P. B. & Smith, F. P. Herbicides used in combination can reduce the probability of herbicide resistance in finite weed populations. Weed Res. 43, 371–382 (2003).
Samoucha, Y. & Ulrich, G. Use of two- and three-way mixtures to prevent buildup of resistance to phenylamide fungicides in Phytophthora and Plasmopara. Phytopathology 77, 1405–1409 (1987).
Lagator, M., Vogwill, T., Mead, A., Colegrave, N. & Neve, P. Herbicide mixtures at high doses slow the evolution of resistance in experimentally evolving populations of Chlamydomonas reinhardtii. New Phytol. 198, 938–945 (2013).
Lagator, M., Vogwill, T., Colegrave, N. & Neve, P. Herbicide cycling has diverse effects on evolution of resistance in Chlamydomonas reinhardtii. Evol. Appl. 6, 197–206 (2013).
Norsworthy, J. K. et al. Reducing the risks of herbicide resistance: best management practices and recommendations. Weed Sci. 60, 31–62 (2012).
Delye, C., Jasieniuk, M. & Le Corre, V. Deciphering the evolution of herbicide resistance in weeds. Trends Genet. 29, 649–658 (2013).
Chikowo, R., Faloya, V., Petit, S. & Munier-Jolain, N. M. Integrated weed management systems allow reduced reliance on herbicides and long-term weed control. Agric. Ecosyst. Environ. 132, 237–242 (2009).
Powles, S. B. & Matthews, J. M. Multiple Herbicide Resistance in Annual Ryegrass ( Lolium rigidum ): A Driving Force for the Adoption of Integrated Weed Management (Springer, Dordrecht, 1992).
Heap, I. The International Survey of Herbicide Resistant Weeds (2017); http://www.weedscience.org/
Höjgard, S. Antibiotic resistance – why is the problem so difficult to solve? Infect. Ecol. Epidemiol. 2, 18165 (2012).
Beckie, H. J. Herbicide-resistant weeds: management tactics and practices. Weed Tech. 20, 793–814 (2006).
Powles, S., Preston, C., Bryan, I. & Jutsum, A. Herbicide resistance: impact and management. Adv. Agron. 58, 57–93 (1997).
Moss, S. R. & Clarke, J. H. Guidelines for the prevention and control of herbicide-resistant black-grass (Alopecurus myosuroides Huds.). Crop Prot. 13, 230–234 (1994).
Comins, H. N. Tactics for resistance management using multiple pesticides. Agric. Ecosyst. Environ. 16, 129–148 (1986).
Neve, P. Challenges for herbicide resistance evolution and management: 50 years after Harper. Weed Res. 47, 365–369 (2007).
Herrmann, J., Hess, M., Strek, H., Richter, O. & Beffa, R. Linkage of the current ALS-resistance status with field history information of multiple fields infested with blackgrass. Jul.-Kuhn-Arch. 443, 273–279 (2016).
Evans, J. A. et al. Managing the evolution of herbicide resistance. Pest Manag. Sci. 72, 74–80 (2016).
Moss, S. R., Perryman, S. A. M. & Tatnell, L. V. Managing herbicide-resistant blackgrass (Alopecurus myosuroides): theory and practice. Weed Technol. 21, 300–309 (2007).
Lutman, P. J. W., Moss, S. R., Cook, S., Welham, S. J. & Kim, D.-S. A review of the effects of crop agronomy on the management of Alopecurus myosuroides. Weed Res. 53, 299–313 (2013).
Godfray, H. C. J. et al. Food security: the challenge of feeding 9 billion people. Science 327, 812–818 (2010).
Oerke, E. C. Crop losses to pests. J. Agric. Sci. 144, 31–43 (2005).
Cousens, R. Theory and reality of weed control thresholds. Plant Prot. Quart. 2, 13–20 (1987).
Maxwell, B. D. Weed thresholds: the space component and considerations for herbicide resistance. Weed Tech. 6, 205–212 (1992).
Foresman, C. & Glasgow, L. US grower perceptions and experiences with glyphosate-resistant weeds. Pest Manag. Sci. 64, 388–391 (2008).
Llewellyn, R. S., Lindner, R. K., Pannell, D. J. & Powles, S. B. Herbicide resistance and the adoption of integrated weed management by Western Australian grain growers. Agric. Econ. 36, 123–130 (2007).
Perez-Jones, A., Park, K.-W., Polge, N., Colquhoun, J. & Mallory-Smith, C. A. Investigating the mechanisms of glyphosate resistance in Lolium multiflorum. Planta 226, 395–404 (2007).
Queenborough, S. A., Burnet, K. M., Sutherland, W. J., Watkinson, A. R. & Freckleton, R. P. From meso- to macroscale population dynamics: a new density-structured approach. Methods Ecol. Evol. 2, 289–302 (2011).
Avery, B. W. Soil Classification for England and Wales (Higher Categories) (Soil Survey Technical Monograph No. 14, Harpenden, 1980).
Clayden, B. and Hollis, J. M. Criteria for Differentiating Soil Series (Soil Survey Technical Monograph No. 17, Harpenden, 1984).
Bates, D., Mächler, M., Bolker, B. & Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Soft. 67, 1–48 (2014).
Harrison, X. A. Using observation-level random effects to model overdispersion in count data in ecology and evolution. PeerJ 2, e616 (2014).
Halekoh, U. & Højsgaard, S. A Kenward–Roger approximation and parametric bootstrap methods for tests in linear mixed models – the R package pbkrtest. J. Stat. Soft. 59, 1–32 (2014).
Kalogirou, S. lctools: Local Correlation, Spatial Inequalities, Geographically Weighted Regression and Other Tools R package version 0.2-5 (2016); https://CRAN.R-project.org/package=lctools
Barton, K. MuMIN: Multi-Model Inference R package version 0.12.2 (2009); https://cran.r-project.org/web/packages/MuMIn/MuMIn.pdf
Stratonovitch, P., Storkey, J. & Semenov, M. A. A process-based approach to modelling impacts of climate change on the damage niche of an agricultural weed. Glob. Change Biol. 18, 2071–2080 (2012).
Perring, F. & Walters, S. M. Atlas of the British Flora (Botanical Society of the British Isles, 1962).
Preston, C. D., Pearman, D. A. & Dines, T. D. New Atlas of the British and Irish Flora (Botanical Society of the Britain and Ireland, 2002).
Acknowledgements
The authors would like to thank all of the farmers who have kindly allowed us to survey their fields and provided field management data for the analyses. This work was funded by BBSRC (BB/L001489/1) and the Agriculture and Horticulture Development Board (Cereals and Oilseeds).
Author information
Authors and Affiliations
Contributions
The project was conceptualized by H.L.H., R.P.F., P.N., D.Z.C. and K.N.; the survey was designed by R.P.F. and H.L.H., the resistance assays were designed by P.N. and D.C. Statistical analysis was undertaken by H.L.H., R.P.F., S.R.C. and D.C. Data were collected by H.L.H., D.C., L.C. and R.H. The inital manuscript was drafted by H.L.H. and R.P.F., with H.L.H., R.P.F., D.Z.C., S.R.C., D.C., P.N. and K.N. contributing to the writing. Funding was acquired by R.P.F., D.Z.C., P.N. and K.N.
Corresponding author
Ethics declarations
Competing interests
R.P.F., D.Z.C., L.C., H.L.H., S.R.C., R.H. and D.C. declare no competing financial interests; P.N. supervises a PhD student co-funded by Bayer (not part of this project).
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figure 1, Supplementary Tables 1–5, Supplementary Experimental Procedures
Rights and permissions
About this article
Cite this article
Hicks, H.L., Comont, D., Coutts, S.R. et al. The factors driving evolved herbicide resistance at a national scale. Nat Ecol Evol 2, 529–536 (2018). https://doi.org/10.1038/s41559-018-0470-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-018-0470-1
This article is cited by
-
Acting pre-emptively reduces the long-term costs of managing herbicide resistance
Scientific Reports (2024)
-
Trade-offs constrain the success of glyphosate-free farming
Scientific Reports (2024)
-
Expanding risk predictions of pesticide resistance evolution in arthropod pests with a proxy for selection pressure
Journal of Pest Science (2023)
-
Drone and sensor technology for sustainable weed management: a review
Chemical and Biological Technologies in Agriculture (2021)
-
Reduction in weed infestation through integrated depletion of the weed seed bank in a rice-wheat cropping system
Agronomy for Sustainable Development (2021)