Abstract
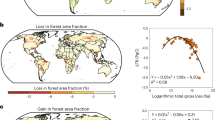
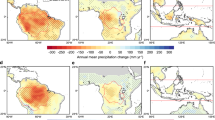
Tropical ecosystems store over half of the world’s aboveground live carbon as biomass, and water availability plays a key role in its distribution. Although precipitation and temperature are shifting across the tropics, their effect on biomass and carbon storage remains uncertain. Here we use empirical relationships between climate and aboveground biomass content to show that the contraction of humid regions, and expansion of those with intense dry periods, results in substantial carbon loss from the neotropics. Under a low emission scenario (Representative Concentration Pathway 4.5) this could cause a net reduction of aboveground live carbon of ~14.4–23.9 PgC (6.8–12%) from 1950–2100. Under a high emissions scenario (Representative Concentration Pathway 8.5) net carbon losses could double across the tropics, to ~28.2–39.7 PgC (13.3–20.1%). The contraction of humid regions in South America accounts for ~40% of this change. Climate mitigation strategies could prevent half of the carbon losses and help maintain the natural tropical net carbon sink.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
CRU v.4.04 data can be downloaded from the CRU website (https://crudata.uea.ac.uk/cru/data/hrg/). Downscaled climate data from the Coupled Model Intercomparison Project Phase 5 (CMIP5) multimodel ensemble can be downloaded from https://gdo-dcp.ucllnl.org/downscaled_cmip_projections/dcpInterface.html. Global biomass data is available from https://developers.google.com/earth-engine/datasets/catalog/WHRC_biomass_tropical)2, https://doi.org/10.5281/zenodo.4161694 (ref. 3) and https://catalogue.ceda.ac.uk/uuid/5f331c418e9f4935b8eb1b836f8a91b8 (ref. 20). MODIS Land Cover data is available at https://lpdaac.usgs.gov/products/mcd12q1v006/. Continent borders are from the Environmental Systems Research Institute’s World Continents shapefiles v.10.3 (http://gis.ucla.edu/geodata/dataset/continent_ln)53. Preprocessed input data (as specified in the manuscript), partial results and final predictions of changes in area and biomass are available in a public repository via Dryad54.
Code availability
Relevant R scripts used to process the data and perform the analyses are available in a public repository via Dryad54.
References
Saatchi, S. S. et al. Benchmark map of forest carbon stocks in tropical regions across three continents. Proc. Natl Acad. Sci. 108, 9899–9904 (2011).
Baccini, A. et al. Estimated carbon dioxide emissions from tropical deforestation improved by carbon-density maps. Nat. Clim. Change 2, 182–185 (2012).
Xu, L. et al. Changes in global terrestrial live biomass over the 21st century. Sci. Adv. 7, eabe9829 (2021).
Betts, R. A. et al. The role of ecosystem-atmosphere interactions in simulated Amazonian precipitation decrease and forest dieback under global climate warming. Theor. Appl. Climatol. 78, 157–175 (2004).
Cox, P. M. et al. Sensitivity of tropical carbon to climate change constrained by carbon dioxide variability. Nature 494, 341–344 (2013).
Rammig, A. et al. Estimating the risk of Amazonian forest dieback. N. Phytol. 187, 694–706 (2010).
Huntingford, C. et al. Towards quantifying uncertainty in predictions of Amazon ‘dieback’. Philos. Trans. R. Soc. B Biol. Sci. 363, 1857–1864 (2008).
Galbraith, D. et al. Multiple mechanisms of Amazonian forest biomass losses in three dynamic global vegetation models under climate change. N. Phytol. 187, 647–665 (2010).
Kumar, D., Pfeiffer, M., Gaillard, C., Langan, L. & Scheiter, S. Climate change and elevated CO2 favor forest over savanna under different future scenarios in South Asia. Biogeosciences 18, 2957–2979 (2021).
Huntingford, C. et al. Simulated resilience of tropical rainforests to CO2-induced climate change. Nat. Geosci. 6, 268–273 (2013).
Brienen, R. J. W. et al. Forest carbon sink neutralized by pervasive growth-lifespan trade-offs. Nat. Commun. 11, 4241 (2020).
Koch, A., Hubau, W. & Lewis, S. L. Earth system models are not capturing present-day tropical forest carbon dynamics. Earths Future 9, e2020EF001874 (2021).
Negrón-Juárez, R. I., Koven, C. D., Riley, W. J., Knox, R. G. & Chambers, J. Q. Observed allocations of productivity and biomass, and turnover times in tropical forests are not accurately represented in CMIP5 Earth system models. Environ. Res. Lett. 10, 064017 (2015).
Fleischer, K. et al. Amazon forest response to CO2 fertilization dependent on plant phosphorus acquisition. Nat. Geosci. 12, 736–741 (2019).
Terrer, C. et al. Nitrogen and phosphorus constrain the CO2 fertilization of global plant biomass. Nat. Clim. Change 9, 684–689 (2019).
Malhi, Y. et al. Exploring the likelihood and mechanism of a climate-change-induced dieback of the Amazon rainforest. Proc. Natl Acad. Sci. 106, 20610–20615 (2009).
Zelazowski, P., Malhi, Y., Huntingford, C., Sitch, S. & Fisher, J. B. Changes in the potential distribution of humid tropical forests on a warmer planet. Philos. Trans. R. Soc. Math. Phys. Eng. Sci. 369, 137–160 (2011).
Huang, L. et al. Drought dominates the interannual variability in global terrestrial net primary production by controlling semi-arid ecosystems. Sci. Rep. 6, 24639 (2016).
Castanho, A. D. A. et al. Potential shifts in the aboveground biomass and physiognomy of a seasonally dry tropical forest in a changing climate. Environ. Res. Lett. 15, 034053 (2020).
Santoro, M. & Cartus, O. ESA Biomass Climate Change Initiative (Biomass_cci): global datasets of forest above-ground biomass for the years 2010, 2017 and 2018, v3. NERC EDS Centre for Environmental Data Analysis https://doi.org/10.5285/5f331c418e9f4935b8eb1b836f8a91b8 (2021).
Baccini, A. et al. Tropical forests are a net carbon source based on aboveground measurements of gain and loss. Science 358, 230–234 (2017).
Harris, N. L. et al. Global maps of twenty-first century forest carbon fluxes. Nat. Clim. Change 11, 234–240 (2021).
Gatti, L. V. et al. Amazonia as a carbon source linked to deforestation and climate change. Nature 595, 388–393 (2021).
Qin, Y. et al. Carbon loss from forest degradation exceeds that from deforestation in the Brazilian Amazon. Nat. Clim. Change 11, 442–448 (2021).
Brienen, R. J. W. et al. Long-term decline of the Amazon carbon sink. Nature 519, 344–348 (2015).
Hubau, W. et al. Asynchronous carbon sink saturation in African and Amazonian tropical forests. Nature 579, 80–87 (2020).
Phillips, O. L. et al. Drought sensitivity of the amazon rainforest. Science 323, 1344–1347 (2009).
Ross, C. W. et al. Woody-biomass projections and drivers of change in sub-Saharan Africa. Nat. Clim. Change 11, 449–455 (2021).
Larjavaara, M., Lu, X., Chen, X. & Vastaranta, M. Impact of rising temperatures on the biomass of humid old-growth forests of the world. Carbon Balance Manag. 16, 31 (2021).
Romps, D. M., Seeley, J. T., Vollaro, D. & Molinari, J. Projected increase in lightning strikes in the United States due to global warming. Science 346, 851–854 (2014).
Gora, E. M., Bitzer, P. M., Burchfield, J. C., Gutierrez, C. & Yanoviak, S. P. The contributions of lightning to biomass turnover, gap formation and plant mortality in a tropical forest. Ecology 102, e03541 (2021).
Magnabosco Marra, D. et al. Windthrows control biomass patterns and functional composition of Amazon forests. Glob. Change Biol. 24, 5867–5881 (2018).
Negrón-Juárez, R. I. et al. Windthrow variability in central amazonia. Atmosphere 8, 28 (2017).
Silva Junior, C. H. L. et al. Persistent collapse of biomass in Amazonian forest edges following deforestation leads to unaccounted carbon losses. Sci. Adv. 6, 40 (2020).
Yin, Y. et al. Fire decline in dry tropical ecosystems enhances decadal land carbon sink. Nat. Commun. 11, 1900 (2020).
Koch, A. & Kaplan, J. O. Tropical forest restoration under future climate change. Nat. Clim. Change 12, 279–283 (2022).
Wang, S. et al. Recent global decline of CO2 fertilization effects on vegetation photosynthesis. Science 370, 1295–1300 (2020).
Case, M. F. & Staver, A. C. Fire prevents woody encroachment only at higher-than-historical frequencies in a South African savanna. J. Appl. Ecol. 54, 955–962 (2017).
Mau, A. C., Reed, S. C., Wood, T. E. & Cavaleri, M. A. Temperate and tropical forest canopies are already functioning beyond their thermal thresholds for photosynthesis. Forests 9, 47 (2018).
Kolby Smith, W. et al. Large divergence of satellite and Earth system model estimates of global terrestrial CO2 fertilization. Nat. Clim. Change 6, 306–310 (2016).
Martens, C. et al. Large uncertainties in future biome changes in Africa call for flexible climate adaptation strategies. Glob. Change Biol. 27, 340–358 (2021).
Doughty, C. E. & Goulden, M. L. Are tropical forests near a high temperature threshold?. J. Geophys. Res. Biogeosciences 113, G00B07 (2008).
Doughty, C. E. & Goulden, M. L. Seasonal patterns of tropical forest leaf area index and CO2 exchange. J. Geophys. Res. Biogeosciences 113, G00B06 (2008).
Langenbrunner, B., Pritchard, M. S., Kooperman, G. J. & Randerson, J. T. Why does amazon precipitation decrease when tropical forests respond to increasing CO2? Earths Future 7, 450–468 (2019).
Harris, I., Osborn, T. J., Jones, P. & Lister, D. Version 4 of the CRU TS monthly high-resolution gridded multivariate climate dataset. Sci. Data 7, 109 (2020).
Maurer, E. P., Brekke, L., Pruitt, T. & Duffy, P. B. Fine-resolution climate projections enhance regional climate change impact studies. EOS Trans. Am. Geophys. Union 88, 504–504 (2007).
Reclamation. Downscaled CMIP3 and CMIP5 Climate and Hydrology Projections: Release of Hydrology Projections, Comparison with preceding Information, and Summary of User Needs. https://gdo-dcp.ucllnl.org/downscaled_cmip_projections/techmemo/BCSD5HydrologyMemo.pdf (2014).
Silva de Miranda, P. L. et al. Using tree species inventories to map biomes and assess their climatic overlaps in lowland tropical South America. Glob. Ecol. Biogeogr. 27, 899–912 (2018).
Beguería, S., Vicente-Serrano, S. M., Reig, F. & Latorre, B. Standardized precipitation evapotranspiration index (SPEI) revisited: parameter fitting, evapotranspiration models, tools, datasets and drought monitoring. Int. J. Climatol. 34, 3001–3023 (2014).
Wright, M. N. & Ziegler, A. ranger: a fast implementation of random forests for high dimensional data in C++ and R. J. Stat. Softw. 77, 1–17 (2017).
Middleton, N., Thomas, D. & UNEP. World Atlas of Desertification (Arnold, 1997).
Staver, A. C., Archibald, S. & Levin, S. A. The global extent and determinants of Savanna and forest as alternative biome states. Science 334, 230–232 (2011).
ESRI Data & Maps. World Continents Version 10.3. (2015).
Uribe, M. R. et al. Net loss of biomass predicted for tropical biomes in a changing climate. Dryad https://doi.org/10.7280/D1D124 (2023).
Acknowledgements
We acknowledge the World Climate Research Programme’s Working Group on Coupled Modelling, which is responsible for CMIP, and we thank the climate modelling groups (listed in Supplementary Table 4) for producing and making available their model outputs. For CMIP, the U.S. Department of Energy’s Program for Climate Model Diagnosis and Intercomparison provided coordinating support and led development of software infrastructure in partnership with the Global Organization for Earth System Science Portals. This research was supported through funding from the National Science Foundation (MSB-ECA award no. 1802754; INFEWS/T1 award no. 1739724).
Author information
Authors and Affiliations
Contributions
M.T.C., M.N.M. and P.M.B. conceived the study. M.R.U. and A.D.A.C. performed the data analysis and prepared the manuscript. P.M.B. and D.V. contributed to the data processing and analysis. All the authors contributed and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Climate Change thanks Manan Bhan and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Distribution of climatic zones in the tropics for Historic and Future 30-year time periods (1950–1979, 1980–2009, 2010–2039, 2040–2069, and 2070–2099).
Colors represent the climatic zones. Contour lines in the maps and gray dashed lines in the scatterplots are plotted for reference to show the distribution and limits of the climatic zones in the period 1950–1979. Climate data from CRU v.4.0445 for 1950–2009 and from CMIP5 projections46,47 for 2010–2099 under RCP 8.5.
Extended Data Fig. 2 Individual model projections of precipitation and MCWD under RCP 8.5.
Dots represent each model’s mean Historic (1950–1979) climate for each climatic zone. Connected arrowheads represent average climate at the end of the century (2070–2099). The black shapes correspond to the average climate from all models. Models used are listed in Supplementary Table 1. Climate data from CRU v.4.0445 for 1950–2009 and from CMIP5 projections46,47 for 2010–2099 under RCP 8.5.
Supplementary information
Supplementary Information
Supplementary Tables 1–8 and Figs. 1–3.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Uribe, M.d.R., Coe, M.T., Castanho, A.D.A. et al. Net loss of biomass predicted for tropical biomes in a changing climate. Nat. Clim. Chang. 13, 274–281 (2023). https://doi.org/10.1038/s41558-023-01600-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41558-023-01600-z
This article is cited by
-
The time since land-use transition drives changes in fire activity in the Amazon-Cerrado region
Communications Earth & Environment (2024)
-
Enhanced future vegetation growth with elevated carbon dioxide concentrations could increase fire activity
Communications Earth & Environment (2024)