Abstract
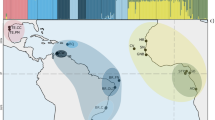
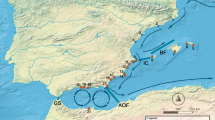
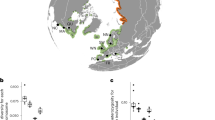
Antarctica has long been considered biologically isolated1. Global warming will make parts of Antarctica more habitable for invasive taxa, yet presumed barriers to dispersal—especially the Southern Ocean’s strong, circumpolar winds, ocean currents and fronts—have been thought to protect the region from non-anthropogenic colonizations from the north1,2. We combine molecular and oceanographic tools to directly test for biological dispersal across the Southern Ocean. Genomic analyses reveal that rafting keystone kelps recently travelled >20,000 km and crossed several ocean-front ‘barriers’ to reach Antarctica from mid-latitude source populations. High-resolution ocean circulation models, incorporating both mesoscale eddies and wave-driven Stokes drift, indicate that such Antarctic incursions are remarkably frequent and rapid. Our results demonstrate that storm-forced surface waves and ocean eddies can dramatically enhance oceanographic connectivity for drift particles in surface layers, and show that Antarctica is not biologically isolated. We infer that Antarctica’s long-standing ecological differences have been the result of environmental extremes that have precluded the establishment of temperate-adapted taxa, but that such taxa nonetheless frequently disperse to the region. Global warming thus has the potential to allow the establishment of diverse new species—including keystone kelps that would drastically alter ecosystem dynamics—even without anthropogenic introductions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Chown, S. L. et al. The changing form of Antarctic biodiversity. Nature 522, 431–438 (2015).
Clarke, A., Barnes, D. K. A. & Hodgson, D. A. How isolated is Antarctica? Trends Ecol. Evol. 20, 1–3 (2005).
Scher, H. D. & Martin, E. E. Timing and climatic consequences of the opening of Drake Passage. Science 312, 428–430 (2006).
Rogers, A. D. Evolution and biodiversity of Antarctic organisms: a molecular perspective. Phil. Trans. R. Soc. Lond. B 362, 2191–2214 (2007).
Dufour, C. O. et al. Role of mesoscale eddies in cross-frontal transport of heat and biogeochemical tracers in the Southern Ocean. J. Phys. Oceanogr. 45, 3057–3081 (2015).
Fraser, C. I., Nikula, R., Ruzzante, D. E. & Waters, J. M. Poleward bound: biological impacts of Southern Hemisphere glaciation. Trends Ecol. Evol. 27, 462–471 (2012).
Barnes, D. K. A., Hodgson, D. A., Convey, P., Allen, C. S. & Clarke, A. Incursion and excursion of Antarctic biota: past, present and future. Glob. Ecol. Biogeogr. 15, 121–142 (2006).
Poulin, E., González-Wevar, C., Díaz, A., Gérard, K. & Hüne, M. Divergence between Antarctic and South American marine invertebrates: what molecular biology tells us about Scotia Arc geodynamics and the intensification of the Antarctic Circumpolar Current. Glob. Planet. Change 123, 392–399 (2014).
Thornhill, D. J., Mahon, A. R., Norenburg, J. L. & Halanych, K. M. Open-ocean barriers to dispersal: a test case with the Antarctic Polar Front and the ribbon worm Parborlasia corrugatus (Nemertea: Lineidae). Mol. Ecol. 17, 5104–5117 (2008).
Leese, F., Agrawal, S. & Held, C. Long-distance island hopping without dispersal stages: transportation across major zoogeographic barriers in a Southern Ocean isopod. Naturwissenschaften 97, 583–594 (2010).
Pisa, S. et al. The cosmopolitan moss Bryum argenteum in Antarctica: recent colonisation or in situ survival? Polar Biol. 37, 1469–1477 (2014).
Fasanella, M., Premoli, A. C., Urdampilleta, J. D., González, M. L. & Chiapella, J. O. How did a grass reach Antarctica? The Patagonian connection of Deschampsia antarctica (Poaceae). Bot. J. Linn. Soc. 185, 511–524 (2017).
De Bruyn, M. et al. Rapid response of a marine mammal species to Holocene climate and habitat change. PLoS Genet. 5, e1000554 (2009).
Griffiths, H. J., Whittle, R. J., Roberts, S. J., Belchier, M. & Linse, K. Antarctic crabs: invasion or endurance? PLoS ONE 8, e66981 (2013).
Fraser, C. I., Kay, G. M., Plessis, M. D. & Ryan, P. G. Breaking down the barrier: dispersal across the Antarctic Polar Front. Ecography 40, 235–237 (2017).
Bromwich, D. H. et al. Central West Antarctica among the most rapidly warming regions on Earth. Nat. Geosci. 6, 139–145 (2013).
Duffy, G. A. et al. Barriers to globally invasive species are weakening across the Antarctic. Divers. Distrib. 23, 982–996 (2017).
Griffiths, H. J., Meijers, A. J. S. & Bracegirdle, T. J. More losers than winners in a century of future Southern Ocean seafloor warming. Nat. Clim. Change 7, 749–754 (2017).
Fraser, C. I., Nikula, R. & Waters, J. M. Oceanic rafting by a coastal community. Proc. R. Soc. B 278, 649–655 (2011).
Smith, S. D. A. Kelp rafts in the Southern Ocean. Glob. Ecol. Biogeogr. 11, 67–69 (2002).
Fraser, C. I., Nikula, R., Spencer, H. G. & Waters, J. M. Kelp genes reveal effects of subantarctic sea ice during the Last Glacial Maximum. Proc. Natl Acad. Sci. USA 106, 3249–3253 (2009).
Graham, M. H., Kinlan, B. P. & Grosberg, R. K. Post-glacial redistribution and shifts in productivity of giant kelp forests. Proc. R. Soc. B 277, 399–406 (2010).
Fach, B. A. & Klinck, J. M. Transport of Antarctic krill (Euphausia superba) across the Scotia Sea. Part I: circulation and particle tracking simulations. Deep Sea Res. I 53, 987–1010 (2006).
Meredith, M. P. et al. Sustained monitoring of the Southern Ocean at Drake Passage: past achievements and future priorities. Rev. Geophys. 49, RG4005 (2011).
Tala, F., Velásquez, M., Mansilla, A., Macaya, E. C. & Thiel, M. Latitudinal and seasonal effects on short-term acclimation of floating kelp species from the South-East Pacific J. Exp. Mar. Biol. Ecol. 483, 31–41 (2016).
Gillespie, R. G. et al. Long-distance dispersal: a framework for hypothesis testing. Trends Ecol. Evol. 27, 47–56 (2012).
Carlton, J. T. et al. Tsunami-driven rafting: transoceanic species dispersal and implications for marine biogeography. Science 357, 1402–1406 (2017).
Barber, H. N., Dadswell, H. E. & Ingle, H. D. Transport of driftwood from South America to Tasmania and Macquarie Island. Nature 184, 203–204 (1959).
Coombs, D. S. & Landis, C. A. Pumice from South Sandwich eruption of March 1962 reaches New Zealand. Nature 209, 289–290 (1966).
Fraser, C. I. in Seaweed Phylogeography: Adaptation and Evolution of Seaweeds Under Environmental Change (eds Hu, Z.-M. & Fraser, C.) 131–143 (Springer, Dordrecht, 2016).
Fraser, C. I., Winter, D. J., Spencer, H. G. & Waters, J. M. Multigene phylogeny of the southern bull-kelp genus Durvillaea (Phaeophyceae: Fucales). Mol. Phylogenet. Evol. 57, 1301–1311 (2010).
Fraser, C. I., McGaughran, A., Chuah, A. & Waters, J. M. The importance of replicating genomic analyses to verify phylogenetic signal for recently evolved lineages. Mol. Ecol. 25, 3683–3695 (2016).
Elshire, R. J. et al. A robust, simple genotyping-by-sequencing (GBS) approach for high diversity species. PLoS ONE 6, e19379 (2011).
Morris, G. P., Grabowski, P. P. & Borevitz, J. O. Genomic diversity in switchgrass (Panicum virgatum): from the continental scale to a dune landscape. Mol. Ecol. 20, 4938–4952 (2011).
Murray, K. D. & Borevitz, J. O. Axe: rapid, competitive sequence read demultiplexing using a trie. Preprint at bioRxiv https://doi.org/10.1101/160606 (2017).
Catchen, J., Hohenlohe, P. A., Bassham, S., Amores, A. & Cresko, W. A. Stacks: an analysis tool set for population genomics. Mol. Ecol. 22, 3124–3140 (2013).
Nguyen, L.-T., Schmidt, H. A., von Haeseler, A. & Minh, B. Q. IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 32, 268–274 (2015).
Kalyaanamoorthy, S., Minh, B. Q., Wong, T. K. F., von Haeseler, A. & Jermiin, L. S. ModelFinder: fast model selection for accurate phylogenetic estimates. Nat. Meth. 14, 587–589 (2017).
Minh, B. Q., Nguyen, M. A. T. & von Haeseler, A. Ultrafast approximation for phylogenetic bootstrap. Mol. Biol. Evol. 30, 1188–1195 (2013).
Cummings, J. A. & Smedstad, O. M. in Data Assimilation for Atmospheric, Oceanic and Hydrologic Applications Vol. II (eds Park, S. K. & Xu, L.) 303–343 (Springer, Berlin, 2013).
Paris, C. B., Helgers, J., van Sebille, E. & Srinivasan, A. Connectivity modeling system: a probabilistic modeling tool for the multi-scale tracking of biotic and abiotic variability in the ocean. Environ. Model. Softw. 42, 47–54 (2013).
Rascle, N. & Ardhuin, F. A global wave parameter database for geophysical applications. Part 2: model validation with improved source term parameterization. Ocean Model. 70, 174–188 (2013).
Lumpkin, R. et al. Removing spurious low-frequency variability in surface drifter velocities. J. Atmos. Ocean. Technol. 30, 353–360 (2013).
Acknowledgements
We thank J. Lee and F. le Bouard for assistance with at-sea observations, and the Antarctic Circumpolar Expedition and Swiss Polar Institute for facilitating the surveys. We thank A. McGaughran and W. Blanchard for analytical advice. Kelp samples used in analyses included some specimens collected by others—see acknowledgments in ref. 31. The oceanographic modelling was undertaken on the National Computational Infrastructure in Canberra, Australia, which is supported by the Australian Commonwealth Government. The research was funded by Australian Research Council grants to C.I.F. (DE140101715 and FT170100281) and A.K.M. (DE170100184), and Fondap-IDEAL grant 15150003 from CONICYT-Chile to E.C.M. and N.V.
Author information
Authors and Affiliations
Contributions
C.I.F. and J.M.W. conceived the research. A.K.M., E.v.S. and A.M.H. conducted all oceanographic modelling. P.G.R. conducted the at-sea kelp surveys. E.C.M. and N.V. discovered and identified the drift samples from beaches in Antarctica. C.I.F. sourced (and in most cases, collected) the kelp samples used in the genomic analyses, and directed the genomic laboratory work carried out by A.P. C.J. contributed bioinformatics expertise and ran the genomic analyses. C.I.F. wrote the first draft of the paper. All authors contributed to editing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information Description: Supplementary Figures 1–7, Supplementary Table 1
Supplementary Figures 1–7, Supplementary Table 1
Supplementary Table 2
Records of at-sea Durvillaea antarctica observations during the ACE voyage (2016-2017)
Supplementary Movie 1
Example trajectories of 140 surface-bound Lagrangian particles released from South Georgia that did (orange trails) and did not (blue trails) reach the Antarctic shelf. The particles are advected by a combination of ocean currents and Stokes drift. Ocean current speed is indicated by colours. The tail on each particle shows its path over the last 50 days
Rights and permissions
About this article
Cite this article
Fraser, C.I., Morrison, A.K., Hogg, A.M. et al. Antarctica’s ecological isolation will be broken by storm-driven dispersal and warming. Nature Clim Change 8, 704–708 (2018). https://doi.org/10.1038/s41558-018-0209-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41558-018-0209-7
This article is cited by
-
Residues of anthropogenic origin found at Stranger Point/Cabo Funes, 25 de Mayo/King George Island, Antarctica
Polar Biology (2024)
-
Extent and reproduction of coastal species on plastic debris in the North Pacific Subtropical Gyre
Nature Ecology & Evolution (2023)
-
A pioneer morphological and genetic study of the intertidal fauna of the Gerlache Strait (Antarctic Peninsula)
Environmental Monitoring and Assessment (2023)
-
Typical and anomalous pathways of surface-floating material in the Northern North Atlantic and Arctic Ocean
Scientific Reports (2022)
-
Potential negative effects of ocean afforestation on offshore ecosystems
Nature Ecology & Evolution (2022)