Abstract
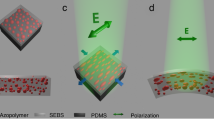
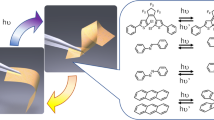
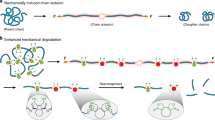
Azobenzene has been widely explored as a photoresponsive element in materials science. Although some studies have investigated the force-induced isomerization of azobenzene, the effect of force on the rupture of azobenzene has not been explored. Here we show that the light-induced structural change of azobenzene can also alter its rupture forces, making it an ideal light-responsive mechanophore. Using single-molecule force spectroscopy and ultrasonication, we found that cis and trans para-azobenzene isomers possess contrasting mechanical properties. Dynamic force spectroscopy experiments and quantum-chemical calculations in which azobenzene regioisomers were pulled from different directions revealed that the distinct rupture forces of the two isomers are due to the pulling direction rather than the energetic difference between the two isomers. These mechanical features of azobenzene can be used to rationally control the macroscopic fracture behaviours of polymer networks by photoillumination. The use of light-induced conformational changes to alter the mechanical response of mechanophores provides an attractive way to engineer polymer networks of light-regulatable mechanical properties.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data are available in the main text or the Supplementary Information. Source data are provided with this paper.
References
Beyer, M. K. & Clausen-Schaumann, H. Mechanochemistry: the mechanical activation of covalent bonds. Chem. Rev. 105, 2921–2948 (2005).
Chen, Y., Mellot, G., van Luijk, D., Creton, C. & Sijbesma, R. P. Mechanochemical tools for polymer materials. Chem. Soc. Rev. 50, 4100–4140 (2021).
Hu, X., McFadden, M. E., Barber, R. W. & Robb, M. J. Mechanochemical regulation of a photochemical reaction. J. Am. Chem. Soc. 140, 14073–14077 (2018).
Muramatsu, T., Sagara, Y., Traeger, H., Tamaoki, N. & Weder, C. Mechanoresponsive behavior of a polymer-embedded red-light emitting rotaxane mechanophore. ACS Appl. Mater. Interfaces 11, 24571–24576 (2019).
Dubach, F. F. C., Ellenbroek, W. G. & Storm, C. How accurately do mechanophores report on bond scission in soft polymer materials? J. Polym. Sci. 59, 1188–1199 (2021).
Robb, M. J. et al. Regioisomer-specific mechanochromism of naphthopyran in polymeric materials. J. Am. Chem. Soc. 138, 12328–12331 (2016).
Kida, J. et al. The photoregulation of a mechanochemical polymer scission. Nat. Commun. 9, 3504 (2018).
Li, J., Nagamani, C. & Moore, J. S. Polymer mechanochemistry: from destructive to productive. Acc. Chem. Res. 48, 2181–2190 (2015).
Nixon, R. & De Bo, G. Three concomitant C–C dissociation pathways during the mechanical activation of an N-heterocyclic carbene precursor. Nat. Chem. 12, 826–831 (2020).
Yang, J. et al. Benzoladderene mechanophores: synthesis, polymerization and mechanochemical transformation. J. Am. Chem. Soc. 141, 6479–6483 (2019).
Aerts, A., Lugger, S. J. D., Heuts, J. P. A. & Sijbesma, R. P. Pyranine based ion-paired complex as a mechanophore in polyurethanes. Macromol. Rapid Commun. 42, e2000476 (2021).
Bian, Q., Fu, L. & Li, H. Engineering shape memory and morphing protein hydrogels based on protein unfolding and folding. Nat. Commun. 13, 137 (2022).
Imato, K., Yamanaka, R., Nakajima, H. & Takeda, N. Fluorescent supramolecular mechanophores based on charge-transfer interactions. Chem. Commun. 56, 7937–7940 (2020).
Sakai, H. et al. Visualization and quantitative evaluation of toughening polymer networks by a sacrificial dynamic cross-linker with mechanochromic properties. ACS Macro Lett. 9, 1108–1113 (2020).
Kida, J., Aoki, D. & Otsuka, H. Self-strengthening of cross-linked elastomers via the use of dynamic covalent macrocyclic mechanophores. ACS Macro Lett. 10, 558–563 (2021).
Pang, X., Lv, J. A., Zhu, C., Qin, L. & Yu, Y. Photodeformable azobenzene-containing liquid crystal polymers and soft actuators. Adv. Mater. 31, e1904224 (2019).
Wu, X. et al. Reversible hydrogels with tunable mechanical properties for optically controlling cell migration. Nano Res. 11, 5556–5565 (2018).
Yager, K. G. & Barrett, C. J. Novel photo-switching using azobenzene functional materials. J. Photochem. Photobiol. A 182, 250–261 (2006).
Cho, W. et al. Photo-triggered shape reconfiguration in stretchable reduced graphene oxide-patterned azobenzene-functionalized liquid crystalline polymer networks. Adv. Funct. Mater. 31, 2102106 (2021).
Ramirez, A. L. et al. Mechanochemical strengthening of a synthetic polymer in response to typically destructive shear forces. Nat. Chem. 5, 757–761 (2013).
Diesendruck, C. E. et al. Mechanically triggered heterolytic unzipping of a low-ceiling-temperature polymer. Nat. Chem. 6, 623–628 (2014).
Versaw, B. A., Zeng, T., Hu, X. & Robb, M. J. Harnessing the power of force: development of mechanophores for molecular release. J. Am. Chem. Soc. 143, 21461–21473 (2021).
Imato, K. et al. Mechanophores with a reversible radical system and freezing-induced mechanochemistry in polymer solutions and gels. Angew. Chem. Int. Ed. 54, 6168–6172 (2015).
Wu, M., Guo, Z., He, W., Yuan, W. & Chen, Y. Empowering self-reporting polymer blends with orthogonal optical properties responsive in a broader force range. Chem. Sci. 12, 1245–1250 (2020).
Ducrot, E., Chen, Y., Bulters, M., Sijbesma, R. P. & Creton, C. Toughening elastomers with sacrificial bonds and watching them break. Science 344, 186–189 (2014).
Krieg, M. et al. Atomic force microscopy-based mechanobiology. Nat. Rev. Phys. 1, 41–57 (2019).
Yao, R., Li, X., Xiao, N., Weng, W. & Zhang, W. Single-molecule observation of mechanical isomerization of spirothiopyran and subsequent click addition. Nano Res. 14, 2654–2658 (2021).
Pill, M. F. et al. Mechanochemical cycloreversion of cyclobutane observed at the single molecule level. Chemistry 22, 12034–12039 (2016).
Kawasaki, K., Aoki, D. & Otsuka, H. Diarylbiindolinones as substituent-tunable mechanochromophores and their application in mechanochromic polymers. Macromol. Rapid Commun. 41, e1900460 (2020).
Vantomme, G., Gelebart, A. H., Broer, D. J. & Meijer, E. W. A four-blade light-driven plastic mill based on hydrazone liquid-crystal networks. Tetrahedron 73, 4963–4967 (2017).
Mamiya, J., Kuriyama, A., Yokota, N., Yamada, M. & Ikeda, T. Photomobile polymer materials: photoresponsive behavior of cross-linked liquid-crystalline polymers with mesomorphic diarylethenes. Chem. Eur. J. 21, 3174–3177 (2015).
Tiu, B. D. B., Delparastan, P., Ney, M. R., Gerst, M. & Messersmith, P. B. Cooperativity of catechols and amines in high-performance dry/wet adhesives. Angew. Chem. Int. Ed. 59, 16616–16624 (2020).
Li, Y. et al. Molecular design principles of lysine-DOPA wet adhesion. Nat. Commun. 11, 3895 (2020).
Berkovich, R., Fernandez, V. I., Stirnemann, G., Valle-Orero, J. & Fernandez, J. M. Segmentation and the entropic elasticity of modular proteins. J. Phys. Chem. Lett. 9, 4707–4713 (2018).
Muller, D. J. et al. Atomic force microscopy-based force spectroscopy and multiparametric imaging of biomolecular and cellular systems. Chem. Rev. 121, 11701–11725 (2021).
Chung, J., Kushner, A. M., Weisman, A. C. & Guan, Z. Direct correlation of single-molecule properties with bulk mechanical performance for the biomimetic design of polymers. Nat. Mater. 13, 1055–1062 (2014).
Perkins, T. T., Smith, D. E. & Chu, S. Single polymer dynamics in an elongational flow. Science 276, 2016–2021 (1997).
Zhang, Y. et al. Distal conformational locks on ferrocene mechanophores guide reaction pathways for increased mechanochemical reactivity. Nat. Chem. 13, 56–62 (2021).
Kersey, F. R., Yount, W. C. & Craig, S. L. Single-molecule force spectroscopy of bimolecular reactions: system homology in the mechanical activation of ligand substitution reactions. J. Am. Chem. Soc. 128, 3886–3887 (2006).
Horst, M. et al. Understanding the mechanochemistry of ladder-type cyclobutane mechanophores by single molecule force spectroscopy. J. Am. Chem. Soc. 143, 12328–12334 (2021).
Razgoniaev, A. O. et al. Single-molecule activation and quantification of mechanically triggered palladium-carbene bond dissociation. J. Am. Chem. Soc. 143, 1784–1789 (2021).
Sha, Y. et al. Generalizing metallocene mechanochemistry to ruthenocene mechanophores. Chem. Sci. 10, 4959–4965 (2019).
Lin, Y. & Craig, S. L. Oxidative regulation of the mechanical strength of a C–S bond. Chem. Sci. 11, 10444–10448 (2020).
Kida, J., Aoki, D. & Otsuka, H. Photoinduced regulation of the heat resistance in polymer networks with diarylethene-conjugated reversible covalent cross-links. ACS Macro. Lett. 8, 1–6 (2019).
Mita, I., Horie, K. & Hirao, K. Photochemistry in polymer solids. 9. Photoisomerization of azobenzene in a polycarbonate film. Macromolecules 22, 558–563 (1989).
Diez Cabanes, V., Van Dyck, C., Osella, S., Cornil, D. & Cornil, J. Challenges for incorporating optical switchability in organic-based electronic devices. ACS Appl. Mater. Interfaces 13, 27737–27748 (2021).
Bisoyi, H. K. & Li, Q. Light-driven liquid crystalline materials: from photo-induced phase transitions and property modulations to applications. Chem. Rev. 116, 15089–15166 (2016).
Hugel, T. et al. Single-molecule optomechanical cycle. Science 296, 1103–1106 (2002).
Holland, N. B. et al. Single molecule force spectroscopy of Azobenzene polymers: switching elasticity of single photochromic macromolecules. Macromolecules 36, 2015–2023 (2003).
Resetic, A., Milavec, J., Zupancic, B., Domenici, V. & Zalar, B. Polymer-dispersed liquid crystal elastomers. Nat. Commun. 7, 13140 (2016).
Surampudi, S. K., Patel, H. R., Nagarjuna, G. & Venkataraman, D. Mechano-isomerization of azobenzene. Chem. Commun. 49, 7519–7521 (2013).
Turansky, R., Konopka, M., Doltsinis, N. L., Stich, I. & Marx, D. Switching of functionalized azobenzene suspended between gold tips by mechanochemical, photochemical and opto-mechanical means. Phys. Chem. Chem. Phys. 12, 13922–13932 (2010).
Lin, Y., Hansen, H. R., Brittain, W. J. & Craig, S. L. Strain-dependent kinetics in the cis-to-trans isomerization of azobenzene in bulk elastomers. J. Phys. Chem. B 123, 8492–8498 (2019).
Oesterhelt, F., Rief, M. & Gaub, H. E. Single molecule force spectroscopy by AFM indicates helical structure of poly(ethylene-glycol) in water. New J. Phys. 1, 6.1–6.11 (1999).
Bandara, H. M. & Burdette, S. C. Photoisomerization in different classes of azobenzene. Chem. Soc. Rev. 41, 1809–1825 (2012).
Garcia-Amoros, J., Sanchez-Ferrer, A., Massad, W. A., Nonell, S. & Velasco, D. Kinetic study of the fast thermal cis-to-trans isomerisation of para-, ortho- and polyhydroxyazobenzenes. Phys. Chem. Chem. Phys. 12, 13238–13242 (2010).
Zhang, H. et al. Spiropyran as a mechanochromic probe in dual cross-linked elastomers. Macromolecules 47, 6783–6790 (2014).
Friddle, R. W., Noy, A. & De Yoreo, J. J. Interpreting the widespread nonlinear force spectra of intermolecular bonds. Proc. Natl Acad. Sci. USA 109, 13573–13578 (2012).
Dudko, O. K., Hummer, G. & Szabo, A. Intrinsic rates and activation free energies from single-molecule pulling experiments. Phys. Rev. Lett. 96, 108101 (2006).
Stevenson, R. & De Bo, G. Controlling reactivity by geometry in retro-Diels-Alder reactions under tension. J. Am. Chem. Soc. 139, 16768–16771 (2017).
Mardyukov, A., Sanchez-Garcia, E., Crespo-Otero, R. & Sander, W. Interaction and reaction of the phenyl radical with water: a source of OH radicals. Angew. Chem. Int. Ed. 48, 4804–4807 (2009).
Kubota, K. et al. Introduction of a luminophore into generic polymers via mechanoradical coupling with a prefluorescent reagent. Angew. Chem. Int. Ed. 60, 16003–16008 (2021).
Wang, Z. J. et al. Azo-crosslinked double-network hydrogels enabling highly efficient mechanoradical generation. J. Am. Chem. Soc. 144, 3154–3161 (2022).
Jiang, J. et al. Computational exploration of polymer mechanochemistry: quantitation of activation force and systematic discovery of reaction sites utilizing two forces. Preprint at https://chemrxiv.org/engage/chemrxiv/article-details/637c72e874b7b6b3ba072adc (2022).
Wang, S. et al. Mechanism dictates mechanics: a molecular substituent effect in the macroscopic fracture of a covalent polymer network. J. Am. Chem. Soc. 143, 3714–3718 (2021).
Wang, S., Panyukov, S., Rubinstein, M. & Craig, S. L. Quantitative adjustment to the molecular energy parameter in the Lake-Thomas theory of polymer fracture energy. Macromolecules 52, 2772–2777 (2019).
Gong, J. P. Why are double network hydrogels so tough? Soft Matter 6, 2583–2590 (2010).
Gong, J. P., Katsuyama, Y., Kurokawa, T. & Osada, Y. Double-network hydrogels with extremely high mechanical strength. Adv. Mater. 15, 1155–1158 (2003).
Wang, Z. et al. Toughening hydrogels through force-triggered chemical reactions that lengthen polymer strands. Science 374, 193–196 (2021).
Matsuda, T. et al. Yielding criteria of double network hydrogels. Macromolecules 49, 1865–1872 (2016).
Chen, Y. et al. Fast reversible isomerization of merocyanine as a tool to quantify stress history in elastomers. Chem. Sci. 12, 1693–1701 (2020).
Brügner, O. & Walter, M. Temperature and loading rate dependent rupture forces from universal paths in mechanochemistry. Phys. Rev. Mater. 2, 113603 (2018).
Walton, E. B., Lee, S. & Van Vliet, K. J. Extending Bell’s model: how force transducer stiffness alters measured unbinding forces and kinetics of molecular complexes. Biophys. J. 94, 2621–2630 (2008).
Kianfar, P., Abolfathi, N. & Karimi, N. Z. Investigating the effect of different transducer stiffness values on the contactin complex detachment by steered molecular dynamics. J. Mol. Graph. Model. 75, 340–346 (2017).
Zheng, P. & Li, H. Direct measurements of the mechanical stability of zinc-thiolate bonds in rubredoxin by single-molecule atomic force microscopy. Biophys. J. 101, 1467–1473 (2011).
Zheng, P. & Li, H. Highly covalent ferric-thiolate bonds exhibit surprisingly low mechanical stability. J. Am. Chem. Soc. 133, 6791–6798 (2011).
Grandbois, M., Beyer, M., Rief, M., Clausen-Schaumann, H. & Gaub, H. E. How strong is a covalent bond? Science 283, 1727–1730 (1999).
Sagara, Y. et al. Rotaxane-based mechanophores enable polymers with mechanically switchable white photoluminescence. ACS. Cent. Sci. 5, 874–881 (2019).
Bertrand, O. & Gohy, J.-F. Photo-responsive polymers: synthesis and applications. Polym. Chem. 8, 52–73 (2017).
Lugger, J. A. M., Marin San Roman, P. P., Kroonen, C. C. E. & Sijbesma, R. P. Nanoporous films with photoswitchable absorption kinetics based on polymerizable columnar discotic liquid crystals. ACS Appl. Mater. Interfaces 13, 4385–4392 (2021).
Yu, H. & Kobayashi, T. Photoresponsive block copolymers containing azobenzenes and other chromophores. Molecules 15, 570–603 (2010).
Becke, A. D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 38, 3098–3100 (1988).
Becke, A. D. A new mixing of Hartree-Fock and local density‐functional theories. J. Chem. Phys. 98, 1372–1377 (1993).
Becke, A. D. Density‐functional thermochemistry. III The role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993).
Lee, C., Yang, W. & Parr, R. G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 37, 785–789 (1988).
Frisch, M. J. et al. Gaussian 16 (Gaussian, 2016).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Grimme, S., Ehrlich, S. & Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 32, 1456–1465 (2011).
McLean, A. D. & Chandler, G. S. Contracted Gaussian basis sets for molecular calculations. I. Second row atoms, Z = 11–18. J. Chem. Phys. 72, 5639–5648 (1980).
Krishnan, R., Binkley, J. S., Seeger, R. & Pople, J. A. Self‐consistent molecular orbital methods. XX. A basis set for correlated wave functions. J. Chem. Phys. 72, 650–654 (1980).
Bauernschmitt, R. & Ahlrichs, R. Stability analysis for solutions of the closed shell Kohn-Sham equation. J. Chem. Phys. 104, 9047–9052 (1996).
Tomasi, J., Mennucci, B. & Cammi, R. Quantum mechanical continuum solvation models. Chem. Rev. 105, 2999–3094 (2005).
Scalmani, G. & Frisch, M. J. Continuous surface charge polarizable continuum models of solvation. I. General formalism. J. Chem. Phys. 132, 114110 (2010).
Maeda, S., Ohno, K. & Morokuma, K. Systematic exploration of the mechanism of chemical reactions: the global reaction route mapping (GRRM) strategy using the ADDF and AFIR methods. Phys. Chem. Chem. Phys. 15, 3683–3701 (2013).
Maeda, S., Harabuchi, Y., Takagi, M., Taketsugu, T. & Morokuma, K. Artificial force induced reaction (AFIR) method for exploring quantum chemical potential energy surfaces. Chem. Rec. 16, 2232–2248 (2016).
Maeda, S. et al. Implementation and performance of the artificial force induced reaction method in the GRRM17 program. J. Comput. Chem. 39, 233–251 (2018).
Maeda, S. & Harabuchi, Y. Exploring paths of chemical transformations in molecular and periodic systems: an approach utilizing force. WIREs Comput. Mol. Sci. 11, e1538 (2021).
Zheng, Y. et al. In situ and real-time visualization of mechanochemical damage in double-network hydrogels by prefluorescent probe via oxygen-relayed radical trapping. J. Am. Chem. Soc. 145, 7376–7389 (2023).
Acknowledgements
We thank Y. Li and X. Wang for helpful discussions. This work was supported mainly by the National Natural Science Foundation of China (nos. T2225016, 12002149, T2322010 and 11934008) to Y. Cao, Y.L., B.X. and W.W., the National Key R&D Program of China (grant no. 2020YFA0908100) to Y. Cao, the Fundamental Research Funds for the Central Universities (grant no. 020514380274) to Y. Cao and the Natural Science Foundation of Jiangsu Province (no. BK20220120) to B.X. This work was partially supported by JSPS KAKENHI (grant no. JP22H04968) to J.P.G. and T.N., by JST, PRESTO, Japan (grant no. JPMJPR2098) to T.N. and by a scholarship of MEXT Japan to Z.J.W.
Author information
Authors and Affiliations
Contributions
Y. Cao, J.P.G., T.N., S.M. and W.W. conceived the idea and designed the study. Y.L. performed the experiments and analysed the results. J.L., J.Z. and Z.Z. performed some of the SMFS experiments. J.Y., B.X. and Z.J.W. prepared the hydrogel samples and tensile tests. Y.Z., M.W. and Y. Chen performed some of the ultrasonication tests. Y.Y., J.J., Y.H., and S.M. performed the quantum calculations. Y. Cao, W.W., S.M. and J.P.G. wrote and refined the paper. Y. Cao and W.W. supervised the project. All authors discussed the results.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Constantino Creton, Anne-Sophie Duwez and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–6, Figs. 1–21, text, references and the Cartesian coordinates of the optimized structures.
Supplementary Data 1
Source data for Supplementary Figures.
Source data
Source Data Fig. 2
Force curves and statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
GPC and UV–vis curves presented in Fig. 4.
Source Data Fig. 5
Distance–force curves presented in Fig. 5.
Source Data Fig. 6
Stress–strain curves and statistical source data presented in Fig. 6.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, Y., Xue, B., Yang, J. et al. Azobenzene as a photoswitchable mechanophore. Nat. Chem. 16, 446–455 (2024). https://doi.org/10.1038/s41557-023-01389-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-023-01389-6
This article is cited by
-
A quirky mechanophore comes to light
Nature Reviews Materials (2023)