Abstract
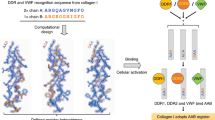
Robust methods for predicting thermal stabilities of collagen triple helices are critical for understanding natural structure and stability in the collagen family of proteins and also for designing synthetic peptides mimicking these essential proteins. In this work, we determine the relative stability imparted on the collagen triple helix by single amino acids and interactions between amino acid pairs. Using this analysis, we create a comprehensive algorithm, SCEPTTr, for predicting melting temperatures of synthetic triple helices. Critically, our algorithm is compatible with every natural amino acid, can evaluate both homotrimers and heterotrimers, and accounts for all possible helix compositions and registers, including non-canonically staggered helices. We test and optimize our algorithm against 431 published collagen triple helices to demonstrate the quality of our predictive system. Finally, we use this algorithm to successfully guide the design of an ABC heterotrimer possessing high assembly specificity.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the paper, in the Supplementary Information and from the corresponding author. In particular, the NMR raw data are available on request due to the size of the files and the lack of appropriate public repositories for raw multi-dimensional NMR data.
Code availability
A compiled standalone Java application is available for download as Supplementary Software 1. The genetic algorithm used to optimize SCEPTTr requires human input to further optimize the values used. For this reason, it is available from the corresponding author upon reasonable request.
References
Celerin, M. et al. Fungal fimbriae are composed of collagen. EMBO J. 15, 4445–4453 (1996).
Rasmussen, M., Jacobsson, M. & Björck, L. Genome-based identification and analysis of collagen-related structural motifs in bacterial and viral proteins. J. Biol. Chem. 278, 32313–32316 (2003).
Thiel, S. Complement activating soluble pattern recognition molecules with collagen-like regions, mannan-binding lectin, ficolins and associated proteins. Mol. Immunol. 44, 3875–3888 (2007).
Yu, Z., An, B., Ramshaw, J. A. M. & Brodsky, B. Bacterial collagen-like proteins that form triple-helical structures. J. Struct. Biol. 186, 451–461 (2014).
O’Shea, E. K., Klemm, J. D., Kim, P. S. & Alber, T. X-ray structure of the GCN4 leucine zipper, a two-stranded, parallel coiled coil. Science 254, 539–544 (1991).
Landschulz, W. H., Johnson, P. F. & McKnight, S. L. The leucine zipper: a hypothetical structure common to a new class of DNA binding proteins. Science 240, 1759–1764 (1988).
O’Shea, E. K., Rutkowski, R., Stafford, W. F. III & Kim, P. S. Preferential heterodimer formation by isolated leucine zippers from fos and jun. Science 245, 646–648 (1989).
Lovejoy, B. et al. Crystal structure of a synthetic triple-stranded α-helical bundle. Science 259, 1288–1293 (1993).
Zimenkov, Y., Conticello, V. P., Guo, L. & Thiyagarajan, P. Rational design of a nanoscale helical scaffold derived from self-assembly of a dimeric coiled coil motif. Tetrahedron 60, 7237–7246 (2004).
Zimenkov, Y. et al. Rational design of a reversible pH-responsive switch for peptide self-assembly. J. Am. Chem. Soc. 128, 6770–6771 (2006).
Moutevelis, E. & Woolfson, D. N. A periodic table of coiled-coil protein structures. J. Mol. Biol. 385, 726–732 (2009).
Fallas, J. A. et al. Computational design of self-assembling cyclic protein homo-oligomers. Nat. Chem. 9, 353–360 (2017).
Rhys, G. G. et al. Maintaining and breaking symmetry in homomeric coiled-coil assemblies. Nat. Commun. 9, 4132 (2018).
Koepnick, B. et al. De novo protein design by citizen scientists. Nature 570, 390–394 (2019).
Persikov, A. V., Ramshaw, J. A. M. & Brodsky, B. Prediction of collagen stability from amino acid sequence. J. Biol. Chem. 280, 19343–19349 (2005).
Holmgren, S. K., Bretscher, L. E., Taylor, K. M. & Raines, R. T. A hyperstable collagen mimic. Chem. Biol. 6, 63–70 (1999).
Gauba, V. & Hartgerink, J. D. Self-Assembled heterotrimeric collagen triple helices directed through electrostatic interactions. J. Am. Chem. Soc. 129, 2683–2690 (2007).
Jalan, A. A., Jochim, K. A. & Hartgerink, J. D. Rational design of a non-canonical “sticky-ended” collagen triple helix. J. Am. Chem. Soc. 136, 7535–7538 (2014).
Persikov, A. V., Ramshaw, J. A. M., Kirkpatrick, A. & Brodsky, B. Peptide investigations of pairwise interactions in the collagen triple-helix. J. Mol. Biol. 316, 385–394 (2002).
Persikov, A. V., Ramshaw, J. A. M., Kirkpatrick, A. & Brodsky, B. Electrostatic interactions involving lysine make major contributions to collagen triple-helix stability. Biochemistry 44, 1414–1422 (2005).
Fallas, J. A., Dong, J., Tao, Y. J. & Hartgerink, J. D. Structural insights into charge pair interactions in triple helical collagen-like proteins. J. Biol. Chem. 287, 8039–8047 (2012).
Wei, F., Fallas, J. A. & Hartgerink, J. D. Sequence position and side chain length dependence of charge pair interactions in collagen triple helices. Macromol. Rapid Commun. 33, 1445–1452 (2012).
Xu, F., Zhang, L., Koder, R. L. & Nanda, V. De novo self-assembling collagen heterotrimers using explicit positive and negative design. Biochemistry 49, 2307–2316 (2010).
Chen, C.-C. et al. Contributions of cation–π interactions to the collagen triple helix stability. Arch. Biochem. Biophys. 508, 46–53 (2011).
Xu, F., Zahid, S., Silva, T. & Nanda, V. Computational design of a collagen A:B:C-type heterotrimer. J. Am. Chem. Soc. 133, 15260–15263 (2011).
Parmar, A. S., Joshi, M., Nosker, P. L., Hasan, N. F. & Nanda, V. Control of collagen stability and heterotrimer specificity through repulsive electrostatic interactions. Biomolecules 3, 986–996 (2013).
Acevedo-Jake, A. M., Ngo, D. H. & Hartgerink, J. D. Control of collagen triple helix stability by phosphorylation. Biomacromolecules 18, 1157–1161 (2017).
Zheng, H., Liu, H., Hu, J. & Xu, F. Using a collagen heterotrimer to screen for cation-π interactions to stabilize triple helices. Chem. Phys. Lett. 715, 77–83 (2019).
Gauba, V. & Hartgerink, J. D. Surprisingly high stability of collagen ABC heterotrimer: evaluation of side chain charge pairs. J. Am. Chem. Soc. 129, 15034–15041 (2007).
Zheng, H. et al. How electrostatic networks modulate specificity and stability of collagen. Proc. Natl Acad. Sci. USA 115, 6207–6212 (2018).
Fallas, J. A. & Hartgerink, J. D. Computational design of self-assembling register-specific collagen heterotrimers. Nat. Commun. 3, 1087 (2012).
Acevedo-Jake, A. M., Clements, K. A. & Hartgerink, J. D. Synthetic, register-specific, AAB heterotrimers to investigate single point glycine mutations in Osteogenesis imperfecta. Biomacromolecules 17, 914–921 (2016).
Clements, K. A., Acevedo-Jake, A. M., Walker, D. R. & Hartgerink, J. D. Glycine substitutions in collagen heterotrimers alter triple helical assembly. Biomacromolecules 18, 617–624 (2017).
Giddu, S., Xu, F. & Nanda, V. Sequence recombination improves target specificity in a redesigned collagen peptide abc-type heterotrimer. Proteins 81, 386–393 (2013).
Parmar, A. S. et al. Design of net-charged abc-type collagen heterotrimers. J. Struct. Biol. 185, 163–167 (2014).
Egli, J., Erdmann, R. S., Schmidt, P. J. & Wennemers, H. Effect of N- and C-terminal functional groups on the stability of collagen triple helices. Chem. Commun. 53, 11036–11039 (2017).
Keshwani, N., Banerjee, S., Brodsky, B. & Makhatadze, G. I. The role of cross-chain ionic interactions for the stability of collagen model peptides. Biophys. J. 105, 1681–1688 (2013).
Acevedo-Jake, A. M., Jalan, A. A. & Hartgerink, J. D. Comparative NMR analysis of collagen triple helix organization from N- to C-termini. Biomacromolecules 16, 145–155 (2015).
O’Leary, L. R. Multi-Hierarchical Self-Assembly of collagen mimetic peptides into AAB type heterotrimers, nanofibers and hydrogels driven by charged pair interactions. PhD thesis, Rice University (2011).
Deprez, P., Doss-Pepe, E., Brodsky, B. & Inestrosa, N. C. Interaction of the collagen-like tail of asymmetric acetylcholinesterase with heparin depends on triple-helical conformation, sequence and stability. Biochem. J. 350, 283–290 (2000).
Shah, N. K., Sharma, M., Kirkpatrick, A., Ramshaw, J. A. M. & Brodsky, B. Gly-Gly-containing triplets of low stability adjacent to a type III collagen epitope. Biochemistry 36, 5878–5883 (1997).
Acknowledgements
This work was funded by the National Science Foundation (CHE 1709631) and the Robert A. Welch Foundation (C1557). I.-C.L. was supported by the Stauffer-Rothrock Fellowship. We thank the NMR and Drug Metabolism Core at Baylor College of Medicine for access to the 800 MHz NMR spectrometer and K. R. MacKenzie for assistance in acquiring NMR spectra.
Author information
Authors and Affiliations
Contributions
D.R.W., S.A.H.H., I.-C.L. and K.J.G. designed, synthesized and characterized deconvolution peptides. D.R.W. assembled the peptide library, designed, wrote and optimized SCEPTTr, performed and analysed NMR experiments, and co-wrote the manuscript. C.M.P. designed and synthesized the novel ABC system and characterized it using circular dichroism spectroscopy. J.D.H. supervised the research, evaluated all of the data and co-wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Correspondence and requests for materials should be addressed to J.D.H.
Peer review information Nature Chemistry thanks the anonymous reviewers for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–76, Discussion and Tables 1–7.
Supplementary Video 1
Movie to show NMR correlations.
Supplementary Software 1
Java application for SCEPTTr.
Rights and permissions
About this article
Cite this article
Walker, D.R., Hulgan, S.A.H., Peterson, C.M. et al. Predicting the stability of homotrimeric and heterotrimeric collagen helices. Nat. Chem. 13, 260–269 (2021). https://doi.org/10.1038/s41557-020-00626-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-020-00626-6
This article is cited by
-
Cis-trans isomerization of peptoid residues in the collagen triple-helix
Nature Communications (2023)
-
Hierarchical metal-peptide assemblies with chirality-encoded spiral architecture and catalytic activity
Science China Chemistry (2023)
-
De novo design of obligate ABC-type heterotrimeric proteins
Nature Structural & Molecular Biology (2022)