Abstract
Transferring structural information from the nanoscale to the macroscale is a promising strategy for developing adaptive and dynamic materials. Here we demonstrate that the knotting and unknotting of a molecular strand can be used to control, and even invert, the handedness of a helical organization within a liquid crystal. An oligodentate tris(2,6-pyridinedicarboxamide) strand with six point-chiral centres folds into an overhand knot of single handedness upon coordination to lanthanide ions, both in isotropic solutions and in liquid crystals. In achiral liquid crystals, dopant knotted and unknotted strands induce supramolecular helical organizations of opposite handedness, with dynamic switching achievable through in situ knotting and unknotting events. Tying the molecular knot transmits information regarding asymmetry across length scales, from Euclidean point chirality (constitutional chirality) via molecular entanglement (conformation) to liquid-crystal (centimetre-scale) chirality. The magnitude of the effect induced by the tying of the molecular knots is similar to that famously used to rotate a glass rod on the surface of a liquid crystal by synthetic molecular motors.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the paper and its Supplementary Information, or are available from the Mendeley data repository (https://bit.ly/3e5chOf) under https://doi.org/10.17632/dw93cmxxs4.2.
References
Jackson, S. E., Suma, A. & Micheletti, C. How to fold intricately: using theory and experiments to unravel the properties of knotted proteins. Curr. Opin. Struct. Biol. 42, 6–14 (2017).
Wasserman, S. A. & Cozzarelli, N. R. Biochemical topology: applications to DNA recombination and replication. Science 232, 951–960 (1986).
Frank-Kamenetskii, M. D., Lukashin, A. V. & Vologodskii, A. V. Statistical mechanics and topology of polymer chains. Nature 258, 398–402 (1975).
Sułkowska, J. I., Sułkowski, P., Szymczak, P. & Cieplak, M. Stabilizing effect of knots on proteins. Proc. Natl Acad. Sci. USA 105, 19714–19719 (2008).
Saitta, A. M., Soper, P. D., Wasserman, E. & Klein, M. L. Influence of a knot on the strength of a polymer strand. Nature 399, 46–48 (1999).
Caraglio, M., Micheletti, C. & Orlandini, E. Stretching response of knotted and unknotted polymer chains. Phys. Rev. Lett. 115, 188301 (2015).
Amin, S., Khorshid, A., Zeng, L., Zimny, P. & Reisner, W. A nanofluidic knot factory based on compression of single DNA in nanochannels. Nat. Commun. 9, 1506 (2018).
Fielden, S. D. P., Leigh, D. A. & Woltering, S. L. Molecular knots. Angew. Chem. Int. Ed. 56, 11166–11194 (2017).
Forgan, R. S., Sauvage, J.-P. & Stoddart, J. F. Chemical topology: complex molecular knots, links, and entanglements. Chem. Rev. 111, 5434–5464 (2011).
Sauvage, J.-P. From chemical topology to molecular machines (Nobel lecture). Angew. Chem. Int. Ed. 56, 11080–11093 (2017).
Lim, N. C. H. & Jackson, S. E. Molecular knots in biology and chemistry. J. Phys. Condens. Matter 27, 354101 (2015).
Goujon, A. et al. Hierarchical self-assembly of supramolecular muscle-like fibers. Angew. Chem. Int. Ed. 55, 703–707 (2016).
Yamaguchi, H. et al. Photoswitchable gel assembly based on molecular recognition. Nat. Commun. 3, 603 (2012).
Cantekin, S. et al. The effect of isotopic substitution on the chirality of a self-assembled helix. Nat. Chem. 3, 42–46 (2011).
Berná, J. et al. Macroscopic transport by synthetic molecular machines. Nat. Mater. 4, 704–710 (2005).
Zhu, K., O’Keefe, C. A., Vukotic, V. A., Schurko, R. W. & Loeb, S. J. A molecular shuttle that operates inside a metal–organic framework. Nat. Chem. 7, 514–519 (2015).
Danowski, W. et al. Unidirectional rotary motion in a metal–organic framework. Nat. Nanotechnol. 14, 488–494 (2019).
Eelkema, R. et al. Nanomotor rotates microscale objects. Nature 440, 163 (2006).
Sakuda, J., Yasuda, T. & Kato, T. Liquid-crystalline catenanes and rotaxanes. Isr. J. Chem. 52, 854–862 (2012).
Baranoff, E. D. et al. A liquid-crystalline [2]catenane and its copper(I) complex. Angew. Chem. Int. Ed. 46, 4680–4683 (2007).
Aprahamian, I. et al. A liquid-crystalline bistable [2]rotaxane. Angew. Chem. Int. Ed. 46, 4675–4679 (2007).
Lancia, F., Ryabchun, A. & Katsonis, N. Life-like motion driven by artificial molecular machines. Nat. Rev. Chem. 3, 536–551 (2019).
Morrow, S. M., Bissette, A. J. & Fletcher, S. P. Transmission of chirality through space and across length scales. Nat. Nanotechnol. 12, 410–419 (2017).
Yashima, E. et al. Supramolecular helical systems: helical assemblies of small molecules, foldamers, and polymers with chiral amplification and their functions. Chem. Rev. 116, 13752–13990 (2016).
Katsonis, N., Lacaze, E. & Ferrarini, A. Controlling chirality with helix inversion in cholesteric liquid crystals. J. Mater. Chem. 22, 7088–7097 (2012).
Tai, J.-S. B. & Smalyukh, I. I. Three-dimensional crystals of adaptive knots. Science 365, 1449–1453 (2019).
Alexander, G. P. Knot your regular crystal of atoms. Science 365, 1377 (2019).
Tkalec, U., Ravnik, M., Čopar, S., Žumer, S. & Muševič, I. Reconfigurable knots and links in chiral nematic colloids. Science 333, 62–66 (2011).
Dang, L.-L. et al. Coordination-driven self-assembly of a molecular figure-eight knot and other topologically complex architectures. Nat. Commun. 10, 2057 (2019).
Danon, J. J. et al. Braiding a molecular knot with eight crossings. Science 355, 159–162 (2017).
Dietrich-Buchecker, C. O. & Sauvage, J.-P. A synthetic molecular trefoil knot. Angew. Chem. Int. Ed. 28, 189–192 (1989).
Guo, J., Mayers, P. C., Breault, G. A. & Hunter, C. A. Synthesis of a molecular trefoil knot by folding and closing on an octahedral coordination template. Nat. Chem. 2, 218–222 (2010).
Barran, P. E. et al. Active metal template synthesis of a molecular trefoil knot. Angew. Chem. Int. Ed. 50, 12280–12284 (2011).
Safarowsky, O., Nieger, M., Fröhlich, R. & Vögtle, F. A molecular knot with twelve amide groups—one-step synthesis, crystal structure, chirality. Angew. Chem. Int. Ed. 39, 1616–1618 (2000).
Ponnuswamy, N., Cougnon, F. B. L., Clough, J. M., Pantos, G. D. & Sanders, J. K. M. Discovery of an organic trefoil knot. Science 338, 783–785 (2012).
Cougnon, F. B. L., Caprice, K., Pupier, M., Bauza, A. & Frontera, A. A strategy to synthesize molecular knots and ́links using the hydrophobic effect. J. Am. Chem. Soc. 140, 12442–12450 (2018).
Marcos, V. et al. Allosteric initiation and regulation of catalysis with a molecular knot. Science 352, 1555–1559 (2016).
Zhang, L. et al. Effects of knot tightness at the molecular level. Proc. Natl Acad. Sci. USA 116, 2452–2457 (2019).
Leigh, D. A., Pirvu, L., Schaufelberger, F., Tetlow, D. J. & Zhang, L. Securing a supramolecular architecture by tying a stopper knot. Angew. Chem. Int. Ed. 57, 10484–10488 (2018).
Ayme, J.-F. et al. Strong and selective anion binding within the central cavity of molecular knots and links. J. Am. Chem. Soc. 137, 9812–9815 (2015).
Benyettou, F. et al. Potent and selective in vitro and in vivo antiproliferative effects of metal-organic trefoil knots. Chem. Sci. 10, 5884–5892 (2019).
Gil-Ramírez, G. et al. Tying a molecular overhand knot of single handedness and asymmetric catalysis with the corresponding pseudo-D3-symmetric trefoil knot. J. Am. Chem. Soc. 138, 13159–13162 (2016).
Kitchen, J. A. Lanthanide-based self-assemblies of 2,6-pyridyldicarboxamide ligands: recent advances and applications as next-generation luminescent and magnetic materials. Coord. Chem. Rev. 340, 232–246 (2017).
Barry, D. E., Caffrey, D. F. & Gunnlaugsson, T. Lanthanide-directed synthesis of luminescent self-assembly supramolecular structures and mechanically bonded systems from acyclic coordinating organic ligands. Chem. Soc. Rev. 45, 3244–3274 (2016).
Zhang, G. et al. Lanthanide template synthesis of trefoil knots of single handedness. J. Am. Chem. Soc. 137, 10437–10442 (2015).
Leigh, D. A., Pirvu, L. & Schaufelberger, F. Stereoselective synthesis of molecular square and granny knots. J. Am. Chem. Soc. 141, 6054–6059 (2019).
Pieraccini, S., Masiero, S., Ferrarini, A. & Spada, G. P. Chirality transfer across length-scales in nematic liquid crystals: fundamentals and applications. Chem. Soc. Rev. 40, 258–271 (2011).
Eelkema, R. & Feringa, B. L. Macroscopic expression of the chirality of amino alcohols by a double amplification mechanism in liquid crystalline media. J. Am. Chem. Soc. 127, 13480–13481 (2005).
Zahn, S., Proni, G., Spada, G. P. & Canary, J. W. Supramolecular detection of metal ion binding: ligand conformational control of cholesteric induction in nematic liquid crystalline phases. Chem. Eur. J. 7, 88–93 (2001).
Meudtner, R. M. & Hecht, S. Helicity inversion in responsive foldamers induced by achiral halide ion guests. Angew. Chem. Int. Ed. 47, 4926–4930 (2008).
Ryabchun, A. & Bobrovsky, A. Cholesteric liquid crystal materials for tunable diffractive optics. Adv. Opt. Mater. 6, 1800335 (2018).
Bisoyi, H. K., Bunning, T. J. & Li, Q. Stimuli-driven control of the helical axis of self-organized soft helical superstructures. Adv. Mater. 30, 1706512 (2018).
Orlova, T. et al. Revolving supramolecular chiral structures powered by light in nanomotor-doped liquid crystals. Nat. Nanotechnol. 13, 304–308 (2018).
Mathews, M. et al. Light-driven reversible handedness inversion in self-organized helical superstructures. J. Am. Chem. Soc. 132, 18361–18366 (2010).
De Gennes, P. G. & Prost, J. The Physics of Liquid Crystals (Oxford Univ. Press, 1997).
Kasyanyuk, D., Slyusarenko, K., West, J., Vasnetsov, M. & Reznikov, Y. Formation of liquid-crystal cholesteric pitch in the centimeter range. Phys. Rev. E 89, 022503 (2014).
Nemati, A. et al. Chirality amplification by desymmetrization of chiral ligand-capped nanoparticles to nanorods quantified in soft condensed matter. Nat. Commun. 9, 3908 (2018).
Ishida, Y. et al. Tunable chiral reaction media based on two-component liquid crystals: regio-, diastereo-, and enantiocontrolled photodimerization of anthracenecarboxylic acids. J. Am. Chem. Soc. 132, 17435–17446 (2010).
Akagi, K. et al. Helical polyacetylene synthesized with a chiral nematic reaction field. Science 282, 1683–1686 (1998).
Thitamadee, S., Tuchihara, K. & Hashimoto, T. Microtubule basis for left-handed helical growth in Arabidopsis. Nature 417, 193–196 (2002).
Gerber, P. R. On the determination of the cholesteric screw sense by the Grandjean-Cano-method. Z. Naturforsch. 35, 619–622 (1980).
Stalder, M. & Schadt, M. Linearly polarized light with axial symmetry generated by liquid-crystal polarization converters. Opt. Lett. 21, 1948–1950 (1996).
Acknowledgements
We thank the Engineering and Physical Sciences Research Council (EPSRC; EP/P027067/1), the Dutch Research Council (Projectruimte, 13PR3105), the European Research Council (ERC Consolidator Grant to N.K., 772564; ERC Advanced Grant to D.A.L., 786630) and the Marie Skłodowska-Curie Actions of the European Union (Individual Postdoctoral Fellowship to F.S., EC 746993) for funding, the University of Manchester Mass Spectrometry Service Centre for high-resolution mass spectrometry and the COST Action CA17139, EUTOPIA, for networking costs. D.A.L. is a Royal Society Research Professor.
Author information
Authors and Affiliations
Contributions
F.S. and L.P. planned and carried out the synthetic work. F.L. and A.R. designed and performed the liquid crystal experiments. D.A.L. and N.K. directed the research. All authors contributed to the analysis of the results and the writing of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
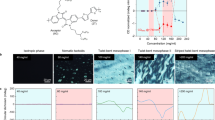
Extended Data Fig. 1 Effect of strand and knot doping in different liquid crystalline hosts.
a, Molecular structures of ZLI-1083 and 5CB which form liquid crystalline phases. b, Table summarizing HTP values for strand 1–3 and knots Λ-Lu1–Λ-Lu3 in ZLI-1083 and 5CB. c, Polarized optical micrograph images (Grandjean-Cano lines and θ-cell measurements) highlighting the chiral helical organization induced by strands 1–3 and corresponding knots in different liquid crystalline hosts. The dashed line shows the rubbing direction of the top substrate. The red arrow shows the rotation of disclination line. Scale bars corresponds to 200 μm. d, CD spectra on ligands 1 (d) and 2 (e) in different solvent mixtures (0.025 mM, 298 K), highlighting how the chiral expressions change with decreasing polarity. Normalized for absorbance.
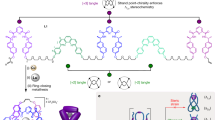
Extended Data Fig. 2 Probing the effect of the knotted conformation with macrocyclic strands.
a, Treatment of macrocycle 5 with Lu(OTf)3 does not produce a trefoil knot due to topological restrictions, nor does this operation invert macroscopic chirality in liquid crystals. Conditions: Lu(OTf)3, MeCN, 80 °C, 24 h. b, CD spectra of the mixture obtained upon complexation of 5 with Lu(OTf)3 (purple trace, MeCN, 298 K) overlaid with reference knot spectrum belonging to Λ-Lu1 (green trace, MeCN, 298 K). Normalized for absorbance. c, d, Polarized optical image of θ-cell filled with ZLI-1083 nematic host doped with 0.1 wt% of 5 after complexation with Lu(OTf)3 (c) and after decomplexation with tetramethylammonium fluoride (d). Notice that handedness of the chiral nematic liquid crystal is not modified. Thickness of the θ-cell is 50 µm. Scale bars correspond to 200 μm.
Supplementary information
Supplementary Information
General experimental information, synthetic procedures, Supplementary Figs. 1–27, Schemes 1–10, Spectra 1–28 and Table 1.
Rights and permissions
About this article
Cite this article
Katsonis, N., Lancia, F., Leigh, D.A. et al. Knotting a molecular strand can invert macroscopic effects of chirality. Nat. Chem. 12, 939–944 (2020). https://doi.org/10.1038/s41557-020-0517-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-020-0517-1
This article is cited by
-
Mechanical scission of a knotted polymer
Nature Chemistry (2024)
-
Construction of a molecular prime link by interlocking two trefoil knots
Nature Synthesis (2022)
-
A molecular endless (74) knot
Nature Chemistry (2021)
-
Open questions in functional molecular topology
Communications Chemistry (2020)