Abstract
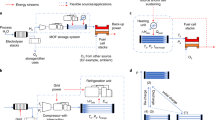
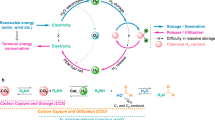
Hydrogen has the highest gravimetric energy density of any energy carrier and produces water as the only oxidation product, making it extremely attractive for both transportation and stationary power applications. However, its low volumetric energy density causes considerable difficulties, inspiring intense efforts to develop chemical-based storage using metal hydrides, liquid organic hydrogen carriers and sorbents. The controlled uptake and release of hydrogen by these materials can be described as a series of challenges: optimal properties fall within a narrow range, can only be found in few materials and often involve important trade-offs. In addition, a greater understanding of the complex kinetics, mass transport and microstructural phenomena associated with hydrogen uptake and release is needed. The goal of this Perspective is to delineate potential use cases, define key challenges and show that solutions will involve a nexus of several subdisciplines of chemistry, including catalysis, data science, nanoscience, interfacial phenomena and dynamic or phase-change materials.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Arbabzadeh, M., Sioshansi, R., Johnson, J. X. & Keoleian, G. A. The role of energy storage in deep decarbonization of electricity production. Nat. Commun. 10, 3413 (2019).
The Future of Hydrogen (IEA, 2019).
Pivovar, B., Rustagi, N. & Satyapal, S. Hydrogen at scale (H2@Scale): key to a clean, economic, and sustainable energy system. Electrochem. Soc. Interface 27, 47–52 (2018).
The National Hydrogen Strategy (Federal Ministry for Economic Affairs and Climate Action, 2020).
Target Explanation Document: Onboard Hydrogen Storage for Light-Duty Fuel Cell Vehicles (US DOE, 2017).
Ahluwalia, R. K. et al. Technical assessment of cryo-compressed hydrogen storage tank systems for automotive applications. Int. J. Hydrog. Energy 35, 4171–4184 (2010).
Allendorf, M. D. et al. An assessment of strategies for the development of solid-state adsorbents for vehicular hydrogen storage. Energy Environ. Sci. 11, 2784–2812 (2018).
Pasini, J. M. et al. Metal hydride material requirements for automotive hydrogen storage systems. Int. J. Hydrog. Energy 38, 9755–9765 (2013).
Bhatia, S. K. & Myers, A. L. Optimum conditions for adsorptive storage. Langmuir 22, 1688–1700 (2006).
Witman, M. et al. Extracting an empirical intermetallic hydride design principle from limited data via interpretable machine learning. J. Phys. Chem. Lett. 11, 40–47 (2020).
Grubel, K., Jeong, H., Yoon, C. W. & Autrey, T. Challenges and opportunities for using formate to store, transport, and use hydrogen. J. Energy Chem. 41, 216–224 (2020).
Zou, Y.-Q., von Wolff, N., Anaby, A., Xie, Y. & Milstein, D. Ethylene glycol as an efficient and reversible liquid–organic hydrogen carrier. Nat. Catal. 2, 415–422 (2019).
Zhu, Q. L. & Xu, Q. Liquid organic and inorganic chemical hydrides for high-capacity hydrogen storage. Energy Environ. Sci. 8, 478–512 (2015).
Preuster, P., Papp, C. & Wasserscheid, P. Liquid organic hydrogen carriers (LOHCs): toward a hydrogen-free hydrogen economy. Acc. Chem. Res. 50, 74–85 (2017).
Mohtadi, R. & Orimo, S.-i The renaissance of hydrides as energy materials. Nat. Rev. Mater. 2, 16091 (2017).
Schneemann, A. et al. Nanostructured metal hydrides for hydrogen storage. Chem. Rev. 118, 10775–10839 (2018).
Chen, L.-N. et al. Efficient hydrogen production from methanol using a single-site Pt1/CeO2 catalyst. J. Am. Chem. Soc. 141, 17995–17999 (2019).
Sordakis, K. et al. Homogeneous catalysis for sustainable hydrogen storage in formic acid and alcohols. Chem. Rev. 118, 372–433 (2018).
Onishi, N., Laurenczy, G., Beller, M. & Himeda, Y. Recent progress for reversible homogeneous catalytic hydrogen storage in formic acid and in methanol. Coord. Chem. Rev. 373, 317–332 (2018).
Jeon, K. J. et al. Air-stable magnesium nanocomposites provide rapid and high-capacity hydrogen storage without using heavy-metal catalysts. Nat. Mater. 10, 286–290 (2011).
White, J. L. et al. The inside-outs of metal hydride dehydrogenation: imaging the phase evolution of the Li-N-H hydrogen storage system. Adv. Mater. Interfaces 7, 1901905 (2020).
Tsivion, E., Long, J. R. & Head-Gordon, M. Hydrogen physisorption on metal–organic framework linkers and metalated linkers: a computational study of the factors that control binding strength. J. Am. Chem. Soc. 136, 17827–17835 (2014).
Runcevski, T. et al. Adsorption of two gas molecules at a single metal site in a metal–organic framework. Chem. Commun. 52, 8251–8254 (2016).
Kapelewski, M. T. et al. Record high hydrogen storage capacity in the metal organic framework Ni2(m-dobdc) at near-ambient temperatures. Chem. Mater. 30, 8179–8189 (2018).
Jawahery, S. et al. Adsorbate-induced lattice deformation in IRMOF-74 series. Nat. Commun. 8, 13945 (2017).
Witman, M. et al. The influence of intrinsic framework flexibility on adsorption in nanoporous materials. J. Am. Chem. Soc. 139, 5547–5557 (2017).
Kanoo, P. et al. Pseudo-gated adsorption with negligible volume change evoked by halogen-bond interaction in the nanospace of MOFs. Chem. Eur. J. 26, 2148–2153 (2020).
Moosavi, S. M. et al. Capturing chemical intuition in synthesis of metal–organic frameworks. Nat. Commun. 10, 539 (2019).
Aaldto-Saksa, P. T., Cook, C., Kiviaho, J. & Repo, T. Liquid organic hydrogen carriers for transportation and storing of renewable energy—review and discussion. J. Power Sources 396, 803–823 (2018).
Gianotti, E., Taillades-Jacquin, M., Roziere, J. & Jones, D. J. High-purity hydrogen generation via dehydrogenation of organic carriers: a review on the catalytic process. ACS Catal. 8, 4660–4680 (2018).
Modisha, P. M., Ouma, C. N. M., Garidzirai, R., Wasserscheid, P. & Bessarabov, D. The prospect of hydrogen storage using liquid organic hydrogen carriers. Energy Fuels 33, 2778–2796 (2019).
Niermann, M., Beckendorff, A., Kaltschmitt, M. & Bonhoff, K. Liquid organic hydrogen carrier (LOHC)—assessment based on chemical and economic properties. Int. J. Hydrog. Energy 44, 6631–6654 (2019).
Clot, E., Eisenstein, O. & Crabtree, R. H.Computational structure-activity relationships in H2 storage: how placement of N atoms affects release temperatures in organic liquid storage materials. Chem. Commun. 2007, 2231–2233 (2007).
Cheng, H., Wu, J., Dong, Y. & Yang, M. Liquid hydrogen storage system. US patent 20180065849A1 (2015).
Verevkin, S. P., Konnova, M. E., Zherikova, K. V. & Pimerzin, A. A. Sustainable hydrogen storage: thermochemistry of amino-alcohols as seminal liquid organic hydrogen carriers. J. Chem. Thermo. 163, 106591 (2021).
Tran, B. L., Johnson, S. I., Brooks, K. P. & Autrey, S. T. Ethanol as a liquid organic hydrogen carrier for seasonal microgrid application: catalysis, theory, and engineering feasibility. ACS Sustain. Chem. Eng. 9, 7130–7138 (2021).
Onoda, M., Nagano, Y. & Fujita, K. Iridium-catalyzed dehydrogenative lactonization of 1,4-butanediol and reversal hydrogenation: new hydrogen storage system using cheap organic resources. Int. J. Hydrog. Energy 44, 28514–28520 (2019).
Hu, P., Fogler, E., Diskin-Posner, Y., Iron, M. A. & Milstein, D. A novel liquid organic hydrogen carrier system based on catalytic peptide formation and hydrogenation. Nat. Commun. 6, 6859 (2015).
Grubel, K. et al. Research requirements to move the bar forward using aqueous formate salts as H2 carriers for energy storage applications. J. Energy Power Technol. 2, 16 (2020).
Su, J., Lu, M. & Lin, H. F. High yield production of formate by hydrogenating CO2 derived ammonium carbamate/carbonate at room temperature. Green Chem. 17, 2769–2773 (2015).
Weisz, P. B. Diffusion and chemical transformation. Science 179, 433–440 (1973).
Weisz, P. B.The science of the possible. ChemTech 12, 424–425 (1982).
Peters, W. et al. Macrokinetic effects in perhydro-N-ethylcarbazole dehydrogenation and H2 productivity optimization by using egg-shell catalysts. Energy Environ. Sci. 8, 3013–3021 (2015).
Alhumaidan, F., Cresswell, D. & Garforth, A. Hydrogen storage in liquid organic hydride: producing hydrogen catalytically from methylcyclohexane. Energy Fuels 25, 4217–4234 (2011).
Andersson, J. Application of liquid hydrogen carriers in hydrogen steelmaking. Energies 14, 1392 (2021).
Yolcular, S. & Olgun, O. Ni/Al2O3 catalysts and their activity in dehydrogenation of methylcyclohexane for hydrogen production. Catal. Today 138, 198–202 (2008).
Al-ShaikhAli, A. H., Jedidi, A., Cavallo, L. & Takanabe, K. Non-precious bimetallic catalysts for selective dehydrogenation of an organic chemical hydride system. Chem. Commun. 51, 12931–12934 (2015).
Yamaguchi, R., Ikeda, C., Takahashi, Y. & Fujita, K. Homogeneous catalytic system for reversible dehydrogenation–hydrogenation reactions of nitrogen heterocycles with reversible interconversion of catalytic species. J. Am. Chem. Soc. 131, 8410–8412 (2009).
Hu, P., Ben-David, Y. & Milstein, D. Rechargeable hydrogen storage system based on the dehydrogenative coupling of ethylenediamine with ethanol. Angew. Chem. Int. Ed. 55, 1061–1064 (2016).
Snider, J. L. et al. Stabilized open metal sites in bimetallic metal–organic framework catalysts for hydrogen production from alcohols. J. Mater. Chem. A 9, 10869–10881 (2021).
Stavila, V. et al. IRMOF-74(n)–Mg: a novel catalyst series for hydrogen activation and hydrogenolysis of C–O bonds. Chem. Sci. 10, 9880–9892 (2019).
Gu, T. T., Gu, J., Zhang, Y. & Ren, H. Metal borohydride-based system for solid-state hydrogen storage. Prog. Chem. 32, 665–686 (2020).
Li, B., Li, J. D., Zhao, H. J., Yu, X. Q. & Shao, H. Y. Mg-based metastable nano alloys for hydrogen storage. Int. J. Hydrog. Energy 44, 6007–6018 (2019).
Sun, Y. H. et al. Tailoring magnesium based materials for hydrogen storage through synthesis: current state of the art. Energy Storage Mater. 10, 168–198 (2018).
Li, J. D., Li, B., Shao, H. Y., Li, W. & Lin, H. J. Catalysis and downsizing in Mg-based hydrogen storage materials. Catalysts 8, 89 (2018).
Yu, X. B., Tang, Z. W., Sun, D. L., Ouyang, L. Z. & Zhu, M. Recent advances and remaining challenges of nanostructured materials for hydrogen storage applications. Prog. Mater Sci. 88, 1–48 (2017).
Rusman, N. A. A. & Dahari, M. A review on the current progress of metal hydrides material for solid-state hydrogen storage applications. Int. J. Hydrog. Energy 41, 12108–12126 (2016).
Niaz, S., Manzoor, T. & Pandith, A. H. Hydrogen storage: materials, methods and perspectives. Renew. Sustain. Energy Rev. 50, 457–469 (2015).
Møller, K. T. et al. Complex metal hydrides for hydrogen, thermal and electrochemical energy storage. Energies 10, 1645 (2017).
Paskevicius, M. et al. Metal borohydrides and derivatives—synthesis, structure and properties. Chem. Soc. Rev. 46, 1565–1634 (2017).
Xiong, Z. T., Wu, G. T., Hu, H. J. & Chen, P. Ternary imides for hydrogen storage. Adv. Mater. 16, 1522–1525 (2004).
Luo, W. F.(LiNH2-MgH2): a viable hydrogen storage system. J. Alloys Compd. 381, 284–287 (2004).
Lu, J., Fang, Z. Z. G., Choi, Y. J. & Sohn, H. Y. Potential of binary lithium magnesium nitride for hydrogen storage applications. J. Phys. Chem. C 111, 12129–12134 (2007).
Wang, J. et al. Potassium-modified Mg(NH2)2/2 LiH system for hydrogen storage. Angew. Chem. Int. Ed. 48, 5828–5832 (2009).
He, T., Cao, H. J. & Chen, P. Complex hydrides for energy storage, conversion, and utilization. Adv. Mater. 31, 1902757 (2019).
Liu, X., Wu, C., Wu, F. & Bai, Y. Light metal complex hydride hydrogen storage systems. Prog. Chem. 27, 1167–1181 (2015).
Orimo, S. I., Nakamori, Y., Eliseo, J. R., Zuttel, A. & Jensen, C. M. Complex hydrides for hydrogen storage. Chem. Rev. 107, 4111–4132 (2007).
Ouyang, L., Chen, K., Jiang, J., Yang, X.-S. & Zhu, M. Hydrogen storage in light-metal based systems: a review. J. Alloys Compd. 829, 154597 (2020).
Berube, V., Radtke, G., Dresselhaus, M. & Chen, G. Size effects on the hydrogen storage properties of nanostructured metal hydrides: a review. Int. J. Energy Res. 31, 637–663 (2007).
Pal, P., Jain, A., Miyaoka, H., Kojima, Y. & Ichikawa, T. Eutectic melting in x(2LiBH4-MgH2) hydrogen storage system by the addition of KH. Int. J. Hydrog. Energy 45, 17000–17005 (2020).
Cho, E. S. et al. Hierarchically controlled inside-out doping of Mg nanocomposites for moderate temperature hydrogen storage. Adv. Funct. Mater. 27, 1704316 (2017).
Witman, M. et al. Data-driven discovery and synthesis of high entropy alloy hydrides with targeted thermodynamic stability. Chem. Mater. 33, 4067–4076 (2021).
Pinkerton, F. E., Meyer, M. S., Meisner, G. P., Balogh, M. P. & Vajo, J. J. Phase boundaries and reversibility of LiBH4/MgH2 hydrogen storage material. J. Phys. Chem. C 111, 12881–12885 (2007).
Lohstroh, W., Roth, A., Hahn, H. & Fichtner, M. Thermodynamic effects in nanoscale NaAlH4. ChemPhysChem 11, 789–792 (2010).
Paskevicius, M., Sheppard, D. A. & Buckley, C. E. Thermodynamic changes in mechanochemically synthesized magnesium hydride nanoparticles. J. Am. Chem. Soc. 132, 5077–5083 (2010).
White, J. L. et al. Identifying the role of dynamic surface hydroxides in the dehydrogenation of Ti-doped NaAlH4. ACS Appl. Mater. Interfaces 11, 4930–4941 (2019).
Frankcombe, T. J. Proposed mechanisms for the catalytic activity of Ti in NaAlH4. Chem. Rev. 112, 2164–2178 (2012).
Wood, B. C., Heo, T. W., Kang, S., Wan, L. F. & Li, S. Beyond idealized models of nanoscale metal hydrides for hydrogen storage. Ind. Eng. Chem. Res. 59, 5786–5796 (2020).
Griessen, R., Strohfeldt, N. & Giessen, H. Thermodynamics of the hybrid interaction of hydrogen with palladium nanoparticles. Nat. Mater. 15, 311–317 (2016).
Liu, W. & Aguey-Zinsou, K.-F. Synthesis of highly dispersed nanosized LaNi5 on carbon: revisiting particle size effects on hydrogen storage properties. Int. J. Hydrog. Energy 41, 14429–14436 (2016).
Zhao-Karger, Z. et al. Altered thermodynamic and kinetic properties of MgH2 infiltrated in microporous scaffold. Chem. Commun. 46, 8353–8355 (2010).
Cho, Y. et al. Reversing the irreversible: thermodynamic stabilization of LiAlH4 nanoconfined within a nitrogen-doped carbon host. ACS Nano 15, 10163–10174 (2021).
Stavila, V. et al. Defying thermodynamics: stabilization of alane within covalent triazine frameworks for reversible hydrogen storage. Angew. Chem. Int. Ed. 60, 25815–25824 (2021).
Rosi, N. L. Hydrogen storage in microporous metal–organic frameworks. Science 300, 1127–1129 (2003).
Murray, L. J., Dinca, M. & Long, J. R. Hydrogen storage in metal–organic frameworks. Chem. Soc. Rev. 38, 1294 (2009).
Anastasopoulou, A. et al. Technoeconomic analysis of metal–organic frameworks for bulk hydrogen transportation. Energy Environ. Sci. 14, 1083–1094 (2021).
Yang, C. & Ogden, J. Determining the lowest-cost hydrogen delivery mode. Int. J. Hydrog. Energy 32, 268–286 (2007).
Witman, M. et al. Design principles for the ultimate gas deliverable capacity material: nonporous to porous deformations without volume change. Mol. Syst. Des. Eng. 5, 1491–1503 (2020).
Kalmutzki, M. J., Hanikel, N. & Yaghi, O. M. Secondary building units as the turning point in the development of the reticular chemistry of MOFs. Sci. Adv. 4, eaat9180 (2018).
Kapelewski, M. T. et al. M2(m-dobdc) (M = Mg, Mn, Fe, Co, Ni) metal–organic frameworks exhibiting increased charge density and enhanced H2 binding at the open metal sites. J. Am. Chem. Soc. 2, 12119–12129 (2014).
Witman, M. et al. Rational design of a low-cost, high-performance metal–organic framework for hydrogen storage and carbon capture. J. Phys. Chem. C 121, 1171–1181 (2017).
Denysenko, D., Grzywa, M., Jelic, J., Reuter, K. & Volkmer, D. Scorpionate-type coordination in MFU-4l metal–organic frameworks: small-molecule binding and activation upon the thermally activated formation of open metal sites. Angew. Chem. Int. Ed. 53, 5832–5836 (2014).
Jaramillo, D. E. et al. Ambient-temperature hydrogen storage via vanadium(ii)–dihydrogen complexation in a metal–organic framework. J. Am. Chem. Soc. 143, 6248–6256 (2021).
Jaramillo, D. E. et al. Selective nitrogen adsorption via backbonding in a metal–organic framework with exposed vanadium sites. Nat. Mater. 19, 517–521 (2020).
Simon, C. M. et al. Optimizing nanoporous materials for gas storage. Phys. Chem. Chem. Phys. 16, 5499 (2014).
Kitagawa, S., Kitaura, R. & Noro, S.-I. Functional porous coordination polymers. Angew. Chem. Int. Ed. 43, 2334–2375 (2004).
Choi, H. J., Dincă, M. & Long, J. R. Broadly hysteretic H2 adsorption in the microporous metal−organic framework Co(1,4-benzenedipyrazolate). J. Am. Chem. Soc. 130, 7848–7850 (2008).
Paragian, K., Li, B., Massino, M. & Rangarajan, S. A computational workflow to discover novel liquid organic hydrogen carriers and their dehydrogenation routes. Mol. Syst. Des. Eng. 5, 1658–1670 (2020).
Hutcheon, M. J., Shipley, A. M. & Needs, R. J. Predicting novel superconducting hydrides using machine learning approaches. Phys. Rev. B 101, 144505 (2020).
Banerjee, D. et al. Metal–organic framework with optimally selective xenon adsorption and separation. Nat. Commun. 7, ncomms11831 (2016).
Goldsmith, J., Wong-Foy, A. G., Cafarella, M. J. & Siegel, D. J. Theoretical limits of hydrogen storage in metal–organic frameworks: opportunities and trade-offs. Chem. Mater. 25, 3373–3382 (2013).
Gomez-Gualdron, D. A., Wilmer, C. E., Farha, O. K., Hupp, J. T. & Snurr, R. Q. Exploring the limits of methane storage and delivery in nanoporous materials. J. Phys. Chem. C 118, 6941–6951 (2014).
Simon, C. M. et al. The materials genome in action: identifying the performance limits for methane storage. Energy Environ. Sci. 8, 1190–1199 (2015).
Simon, C. M., Mercado, R., Schnell, S. K., Smit, B. & Haranczyk, M. What are the best materials to separate a xenon/krypton mixture? Chem. Mater. 27, 4459–4475 (2015).
Ahmed, A. et al. Exceptional hydrogen storage achieved by screening nearly half a million metal–organic frameworks. Nat. Commun. 10, 1568 (2019).
Bobbitt, N. S., Chen, J. & Snurr, R. Q. High-throughput screening of metal–organic frameworks for hydrogen storage at cryogenic temperature. J. Phys. Chem. C 120, 27328–27341 (2016).
Thornton, A. W. et al. Materials genome in action: identifying the performance limits of physical hydrogen storage. Chem. Mater. 29, 2844–2854 (2017).
Rappe, A. K., Casewit, C. J., Colwell, K. S., Goddard, W. A. & Skiff, W. M. UFF, a full periodic table force field for molecular mechanics and molecular dynamics simulations. J. Am. Chem. Soc. 114, 10024–10035 (1992).
Dzubak, A. L. et al. Ab initio carbon capture in open-site metal–organic frameworks. Nat. Chem. 4, 810–816 (2012).
Lee, K., Howe, J. D., Lin, L.-C., Smit, B. & Neaton, J. B. Small-molecule adsorption in open-site metal–organic frameworks: a systematic density functional theory study for rational design. Chem. Mater. 27, 668–678 (2015).
Lin, L.-C., Lee, K., Gagliardi, L., Neaton, J. B. & Smit, B. Force-field development from electronic structure calculations with periodic boundary conditions: applications to gaseous adsorption and transport in metal–organic frameworks. J. Chem. Theory Comput. 10, 1477–1488 (2014).
Pham, T. et al. Understanding the H2 sorption trends in the M-MOF-74 series (M = Mg, Ni, Co, Zn). J. Phys. Chem. C 119, 1078–1090 (2015).
Pham, T., Forrest, K. A., McLaughlin, K., Eckert, J. & Space, B. Capturing the H2–metal interaction in Mg-MOF-74 using classical polarization. J. Phys. Chem. C 118, 22683–22690 (2014).
Pham, T. et al. Simulations of hydrogen sorption in rht-MOF-1: identifying the binding sites through explicit polarization and quantum rotation calculations. J. Mater. Chem. A 2, 2088–2100 (2014).
Chung, Y. G. et al. Advances, updates, and analytics for the computation-ready, experimental metal–organic framework database: CoRE MOF 2019. J. Chem. Eng. Data 64, 5985–5998 (2019).
Alapati, S. V., Johnson, J. K. & Sholl, D. S. Identification of destabilized metal hydrides for hydrogen storage using first principles calculations. J. Phys. Chem. B 110, 8769–8776 (2006).
Jain, A. et al. Commentary: the Materials Project: a materials genome approach to accelerating materials innovation. APL Mater. 1, 011002 (2013).
Saal, J. E., Kirklin, S., Aykol, M., Meredig, B. & Wolverton, C. Materials design and discovery with high-throughput density functional theory: the Open Quantum Materials Database (OQMD). JOM 65, 1501–1509 (2013).
Nazarian, D., Ganesh, P. & Sholl, D. S. Benchmarking density functional theory predictions of framework structures and properties in a chemically diverse test set of metal–organic frameworks. J. Mater. Chem. A 3, 22432–22440 (2015).
Hattrick-Simpers, J. R., Choudhary, K. & Corgnale, C. A simple constrained machine learning model for predicting high-pressure-hydrogen-compressor materials. Mol. Syst. Des. Eng. 3, 509–517 (2018).
Bartok, A. P., Payne, M. C., Kondor, R. & Csanyi, G. Gaussian approximation potentials: the accuracy of quantum mechanics, without the electrons. Phys. Rev. Lett. 104, 136403 (2010).
Behler, J. & Parrinello, M. Generalized neural-network representation of high-dimensional potential-energy surfaces. Phys. Rev. Lett. 98, 146401 (2007).
Jinnouchi, R., Lahnsteiner, J., Karsai, F., Kresse, G. & Bokdam, M. Phase transitions of hybrid perovskites simulated by machine-learning force fields trained on the fly with Bayesian inference. Phys. Rev. Lett. 122, 225701 (2019).
Suh, M. P., Park, H. J., Prasad, T. K. & Lim, D.-W. Hydrogen storage in metal–organic frameworks. Chem. Rev. 112, 782–835 (2012).
Allendorf, M. D., Stavila, V., Witman, M., Brozek, C. K. & Hendon, C. H. What lies beneath a metal–organic framework crystal structure? New design principles from unexpected behaviors. J. Am. Chem. Soc. 143, 6705–6723 (2021).
Technology Roadmap—Hydrogen and Fuel Cells (IEA, 2015).
Cao, W., Zhang, J. & Li, H. Batteries with high theoretical energy densities. Energy Storage Mater. 26, 46–55 (2020).
May, G. J., Davidson, A. & Monahov, B. Lead batteries for utility energy storage: a review. J. Energy Storage 15, 145–157 (2018).
Pasquini, L. Design of nanomaterials for hydrogen storage. Energies 13, 3503 (2020).
Klebanoff, L. E. Hydrogen Storage Technology: Materials and Applications (CRC Press, 2013).
Chen, P., Xiong, Z., Luo, J., Lin, J. & Tan, K. L. Interaction of hydrogen with metal nitrides and imides. Nature 420, 302–304 (2002).
Ding, Z. et al. LiBH4 for hydrogen storage—new perspectives. Nano Mater. Sci. 2, 109–119 (2020).
Liu, M. et al. Novel 1D carbon nanotubes uniformly wrapped nanoscale MgH2 for efficient hydrogen storage cycling performances with extreme high gravimetric and volumetric capacities. Nano Energy 61, 540–549 (2019).
Von Colbe, J. B. et al. Application of hydrides in hydrogen storage and compression: achievements, outlook and perspectives. Int. J. Hydrog. Energy 44, 7780–7808 (2019).
Zavorotynska, O., El-Kharbachi, A., Deledda, S. & Hauback, B. C. Recent progress in magnesium borohydride Mg(BH4)2: fundamentals and applications for energy storage. Int. J. Hydrog. Energy 41, 14387–14403 (2016).
Zidan, R. et al. Aluminium hydride: a reversible material for hydrogen storage. Chem. Commun. 2009, 3717–3719 (2009).
Hydrogen and Fuel Cell Technologies Office Multi-Year Research, Development, and Demonstration Plan Section 3.3 (Hydrogen and Fuel Cell Technologies Office, 2015).
Brooks, K. P., Semelsberger, T. A., Simmons, K. L. & van Hassel, B. Slurry-based chemical hydrogen storage systems for automotive fuel cell applications. J. Power Sources 268, 950–959 (2014).
Tamburello, D., Hardy, B., Corgnale, C., Sulic, M. & Anton, D. Cryo-adsorbent hydrogen storage systems for fuel cell vehicles. In Proc. ASME 2017 Fluids Engineering Division Summer Meeting (American Society of Mechanical Engineers, 2017).
Bartscher, W., Rebizant, J. & Haschke, J. M. Equilibria and thermodynamic properties of the ThZr2·H system. J. Less Common Met. 136, 385–394 (1988).
Vajo, J. J., Mertens, F., Ahn, C. C., Bowman, R. C. & Fultz, B. Altering hydrogen storage properties by hydride destabilization through alloy formation: LiH and MgH2 destabilized with Si. J. Phys. Chem. B 108, 13977–13983 (2004).
Cahen, S., Eymery, J.-B., Janot, R. & Tarascon, J.-M. Improvement of the LiBH4 hydrogen desorption by inclusion into mesoporous carbons. J. Power Sources 189, 902–908 (2009).
Aakko-Saksa, P. T., Cook, C., Kiviaho, J. & Repo, T. Liquid organic hydrogen carriers for transportation and storing of renewable energy—review and discussion. J. Power Sources 396, 803–823 (2018).
Sahlberg, M., Karlsson, D., Zlotea, C. & Jansson, U. Superior hydrogen storage in high entropy alloys. Sci. Rep. 6, 36770 (2016).
Reilly, J. J. & Wiswall, R. H. Formation and properties of iron titanium hydride. Inorg. Chem. 13, 218–222 (1974).
Marcinkoski, J. et al. Hydrogen Class 8 Long Haul Truck Targets. Report No. 19006 (Hydrogen and Fuel Cell Technologies Office, US DOE, 2019).
Brewer, E & Hanlin, J. Fuel Cell Hybrid Electric Delivery Van Project. In 2021 Annual Merit Review and Peer Evaluation Meeting (Center for Transportation and the Environment, 2021); https://www.hydrogen.energy.gov/pdfs/review21/ta016_hanlin_2021_o.pdf
Pratt, J. W. & Klebanoff, L. E. Feasibility of the SF-BREEZE: a Zero-Emission, Hydrogen Fuel Cell, High-Speed Passenger Ferry. Report No. SAND2016-9719 (Sandia National Laboratories, 2016).
FY 2018 Progress Report for the DOE Hydrogen and Fuel Cells Program. Report No. DOE/GO-102019-5156 (US DOE, 2019).
Devlin, P. & Moreland, G. Industry Deployed Fuel Cell Backup Power (BuP). Report No. 17004 (Hydrogen and Fuel Cell Technologies Office, US DOE, 2017).
Pauksztat, A., Saliger, R. & Boyanov, N. Residential energy supply concept with integration of renewable energies and energy storage. Chem. Eng. Technol. 42, 1907–1913 (2019).
Kumagai, T. AHEAD Launches Brunei–Japan Hydrogen Supply Chain for Power Generation in Tokyo Bay (S&P Global, 2020).
Energy Savings Analysis: ANSI/ASHRAE/IES Standard 90.1-2016 (Office of Energy Efficiency & Renewable Energy & US DOE, 2017).
Saur, G., Arjona, V., Clutterbuck, A. & Parker, E. Hydrogen and Fuel Cells for Data Center Applications Project Meeting: Workshop Report (National Renewable Energy Laboratory, 2019).
Mongird, K. et al. 2020 Grid Energy Storage Technology Cost and Performance Assessment. Report No. DOE/PA-0204 (US DOE, 2020).
Acknowledgements
We gratefully acknowledge research support from the Hydrogen Materials Advanced Research Consortium (HyMARC), which was established as part of the Energy Materials Network under the US DOE Office of Energy Efficiency and Renewable Energy’s Hydrogen and Fuel Cell Technologies Office, under contract numbers DE-AC04-94AL85000 and DE-AC52-07NA27344. Sandia National Laboratories is a multi-mission laboratory managed and operated by National Technology and Engineering Solutions of Sandia—a wholly owned subsidiary of Honeywell International—for the US DOE’s National Nuclear Security Administration under contract DE-NA-0003525. The Pacific Northwest National Laboratory is operated by Battelle for the US DOE under contract DE-AC05-76RL01830.
Author information
Authors and Affiliations
Contributions
M.D.A., V.S., M.W., J.L.S., T.A., M.E.B., K.B. and B.L.T. contributed to discussions and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Linear regression of entropy and enthalpy of dehydrogenation for bulk and nanoscale hydrides.
Figure 1 shows linear least-squares fits to the thermodynamic data for (a) bulk and (b) nanoscale hydrides. The bulk data, which are a subset of the full HydPARK database10, at best show a weak correlation between the entropy ΔS° and enthalpy ΔH° of H2 dehydrogenation, as indicated by the low values of R2 and the Spearman Rank Correlation Coefficient R. Excluding outlier compositions, as detailed in Ref. 10, improves the fit somewhat, yielding R2 = 0.42 and R=0.68 across the entire ML-ready HydPARK dataset, suggesting a moderate correlation. In contrast, the data for nanoscale hydrides, although admittedly limited, exhibit a fairly strong correlation, with R2 = 0.738 and R = 0.891. Within specific hydride classes, stronger ΔH° and ΔS° correlations can be found. For example, for nano-PdH R2 = 0.954 and R = 0.939 and for bulk AB materials R2 = 0.924 and R = 0.964.
Supplementary information
Supplementary Information
Supplementary Fig. 1 and Tables 1 and 2, Supplementary Discussion and Supplementary References.
Supplementary Data 1
Source data for Supplementary Fig. 1 (thermodynamics data for hydrogen release from nanoscale metal hydrides).
Source data
Source Data Fig. 1
Energy and specific energy for various hydrogen storage materials.
Source Data Fig. 3
Enthalpy and entropy of H2 release from bulk and nanoscale metal hydrides.
Source Data Extended Data Fig. 1
Thermodynamics data for hydrogen release from bulk metal hydrides.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Allendorf, M.D., Stavila, V., Snider, J.L. et al. Challenges to developing materials for the transport and storage of hydrogen. Nat. Chem. 14, 1214–1223 (2022). https://doi.org/10.1038/s41557-022-01056-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-022-01056-2
This article is cited by
-
Experimentally validated design principles of heteroatom-doped-graphene-supported calcium single-atom materials for non-dissociative chemisorption solid-state hydrogen storage
Nature Communications (2024)
-
Analyzing microstructure relationships in porous copper using a multi-method machine learning-based approach
Communications Materials (2024)
-
Decoupled electrolysis for hydrogen production and hydrazine oxidation via high-capacity and stable pre-protonated vanadium hexacyanoferrate
Nature Communications (2024)
-
Light-driven ammonia synthesis under mild conditions using lithium hydride
Nature Chemistry (2024)
-
Spectroscopic visualization of reversible hydrogen spillover between palladium and metal–organic frameworks toward catalytic semihydrogenation
Nature Communications (2024)